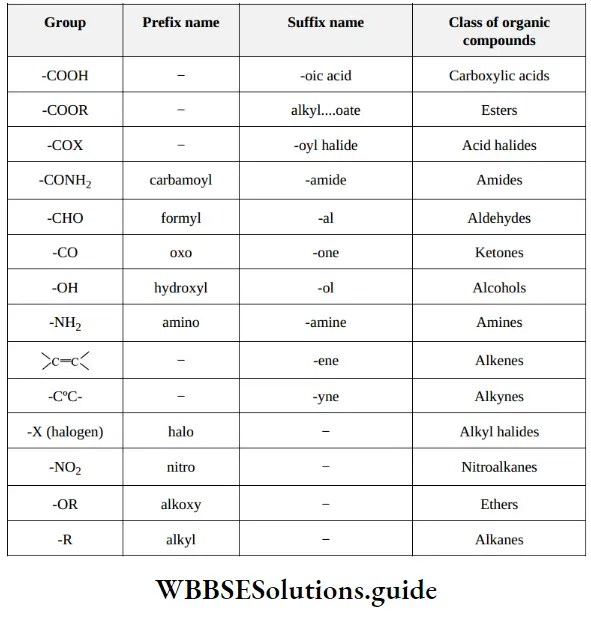
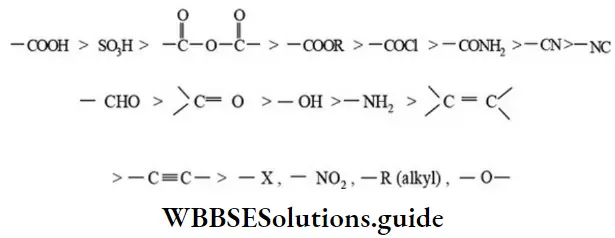
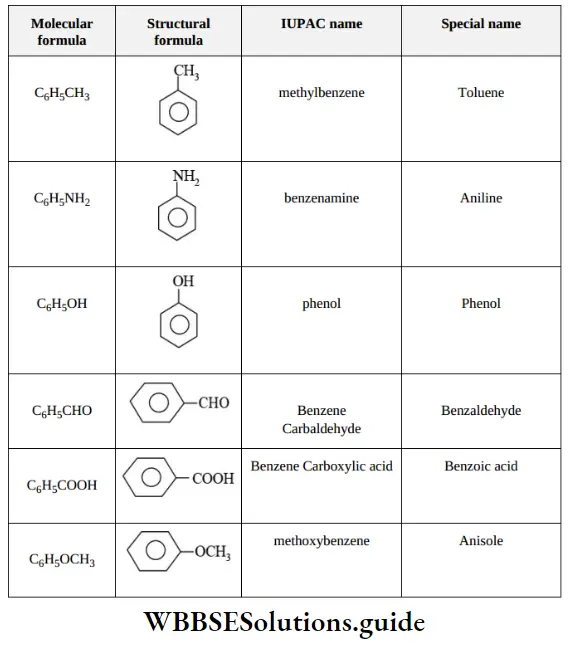
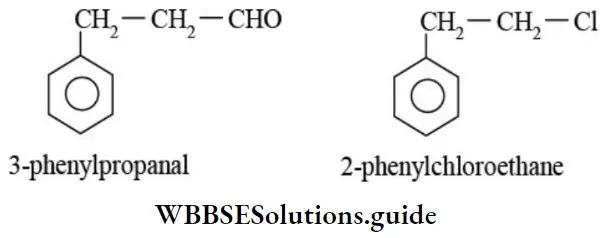
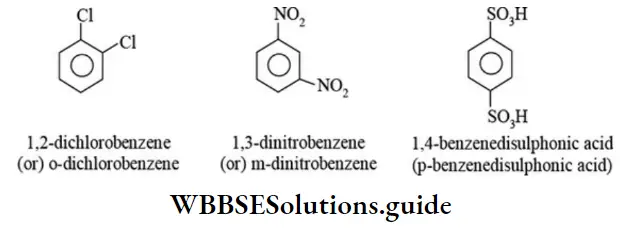
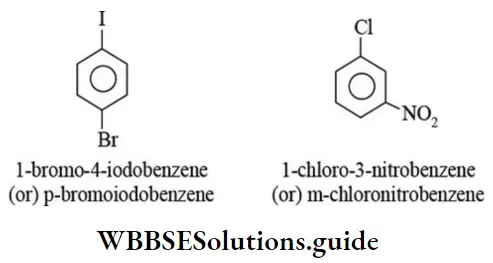
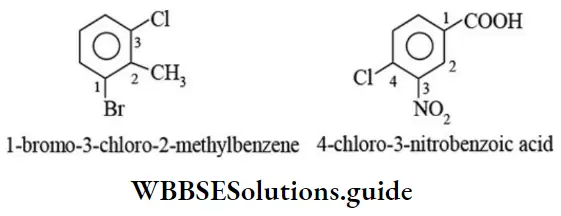
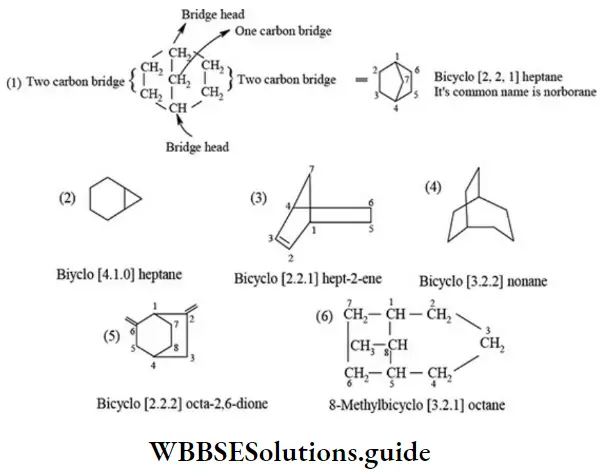
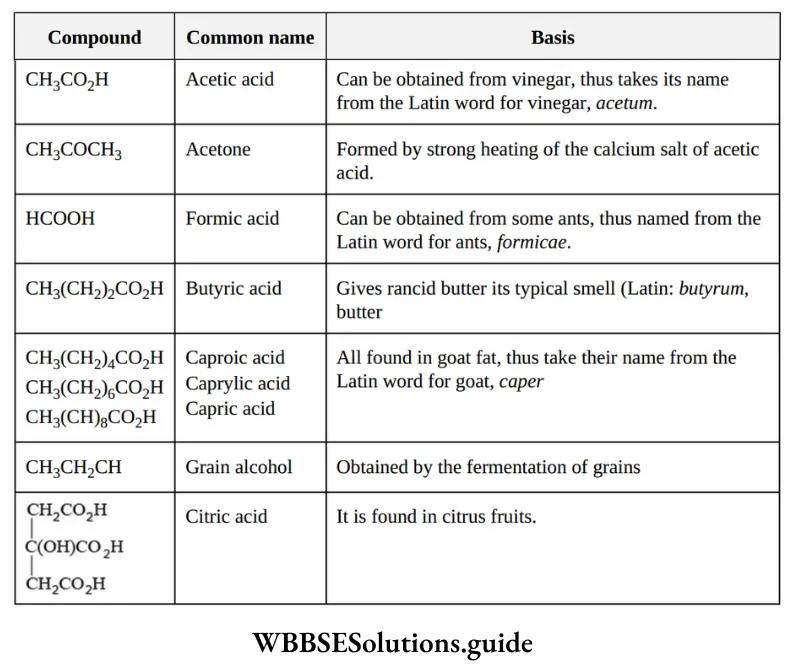
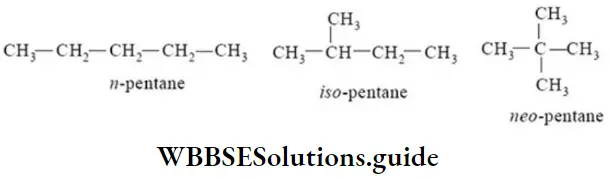
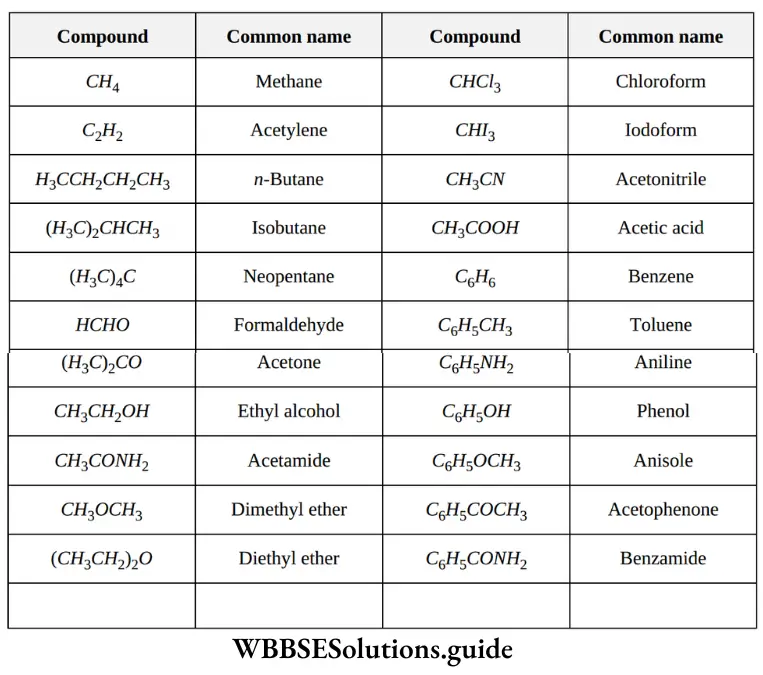
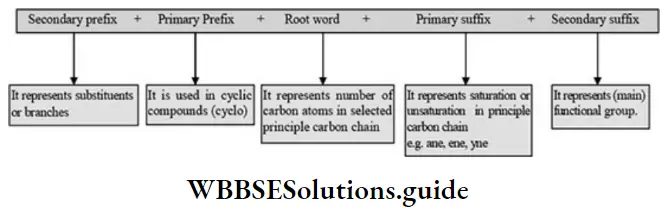
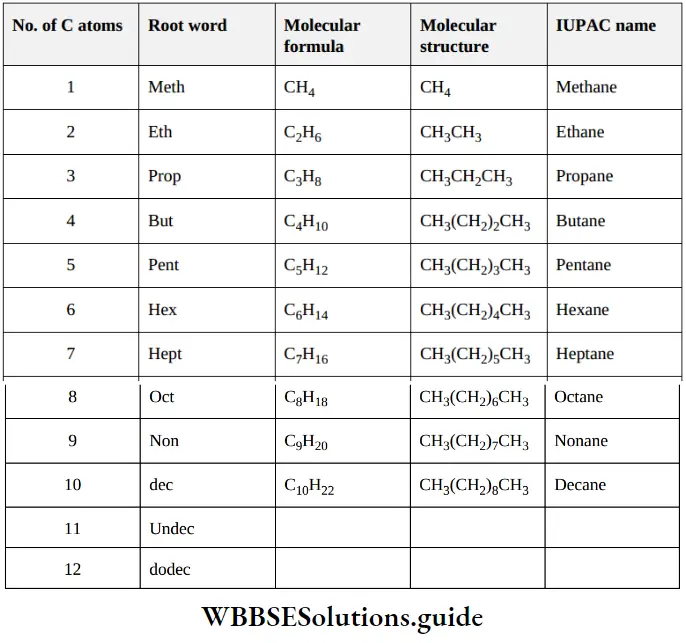
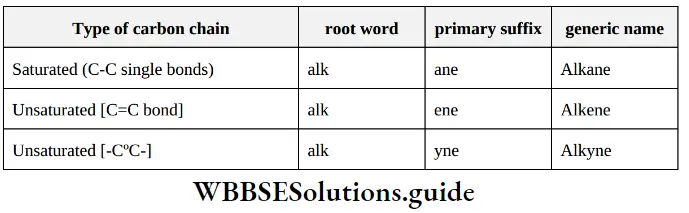
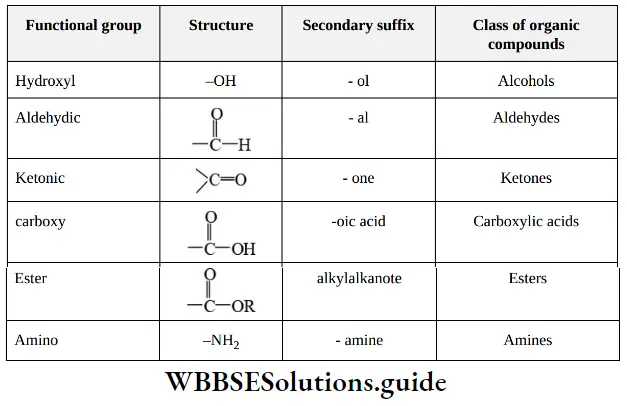
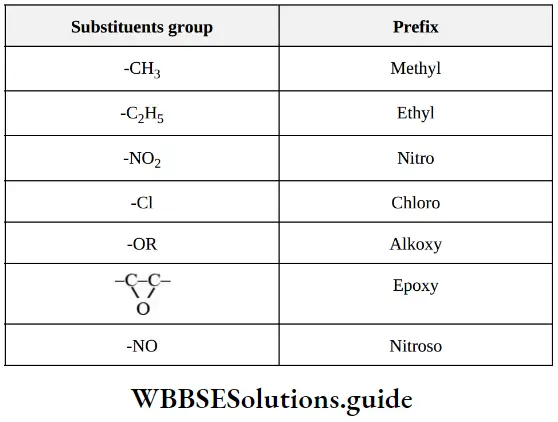
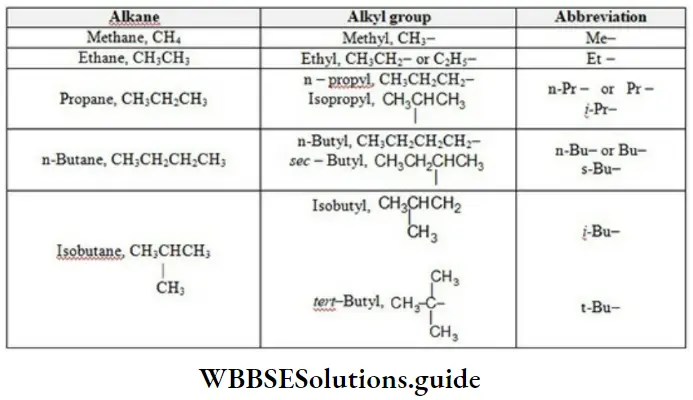
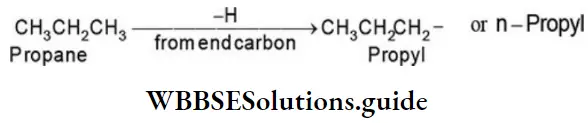
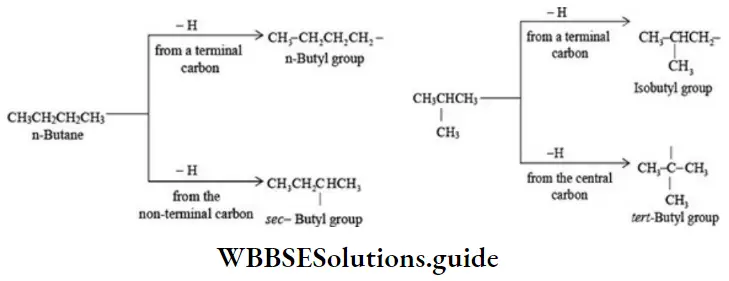
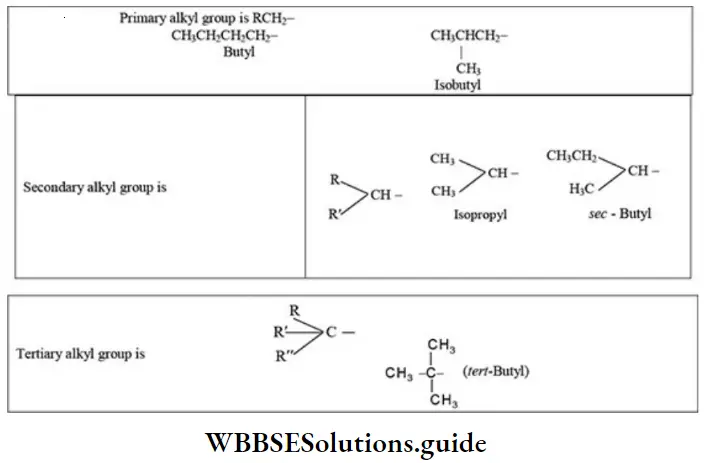
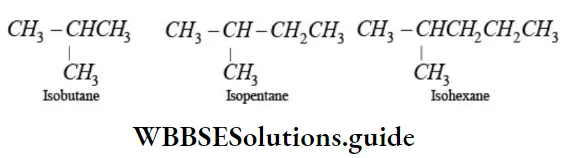
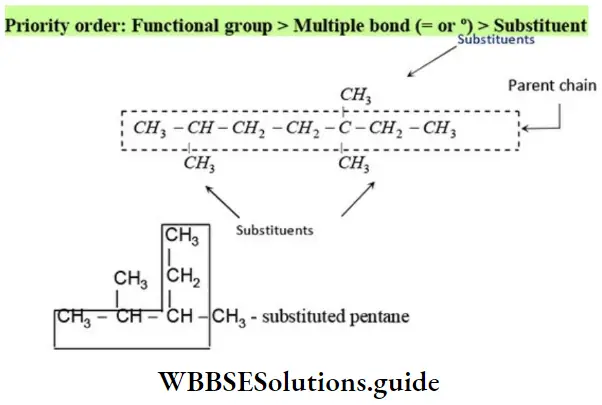
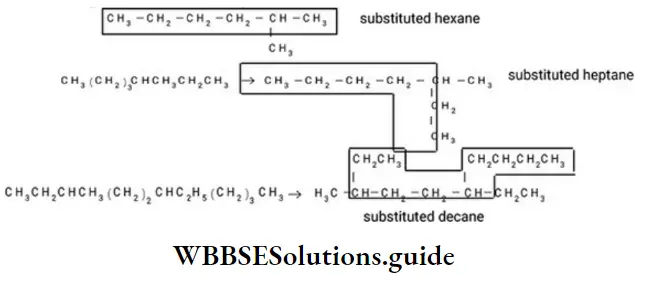
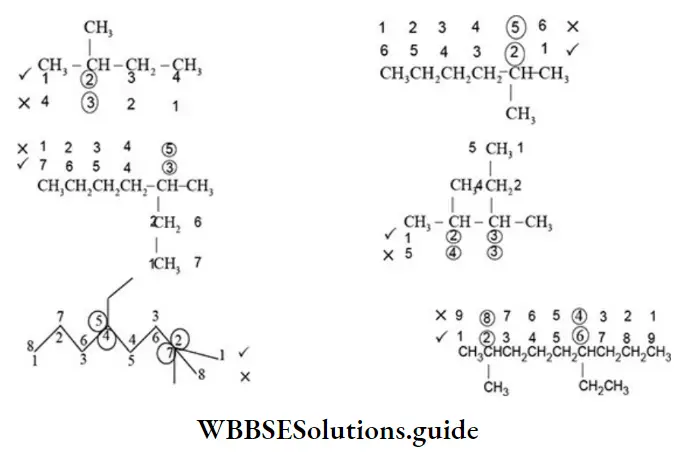
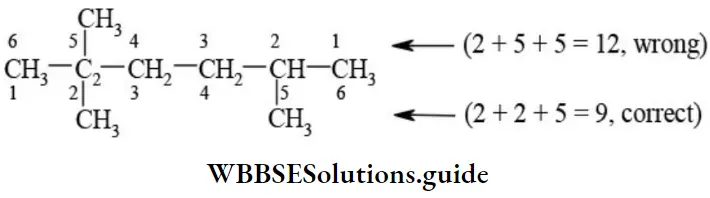
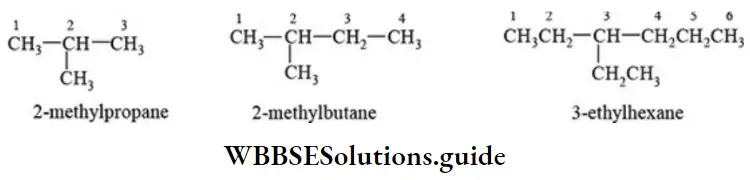
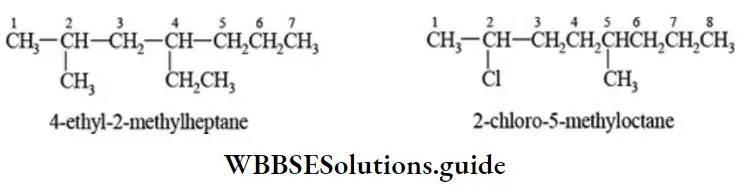
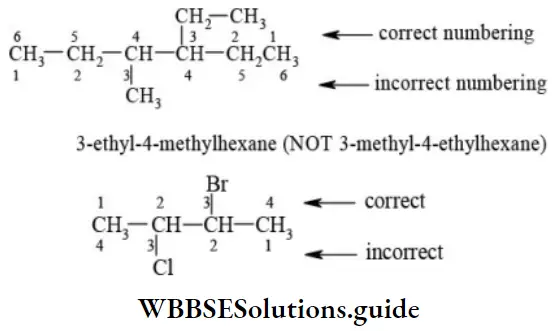
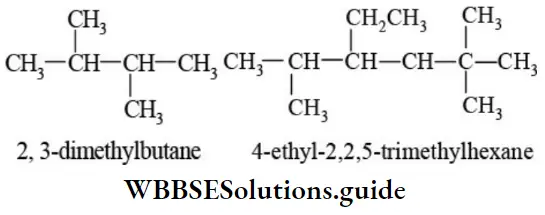
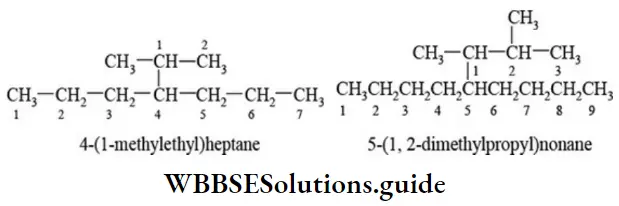
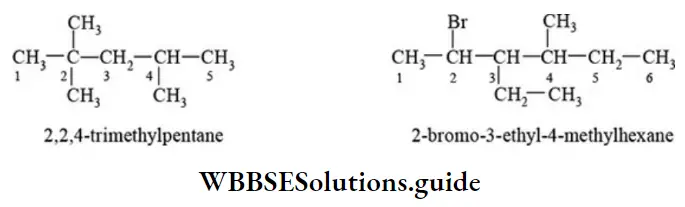
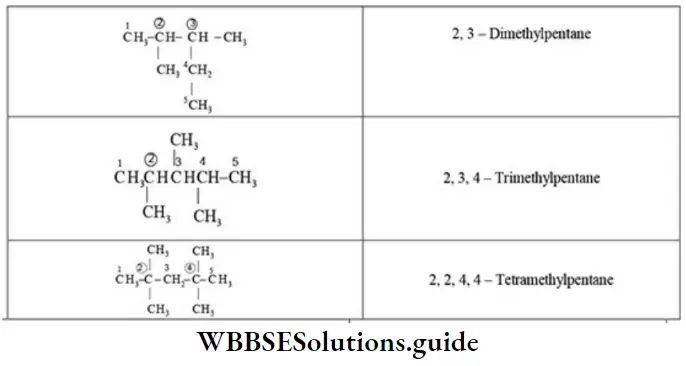
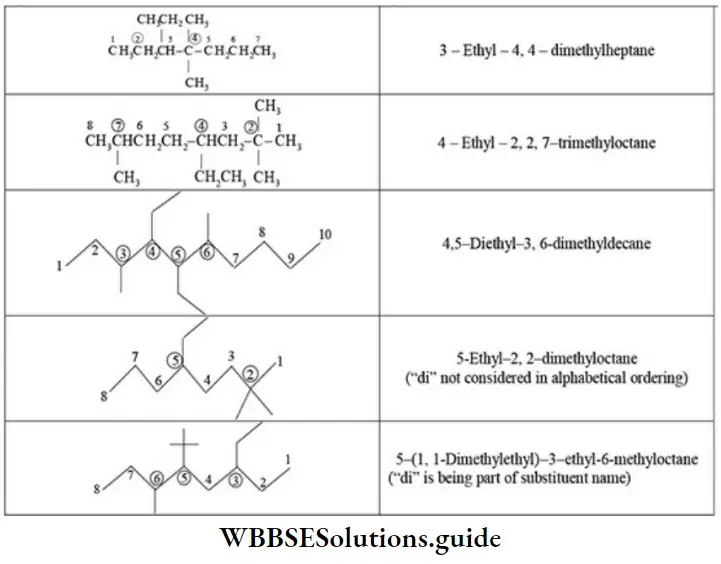
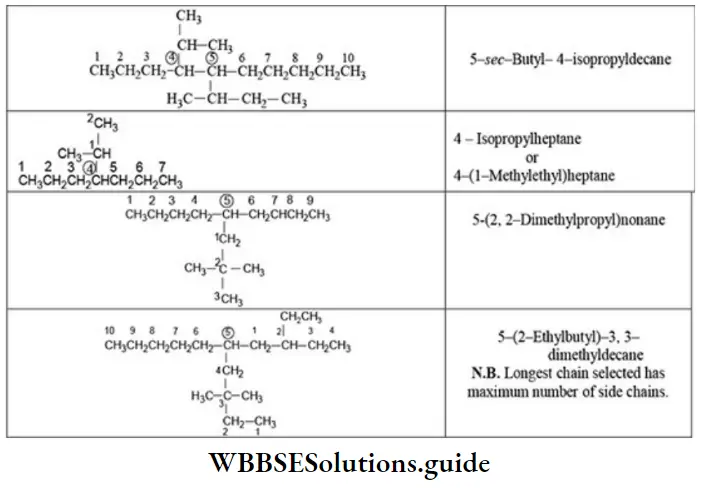
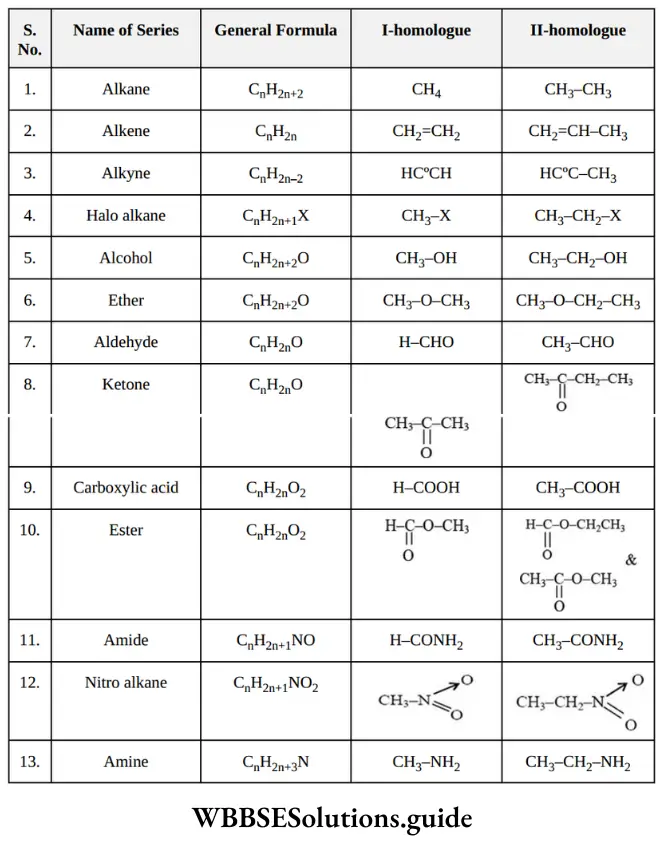
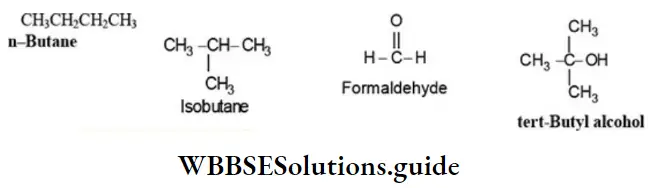
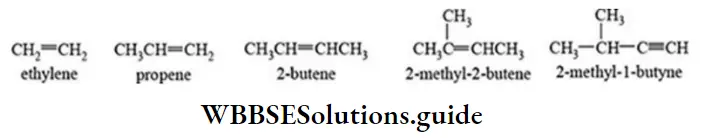
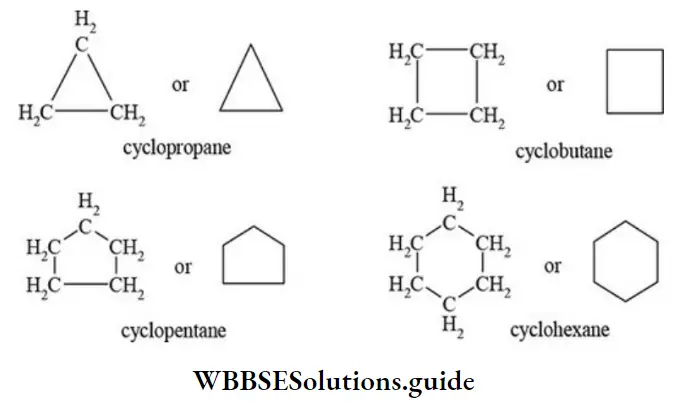
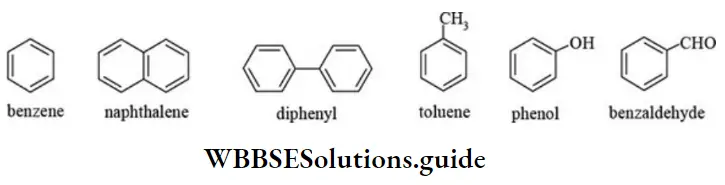
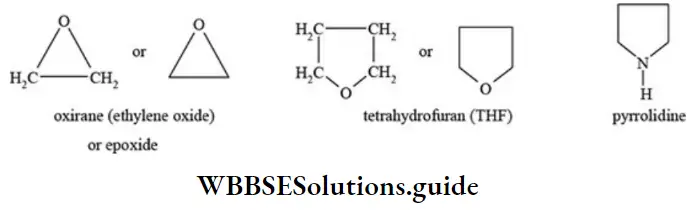
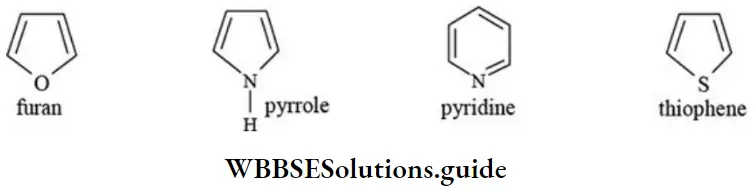
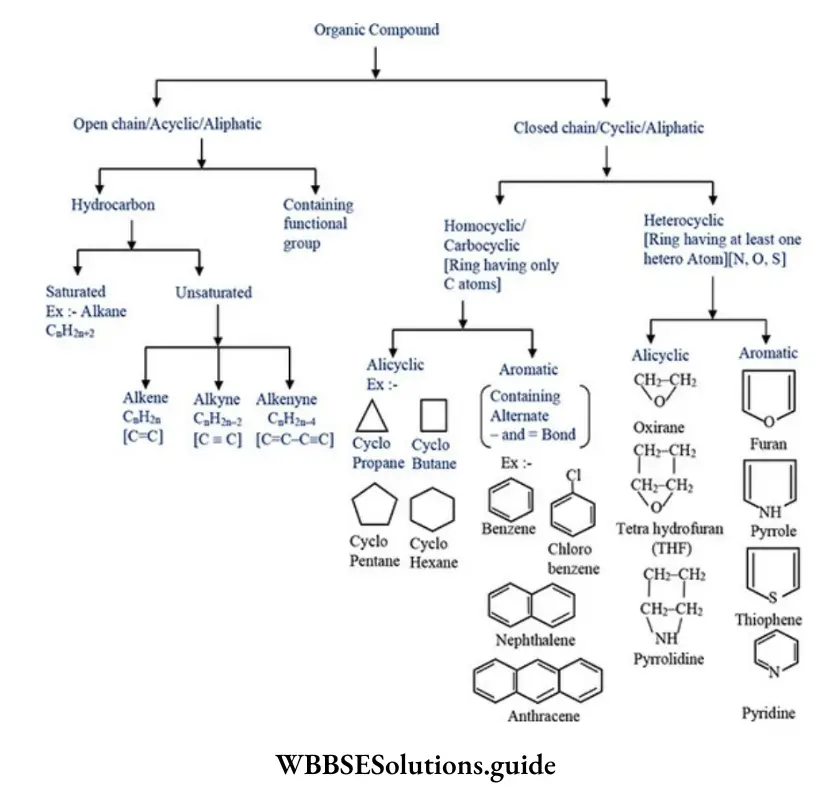
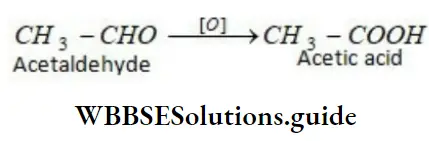
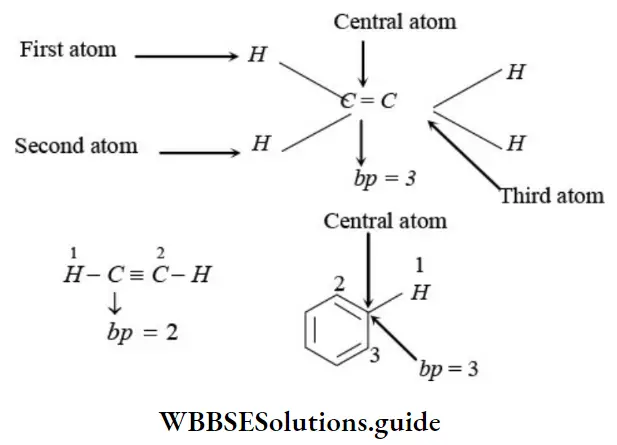
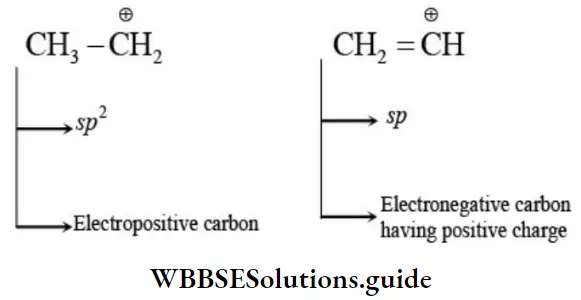
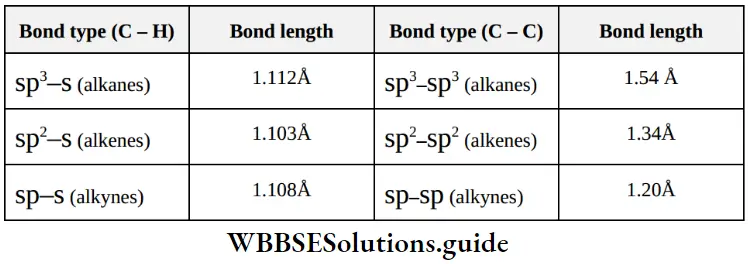
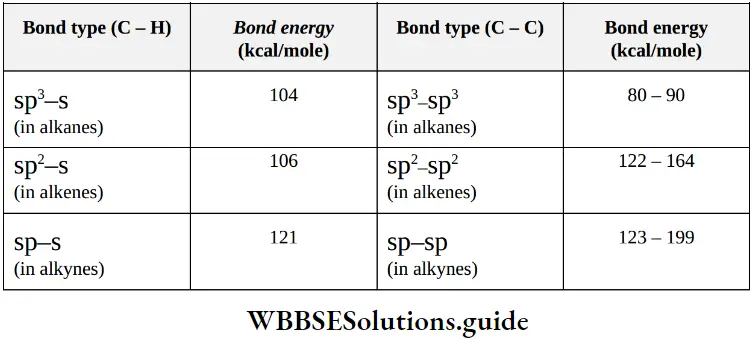
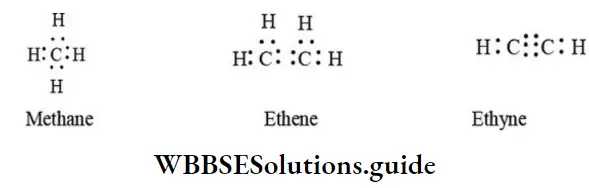
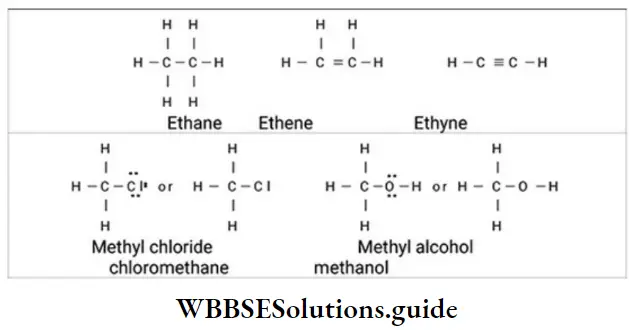
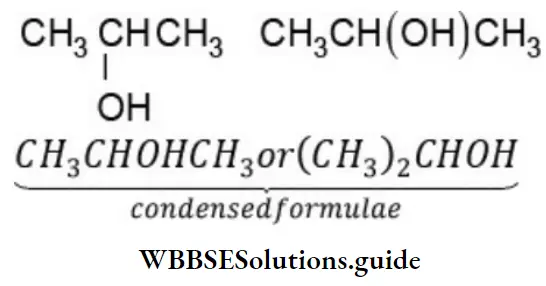
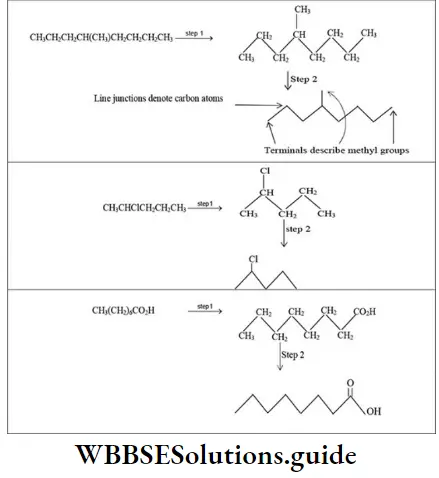
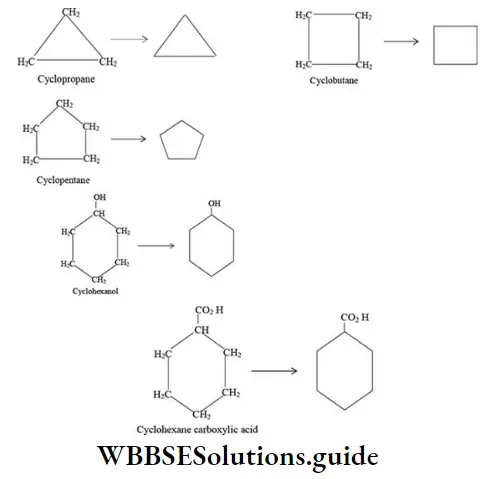
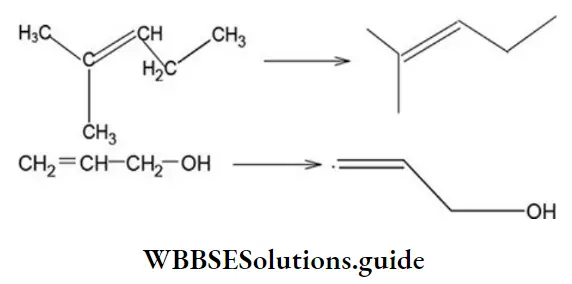
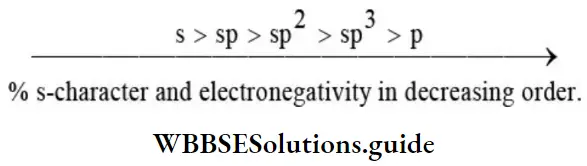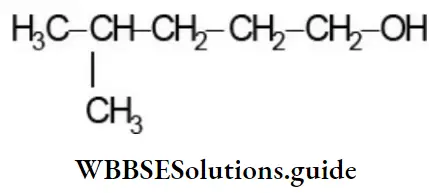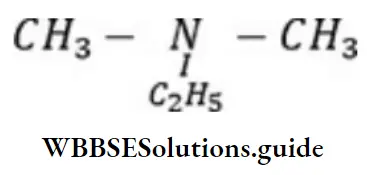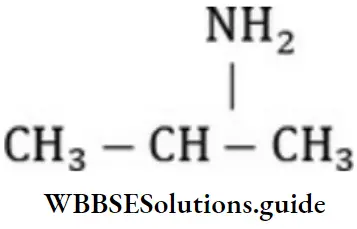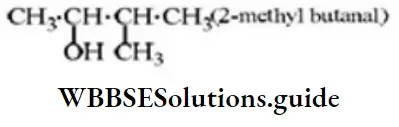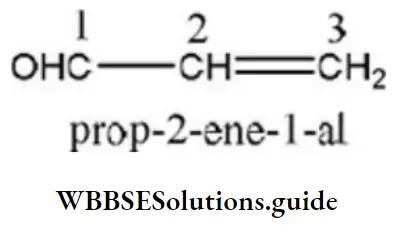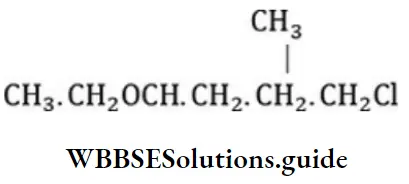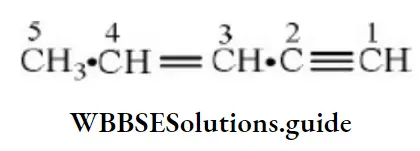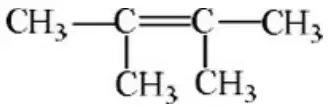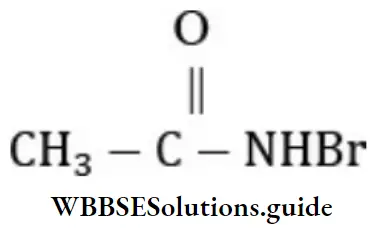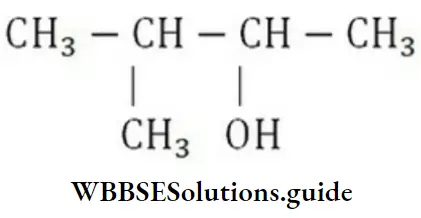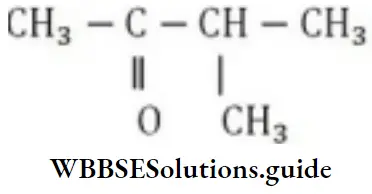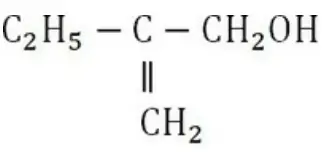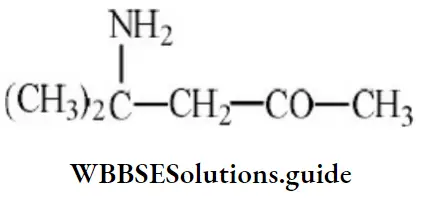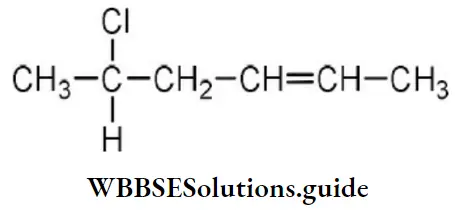Nomenclature Of Organic Compounds
Question 1. The IUPAC name of the compound (CH3)2CH-CH=CH-CHOH-CH3 is
- 5-methyl-hex-3-en-2-ol
- 2-methyl-hex-3-en-5-ol
- 2-hydroxy-5-nethyl-3-hexene
- 5-hydroxy-2-methyl-3-hexene
Answer: 1. 5-methyl-hex-3-en-2-ol
Solution: Follow IUPAC rules.
The IUPAC name of the compound (CH3)2CH-CH=CH-CHOH-CH3 is 5-methyl-hex-3-en-2-ol
Read And Learn More: NEET General Organic Chemistry Notes, Question And Answers
Question 2. Which of the following represents the condensed formula for pentanes?
- CH3(CH2)3CH3
- (CH3)3CCH3
- (CH3)2CHCH2 CH3
- All of these
Answer: 4. All of these
Solution: In condensed structural formula, parenthesis is used for the identical groups of atoms.
NEET organic chemistry nomenclature questions and answers
Question 3. Which is the most stable carbocation?
- isopropyl cation
- Triphenylmethyl cation
- Ethyl cation
- n-propyl cation
Answer: 2. Triphenylmethyl cation
Solution: Aryl carbonium ions are more stable than the alkyl carbonium ions. The order of stability of carbocation is
Triphenyl methyl >Diphenyl methyl> Benzyl>Allyl>30>2°>1°> methyl carbocation.
Question 4. Which of the following is phenyl ethanoate?
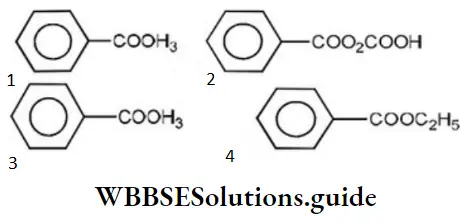
Answer: 3
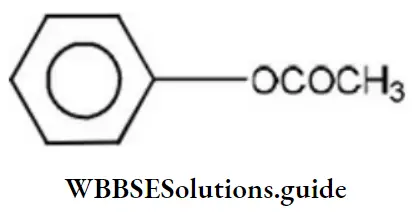
Solution: Esters are named by prefixing the name of the alkyl or aryl group (of OR’ part) before the name of the parent acid and changing the suffix “ic” acid to ate. Hence, the structure of phenyl ethanoate is
IUPAC nomenclature NEET previous year questions with solutions
Question 5. IUPAC name of CH2=CH—CH(CH3)2 is:
- 1,1-dimethyl-2-propene
- 3-methyl-1-butene
- 2-vinyl propane
- 1-isopropyl ethylene
Answer: 2. 3-methyl-1-butene
Solution: Follow IUPAC rules.

NEET chemistry nomenclature MCQs with answers
Question 6. In the structure, the number of carbon atoms are:
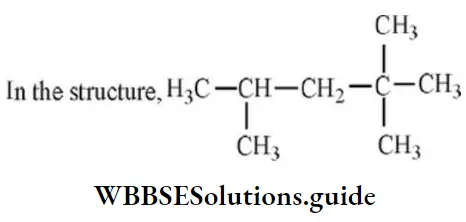
- One primary, two secondary and one tertiary
- Four primary, two tertiary and one secondary
- One primary, one secondary, one tertiary and one quaternary
- Five primary, one secondary, one tertiary and one quaternary
Answer: 4. Five primary, one secondary, one tertiary and one quaternary
Solution: A primary carbon is one which is joined to one carbon atom. A secondary carbon atom is joined to two carbon atoms and the tertiary carbon is attached to three carbon atoms. A quaternary carbon has all its four valencies attached to carbon atoms.
Question 7. IUPAC name of the following compound is
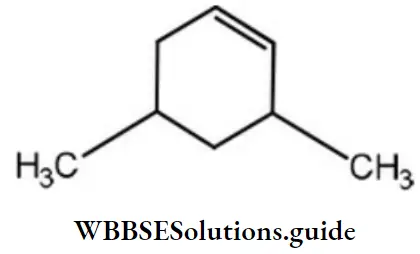
- 3, 5-dimethylcyclohexene
- 3, 5-dimethyl-1-cyclohexene
- 1, 5-dimethyl-5-cyclohexene
- 1, 3-dimethyl-5-cyclohexene
Answer: 1. 3, 5-methylcyclohexane
Solution: Its IUPAC name is 3,5-dimethylcyclohexene.
Important nomenclature questions for NEET organic chemistry
Question 8. IUPAC name ofis
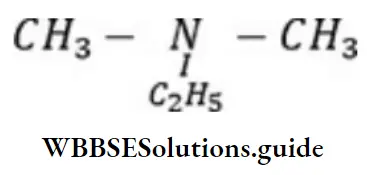
- N, N- dimethylethanolamine
- N-methyl, N-ethylmethanamine
- Dimethyl-ethylamine
- None of the above
Answer: 1. N, N- dimethylethanolamine
Solution: Follow IUPAC rules.
Question 9. The IUPAC name of CH3-CH=CH-C=CH is
- Pent-3-en-1-yne
- Pent-3-en-4-yne
- Pent-2-en-4-yne
- Pent-2-en-3-yne
Question 10.
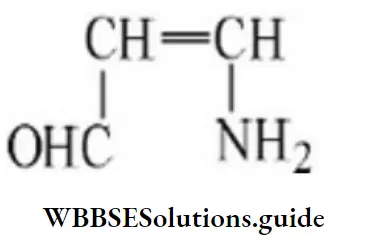
- 1-amino prop-2-enal
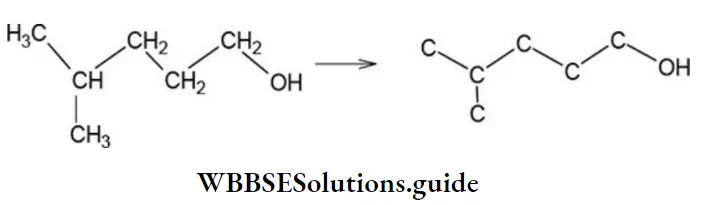
- 3-amino prop-2-enal
- 1-amino-2-formyl ethene
- 3-amino-1-oxoprop-2-ene
Answer: 2. 3-amino prop-2-enal
Solution: Follow IUPAC rules.
Question 11. The IUPAC name of an unsymmetrical ether with the molecular formula of C4H10O is
- Ethoxy propane
- Methoxy ethane
- Ethoxy ethane
- Methoxy propane
Answer: 4. Methoxy propane
Solution: Only two unsymmetrical ethers are possible from the formula C4H10O viz
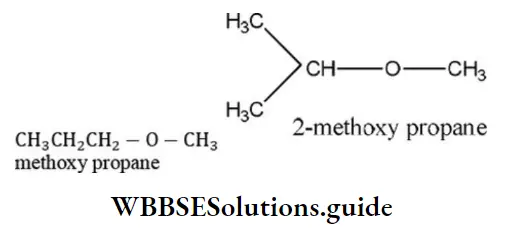
CH3 CH2 CH3-O-CH3
Question 12. IUPAC name of urea is:
- Diamino ketone
- 1-aminoethanamide
- 1-aminomethanamide
- amino acetamide
Question 13. IUPAC name of C6 H5 CN is:
- Phenyl nitrile
- Benzene nitrile
- Benzyl nitrile
- Phenyl cyanide
Answer: 2. Benzene nitrile
Solution: Follow IUPAC rules.
Question 14. The IUPAC name of the compound is
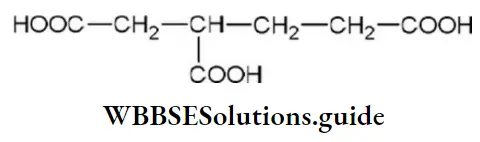
- 2(carboxymethyl)-pentane-1,5-dioic acid
- 3-carboxy hexane-1, 6-dioic acid
- Butane-1, 2, 4-tricarboxylic acid
- 4-carboxy hexane-1, 6-dioic acid
Answer: 2. 3-carboxy hexane-1, 6-dioic acid
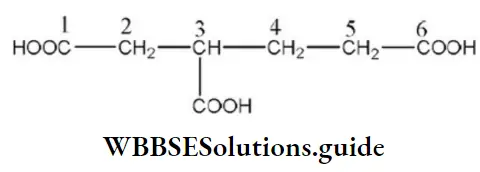
Solution: IUPAC name of the above compound is 3-carboxyhexane-1, 6-dioic acid.
NEET organic chemistry IUPAC naming practice questions
Question 15. IUPAC name of
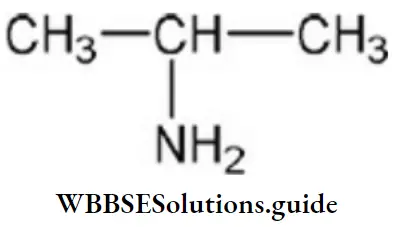
- Dimethyl amine
- 2-amino propane
- Isopropyl amine
- 2-propanamide
Answer: 4. 2-propanamide
Solution: 2-propanamide
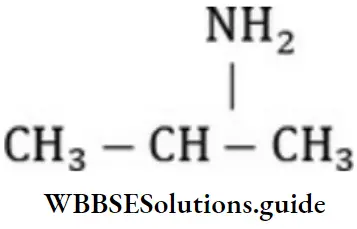
Question 16. The IUPAC name of
- 1, 1-dimethyl-1, 3-butanediol
- 2-methyl-2, 4-pentanediol
- 4-methyl-2, 4-pentanediol
- l, 3, 3-trimethyl-1, 3-propanediol
Answer: 2. 2-methyl-2, 4-pentanediol
Solution: Follow IUPAC rules.
Question 17. The IUPAC name of the compound is
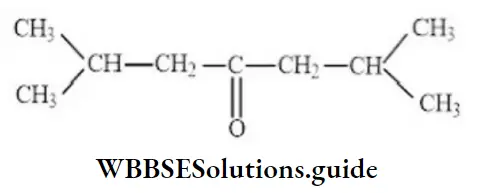
- 2,4-dimethylhexanone-3
- 2,6-dimethylheptanone-4
- 2,6-dimethylhexanone-4
- 2,6-dimethylheptanone-5
Answer: 2. 2,6-dimethylheptanone-4
Solution: Follow IUPAC rules.
Question 18. Lactic acid is:
- Propionic acid
- ^-hydroxy propanoic acid
- a-hydroxy propanoic acid
- None of the above
Answer: 3. a-hydroxy propanoic acid
Solution: Lactic acid is also called as a-hydroxy propionic acid or 2-hydroxy propanoic acid.
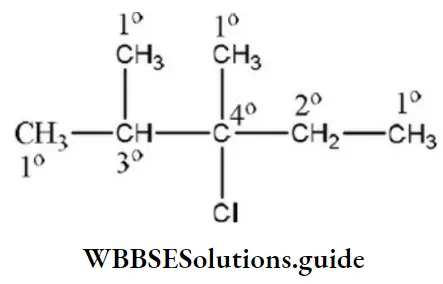
Question 19. The compound which contains all the four 1°, 2°, 3° and 4° carbon atoms is
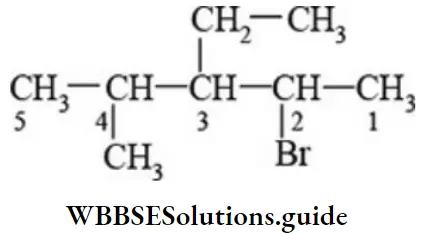
- 2, 3-dimethyl pentane
- 3-chloro-2, 3-dimethyl pentane
- 2, 3, 4-trimethylpentane
- 3, 3-dimethyl pentane
Answer: 2. 3-chloro-2, 3-dimethyl pentane
Solution: 3-chloro-2, 3-dimethyl pentane contains all the four 1°,2°, 3° and 4° carbon atoms.
Question 20. The name formic acid was given for HCOOH because it was prepared from:
- Acetum
- Ant
- Wood
- Oxalis plant
Answer: 2. Ant
Solution: Formic acid was obtained from ant (Formica in Greek). This is the trivial name for HCOOH.
Question 21. The IUPAC name of the compound having the formula CH= C — CH=CH2 is:
- 1-butene-3-yne
- 3-butene-1-yne
- 1-butyn-3-ene
- But-1-yne-3-ene
Answer: 1. 1-butene-3-yne
Solution: Follow IUPAC nomenclature.
Question 22. The IUPAC name of the below compound is
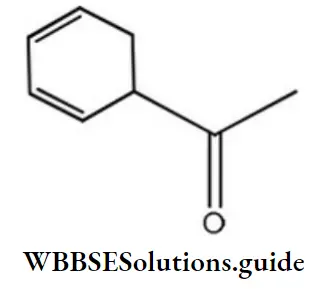
- 1- cyclohexane-2,4-dienylethanone
- 3- cyclohexane-2,4-dienylethanone
- 1- cyclohexane-3,5-dienylethanone
- 3- cyclohexane-3,5-dienylethanone
Answer: 1. 1- cyclohexane-2,4-dienylethanone
Solution: Follow IUPAC nomenclature.
Question 23. The IUPAC name of the below compound is
- 1-methyl-1-amino propane
- 2-amino butane
- 2-methyl-3-amino propane
- None of the above
Answer: 2. 2-amino butane
Solution: Follow IUPAC rules.
Question 24. The correct IUPAC name of (C2 H5)4 C is:
- Tetraethyl methane
- 2-ethyl pentane
- 3,3-diethyl pentane
- None of these
Answer: 3. 3,3-diethyl pentane
Solution: Follow IUPAC rules.
Nomenclature of organic compounds NEET question bank
Question 25. Which IUPAC name is wrong?
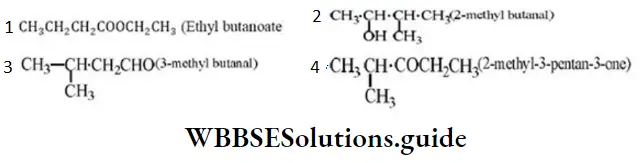
Answer: 3
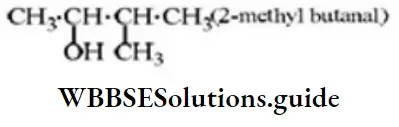
Solution: It is 3-methyl butan-2-ol.
Question 26. The structural formula of 2,2,3-trimethyl hexane is:
Answer: 3
Solution: Follow IUPAC rules.
Question 27. IUPAC name of acraldehyde is
- But-3-en-1-al
- Propenyl aldehyde
- But-2-ene-1-al
- Prop-2-en-1-al
Answer: 4. Prop-2-en-1-al
Solution: IUPAC name of acraldehyde
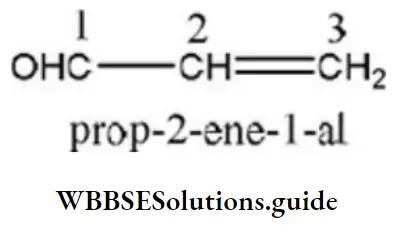
Question 28. The IUPAC name of the compound is
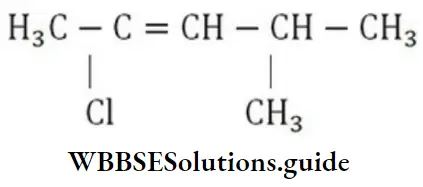
- 2-chloro-4-methyl-2-pentene
- 4-chloro-2-methyl-3-pentene
- 4-methyl-2-chloro-2-pentene
- 2-chloro-4, 4-dimethyl-2-butene
Answer: 1. 2-chloro-4-methyl-2-penton
Solution: Follow IUPAC rules.
Question 29. 2-methyl pent-3-ene is achiral because it has:
- A centre of symmetry
- A plane of symmetry
- Symmetry at C2 carbon
- Both centre and a plane of symmetry
Answer: 4. Both centre and a plane of symmetry
Solution: If the structure of the compound is drawn then we find that a plane of symmetry is present in this compound. The two halves are equal hence the compound does not have a chiral point, in addition to this it has a Centre. The two sides of the compound on the different sides of the centre atom are similar. Both the reasons are present which signifies that the molecule is achiral.
Question 30. The IUPAC name of the compound is
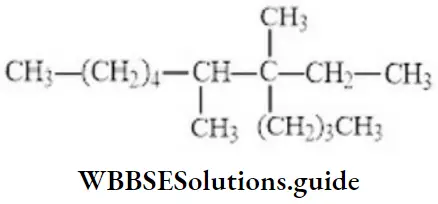
- 3,4-dimethyl-3-n-propyl nonane
- 4-ethyl-4,5-dimethyl decane
- 6,7-dimethyl-7-n-propyl nonane
- 6,7-dimethyl-7-ethylene
Answer: 2. 4-ethyl-4,5-dimethyl decane
Solution: Follow IUPAC rules.
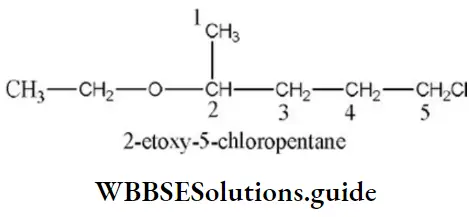
Question 31. Give the correct IUPAC name for
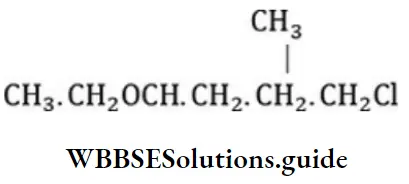
- 2-ethoxy-5-chloroethane
- l-chloro-4-ethoxy-4-methyl butane
- 1-chloro-4-ethoxy pentane
- Ethyl-1-chloropentylether
Answer: 1. 2-ethoxy-5-chloroethane
Solution: According to the IUPAC system, ether is named as alkoxy alkane. The larger alkyl group forms the parent chain while the lower alkyl group takes in ethereal oxygen and forms a part of the alkoxy group.
Question 32. The IUPAC name of the compound, CH3 CH = CHC = CH is:
- Pent-4-yn-2-ene
- Pent-3-en-1-yne
- Pent-2-en-4-yne
- Pent-1-yn-3-ene
Answer: 2. Pent-3-en-1-yne
Solution: Follow IUPAC rules.
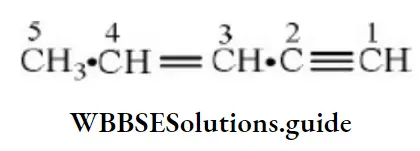
Question 33. The IUPAC name of C6 H5 COCl is
- Benzoyl chloride
- Benzene chloro ketone
- Benzene carbonyl chloride
- Chloro phenyl ketone
Answer: 1. Benzoyl chloride
Solution: Follow IUPAC rules.
NEET chemistry IUPAC nomenclature quiz with answers
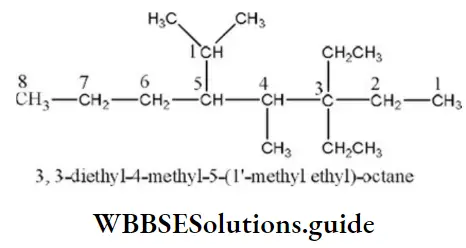
Question 34. IUPAC name of the compound
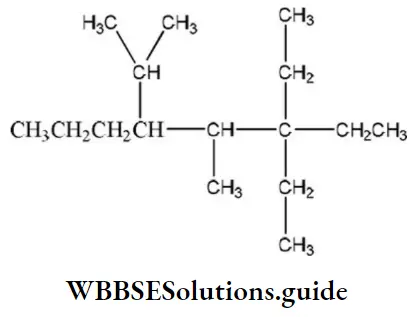
- 5- methyl-4-isopropyl-6, 6’diethyloctane
- 3, 3-dimethyl, 3-ethyl-5- isopropyl octane
- 3, 3-diethyl-4-methyl-5-(1,1-dimethyl) octane
- 3, 3- diethyl-4-methyl-5-(1’-methylethyl) octane
Answer: 4. 3, 3- diethyl-4-methyl-5-(1’-methyl ethyl) octane
Solution: Select the longest possible carbon atom chain, number it and name the compound according to IUPAC rules.
Question 35. The IUPAC name of is
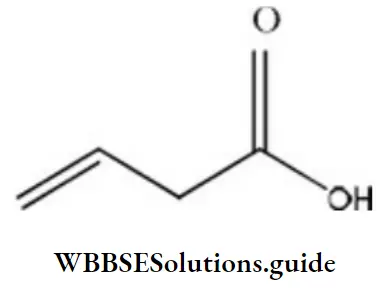
- But-3-enoic acid
- But-1-enoic acid
- Pent-4-enoic acid
- Prop-2-enoic acid
Question 36. The correct IUPAC name of the below molecule is
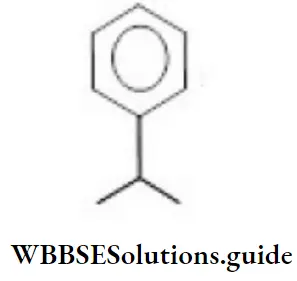
- Isopropyl benzene
- Cumene
- Phenyl isopropanol
- 2-phenyl propane
Answer: 4. 2-phenyl propane
Solution: Follow IUPAC rules.
Question 37. The IUPAC name of the compound is
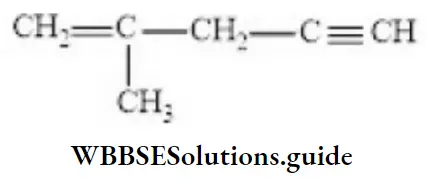
- 2-methyl pent-1-en-4-yne
- 4-methyl pent-4-en-1-yne
- 2-methyl pent-2-en-4-yne
- 4-methyl pent-1-en-4-yne
Answer: 1. 2-methylpent-1-en-4-yne
Solution: Follow IUPAC rules.
Question 38. Freon-114 is an organic compound. k is chemically called 1,2dichlorotetrafluoroethane. Its correct structural formula is:
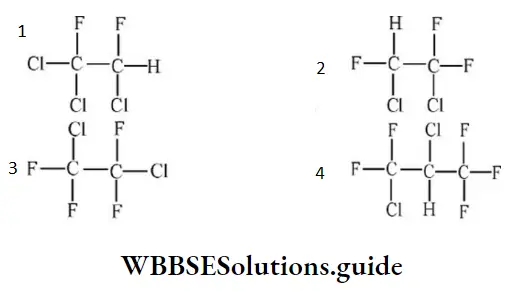
Answer: 3
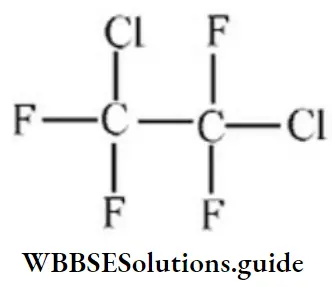
Solution: Follow IUPAC rules.
Question 39. The IUPAC name of the following compound is
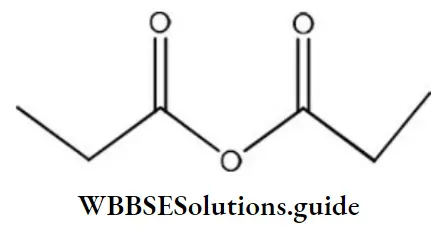
- Propionic anhydride
- Di propanoic anhydride
- Ethoxy propanoic acid
- Propanoic anhydride
Answer: 4. Propanoic anhydride
Solution: In the IUPAC system, anhydrides are named as alkanoic anhydride.
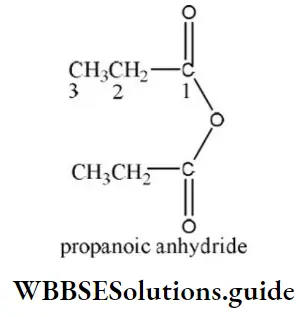
Question 40. IUPAC name of the compound is
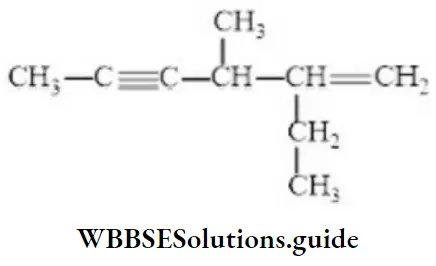
- 2-ethyl-3-methyl-hexa-l-en-4-yne
- 5-ethyl-4-methyl-hexa-2-yn-5-ene
- 3-methylene-4-methylhepta-5-yne
- 5-methylene-5-ethyl-4-methylhepta-2-yne
- Answer: 1. 2-ethyl-3-methyl-hexa-l-en-4-yne
Solution:-do-
Question 41. Bicyclo (1,1,0) butane is
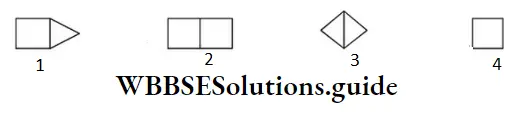
Answer: 3

Its IUPAC name is bicyclo [1,1,0] butane.
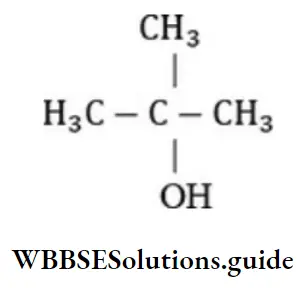
Question 42. t-butyl alcohol is
- 2-methyl propane-2-ol
- 2-methyl propane-1-ol
- 3-methyl butane-1-ol
- 3-methyl butane-2-ol
Answer: 1. 2-methyl propan-2-ol
Solution: 2-methyl propan-2-ol is tert-butyl alcohol.
Question 43. IUPAC name of C6 H5 COCl is:
- Benzal chloride
- Benzenechloro ketone
- Benzene carbonyl chloride
- Chloro phenyl ketone
Answer: 3. Benzene carbonyl chloride
Solution: Follow IUPAC rules.
Question 44. The IUPAC name for tertiary butyl iodide is
- 4-iodo butane
- 2-iodo butane
- 1-iodo-3-methyl propane
- 2-iodo-2-methylpropane
Answer: 4. 2-iodo-2-methylpropane
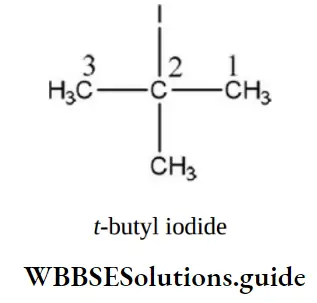
Solution: Its IUPAC name is 2-iodo-2-methyl propane.
Question 45. The correct structure of dimethyl butane is:
- CH3CH3—C=C—CH2 CH3
- (CH3)3C —C —C≡CH
- CH3—C=CCH(CH3)2
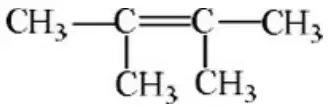
Answer: 2. (CH3)3C —C —C≡CH
Question 46. Glyoxal is
- CH2OH-CH2OH
- CHO-CH2OH
- COOH-CO-COOH
- CHO-CHO
Answer: 4. CHO-CHO
Solution:
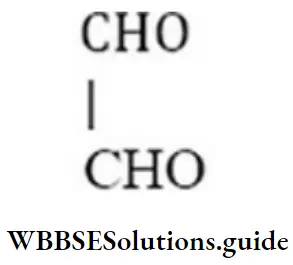
Question 47. Give the IUPAC name for,
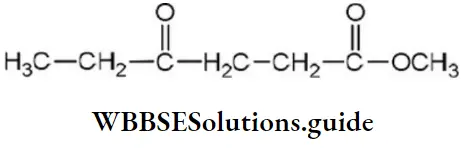
- Ethyl-4- oxo heptanoate
- Methyl-4- oxo heptanoate
- Ethyl-4- oxo hexanoate
- Methyl 4- oxo hexanoate
Answer: 4. Methyl 4- oxo hexanoate
Solution: Follow IUPAC rules.
Question 48. The IUPAC name of the compound,
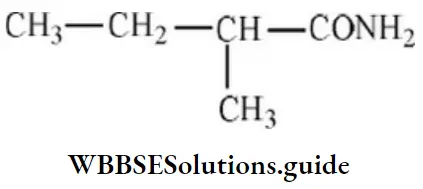
- 2-ethylbutanamide
- 2-methylbutanamide
- 1-amino-2-methylpropane
- None of the above
Answer: 2. 2-methylbutanamide
Solution: Follow IUPAC rules.
Question 49. The IUPAC name of CH3 COCH(CH3)2 is
- Isopropyl methyl ketone
- 2-methyl-3-butanone
- 4-methyl isopropyl ketone
- 3-methyl-2-butanone
Answer: 4. 3-methyl-2-butanone
Solution:
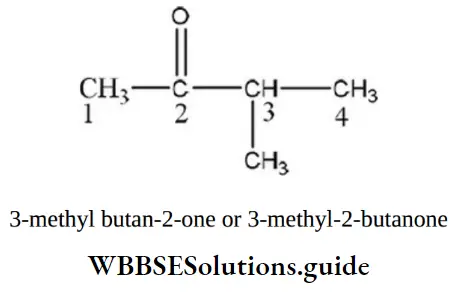
The keto functional group is given priority.
Question 50. The IUPAC name of the following compound is
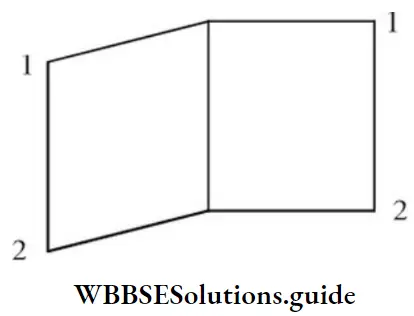
- Bicyclo [2,2,0] octane
- Bicyclo [0,2,2] hexane
- Bicyclo [2,1,1] hexane
- Bicyclo [2,2,0] hexane
Answer: 4. Bicyclo [2,2,0] hexane
Solution: Hence, the correct IUPAC name is bicyclo [2,2,0] hexane.
Nomenclature of organic compounds NEET sample paper
Question 51. The IUPAC name of the compound,
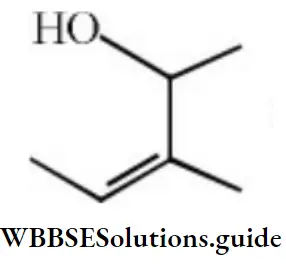
- 1,2-dimethyl-2-butenol
- 3-methylpent-3-en-2-ol
- 3,4-dimethyl-2-buten-4-ol
- 2,3-dimethyl-3-pentenol
Answer: 2. 3-methyl pent-3-en-2-ol
Solution: Follow IUPAC rules.
Question 52. IUPAC name of (CH3)3 CCl is
- n-butyl chloride
- 3-chloro butane
- 2-chloro 2-methyl propane
- t-butyl chloride
Answer: 3. 2-chloro 2-methyl propane
Solution: Follow IUPAC rules.
Question 53. What is the correct IUPAC name of the compound?
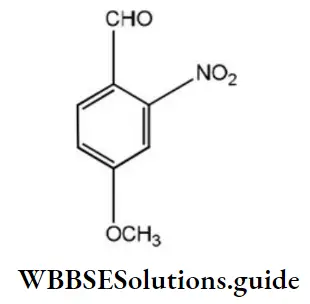
- 4-methoxy-2-nitrobenzaldehyde
- 4-formyl-3-nitro anisole
- 4-methoxy-6-nitrobenzaldehyde
- 2-formyl-5-methoxy nitrobenzene
Answer: 1. 4-methoxy-2-nitrobenzaldehyde
Solution: Follow IUPAC rules.
Question 54. What is the correct IUPAC name of the compound?
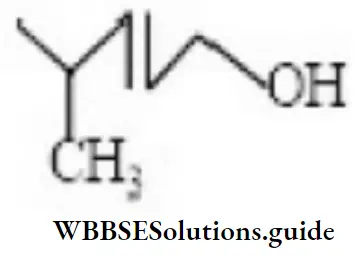
- 5-methyl hexanol
- 2-methyl hexanol
- 2-methylhex-3-enol
- 4-methyl pent-2-enol
Answer: 4. 4-methyl pent-2-enol
Solution: —do—
Question 55. The IUPAC name of the compound is:
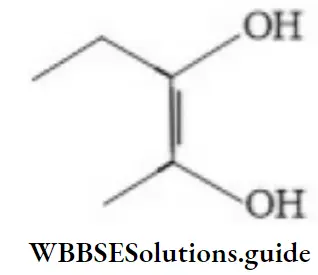
- But-2-ene-2,3-diol
- Pent-2-ene-2,3-diol
- 2-methylbut-2-ene-2,3-diol
- Hex-2-ene-2,3-diol
Answer: 2. Pent-2-ene-2,3-diol
Solution: Follow IUPAC rules.
Question 56. Which nomenclature in IUPAC is incorrect?
- Pentyne-3
- Pentyne-2
- Hexyne-3
- None of these
Answer: 1. Pentyne-3
Solution: CH3 C = CCH2 CH3; It is always pentyne-2 and never pentyne-3.
Question 57. The IUPAC name of the compound CH3 CONHBr is
- 1-bromoacetate
- Ethanoylbromide
- N-Bromoethanamide
- None of these
Answer: 3. N-Bromoethanamide
Solution: Follow IUPAC rules.
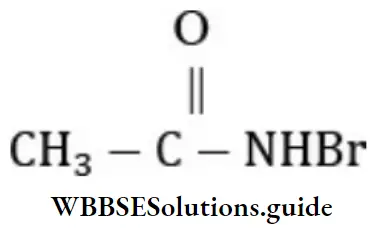
Question 58. The IUPAC name of the compound is:
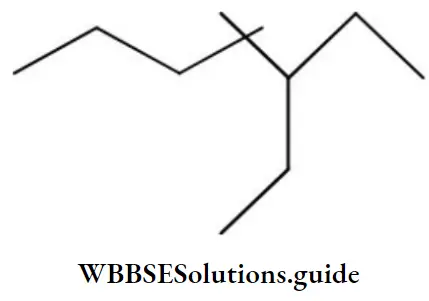
- 3-ethyl-4,4-dimethyl heptane
- 1,1-diethyl-2,2-dimethyl pentane
- 4,4-dimethyl-5,5-diethyl pentane
- 5,5-diethyl-4, 4-dimethyl pentane
Answer: 1. 3-ethyl-4,4-dimethyl heptane
Solution: Follow IUPAC rules.
Question 59. The correct IUPAC name of the compound is:
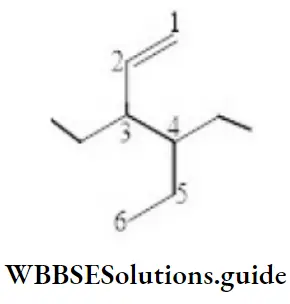
- 3-(1-ethyl propyl) hex-1-ene
- 4-Ethyl-3-propyl hex-1-ene
- 3-Ethyl-4-ethenyl heptane
- 3-Ethyl-4-propyl hex-5-ene
Answer: 2. 4-Ethyl-3-propyl hex-1-ene
Solution: Follow IUPAC rules.
Question 60. The IUPAC name of the following compound is
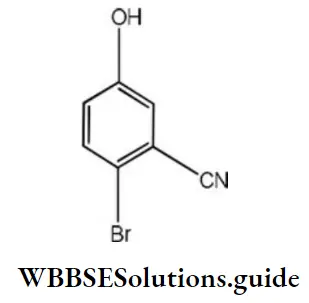
- 4-bromo-3-cynophenoal
- 2-bromo-5-hydroxy benzonitrile
- 2-cyano-4-hydroxybromobenzene
- 6-bromo-3-hydroxy benzonitrile
Answer: 2. 2-bromo-5-hydroxy benzonitrile
Solution: The Cyano group has the highest priority therefore, parent name must be benzonitrile. Br occurs at 2- position, and hydroxyl at 3-position, hence the IUPAC name is 2-bromo-5-hydroxy benzonitrile.
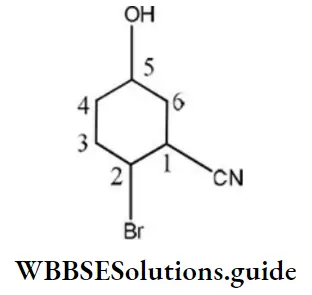
Question 61. IUPAC name of the compound is
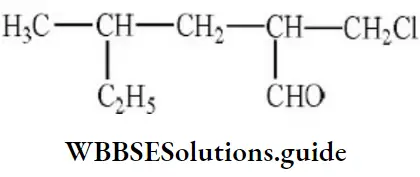
- 2-chloromethyl-4-methyl-hexanal
- 1-chloro-4-ethyl-2-pentanal
- 1-chloro-4-methyl-2-hexanal
- 1-chloro-2-aldo-4-methyl hexane
Answer: 1. 2-chloromethyl-4-methyl-hexanal
Solution: Follow IUPAC rules.
Question 62. Which one is not in the IUPAC system?
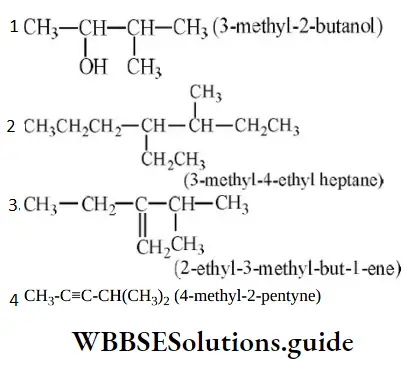
Answer: 2
Solution: It should be 4-ethyl-3-methyl heptane.
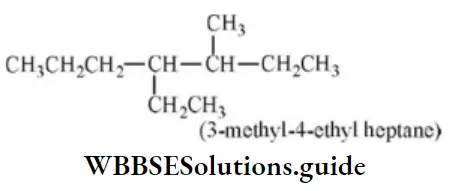
Question 63. The correct IUPAC name is
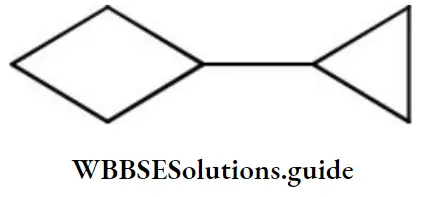
- 1- cyclopropyl Cyclobutane
- 1, 1-cyclobutane
- 1- cyclobutane-1- cyclopropane
- None of the above
Answer: 1. 1- cyclopropyl Cyclobutane
Solution: Follow IUPAC rules.
Question 64. The IUPAC name of the compound
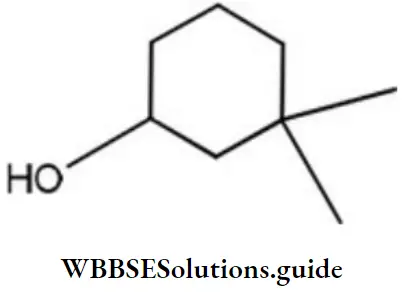
- 3, 3-dimethyl-1-hydroxy cyclohexane
- 1, 1-dimethyl-3- hydroxy cyclohexane
- 3, 3- dimethy-1- cyclohexanol
- 1,1-dimethyl-3-cyclohexanol
Answer: 3. 3, 3- dimethyl-1- cyclohexanol
Solution: Carbon with – OH group is given C1 thus it is 3, 3-dimethyl-1-cyclohexanol.
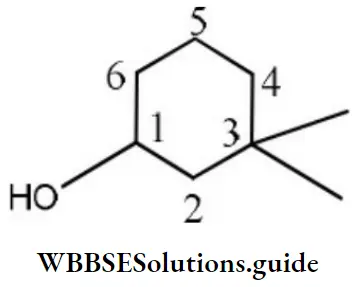
Question 65. The IUPAC name of the compound is

- But-2-en-l-ol
- l-hydroxy but-l-ene
- 4-hydroxy butene-3
- But-l-en-l-ol
Answer: 4. But-l-en-l-ol
Solution: Follow IUPAC rules.
Question 66. The name of (CH3)2 HC—O—CH2—CH2—CH3 is:
- Isopropyl propyl ether
- Dipropyl ether
- Di-isopropyl ether
- Isopropyl propyl ketone
Answer: 1. Isopropyl propyl ether
Solution: Ether group (—O—) has propyl and isopropyl groups on its two sides.
Question 67. IUPAC name of, CH3 CH(OH) CH2 CH2 COOH is:
- 4-hydroxy pentanoic acid
- 1-carboxy-3-butanoic acid
- 1-carboxy-4-butanol
- 4-carboxy-2-butanol
Answer: 1. 4-hydroxy pentanoic acid
Shortcut tricks for IUPAC nomenclature NEET with practice questions
Question 68. The prefix name of —SH group in IUPAC system is:
- Mercapto
- Thiol
- Sulphide
- None of these
Answer: 1. Mercapto
Solution: Follow IUPAC rules.
Question 69. The correct structure of 4-bromo-3-methyl-but-1-ene.
- Br-CH=C(CH3)2
- CH2=CH-CH(CH3)-CH2 Br
- CH2=C(CH3)CH2 CH2 Br
- CH3-C(CH3)=CHCH2-Br
Answer: 2. CH2=CH-CH(CH3)-CH2 Br
Solution: Follow IUPAC rules.
Question 70. Which of the following represents neo-pentyl alcohol?
- CH3CH(CH3)CH2CH2OH
- (CH3)3C-CH2OH
- CH3(CH2)3 OH
- CH3CH2CH(OH)C2H5
Answer: 2. (CH3)3C-CH2OH
Solution: (CH3)3C-CH2OH is neo-pentyl alcohol.
Question 71. IUPAC name of the compound, ClCH2 CH2 COOH is:
- 3-chloropropanoic acid
- 2-chloropropanoic acid
- 2-chloroethanoic acid
- Chlorosuccinic acid
Answer: 1. 3-chloropropanoic acid
Solution: Follow IUPAC rules.
Question 72. Which of the following IUPAC names is correct?
- 2-methyl-3-ethyl pentane
- 2-ethyl-3-methyl pentane
- 3-ethyl-2-methyl pentane
- 3-methyl-2-ethyl pentane
Answer: 3. 3-ethyl-2-methyl pentane
Solution: -do-
Question 73. IUPAC name of, (C2H5)2CHCH2OH is:
- 2-ethyl butanol-1
- 2-methyl pentanol-1
- 2-ethyl pentanol-1
- 3-ethyl butanol-1
Answer: 1. 2-ethyl butanol-1
Solution: Follow IUPAC rules.
Question 74. 3-phenylpropanoid acid is IUPAC name of:
- Mendaleic acid
- Pyruvic acid
- Succinic acid
- Cinnamic acid
Answer: 4. Cinnamic acid
Solution: C6 H5—CH=CHCOOH is cinnamic acid.
Question 75. IUPAC name of the compound is:
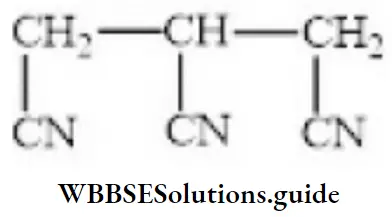
- 1,2,3-trieyanopropane
- Propane-1,2,3-tricarbonitrile
- 1,2,3-cyclopropane
- Propane triarylamine
Answer: 2. Propane-1,2,3-tricarbonitrile
Solution: Follow IUPAC rules.
Question 76. IUPAC name of the compound is
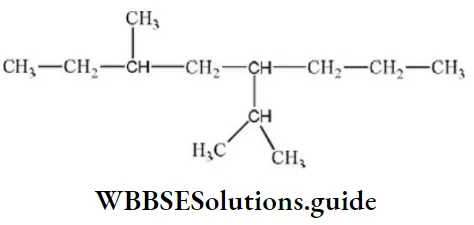
- 4-isopropyl, 6-methyl octane
- 3-methyl, 5-(1-methyl ethyl) octane
- 3-methyl, 5-isopropyl octane
- 6-methyl, 4-(1-methyl ethyl) octane
Answer: 2. 3-methyl, 5-(1-methyl ethyl) octane
Solution: IUPAC name of compound.
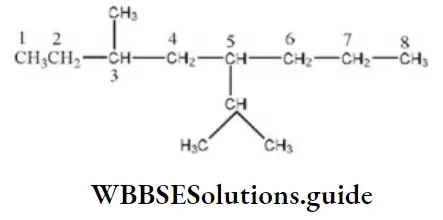
Straight chain which contains a large number of side chains is taken as the parent chain and the counting starts from that side where the side chain is nearest.
Question 77. IUPAC name of the compound is:

- 4-bromo-3-ethyl-1,4-pentadiene
- 2-bromo-3-ethyl-1,4pentadiene
- 2-bromo-3-ethyl-1-5-pentadiene
- None of the above
Answer: 2. 2-bromo-3-ethyl-1,4pentadiene
Solution: The IUPAC name of this molecule is 2-bromo-3-ethyl-1, 4-pentadiene.
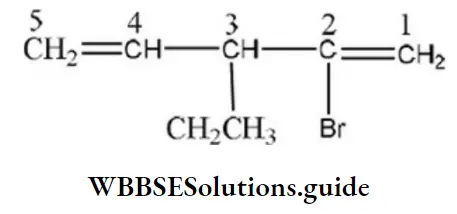
Question 78. IUPAC name of the compound is:
- 4,5-dimethyl oct-4-ene
- 3,4-dimethyl oct-5-ene
- 4,5-dimethyl oct-5-ene
- None of the above
Answer: 1. 4,5-dimethyl oct-4-ene
Solution: Follow IUPAC rules.
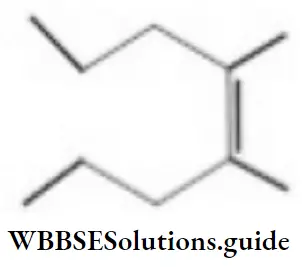
Question 79. The correct IUPAC name of the following compound is

- 5, 6-dimethyl-8-methyl dec-6-ene
- 6-butyl-5-ethyl-3-methyl oct-4-ene
- 5, 6-diethyl-3-methyl dec-4-ene
- 2, 4, 5-triethyl non-3-ene
Answer: 3. 5, 6-diethyl-3-methyl dec-4-ene
Question 80. Which of the following compounds has incorrect IUPAC nomenclature?
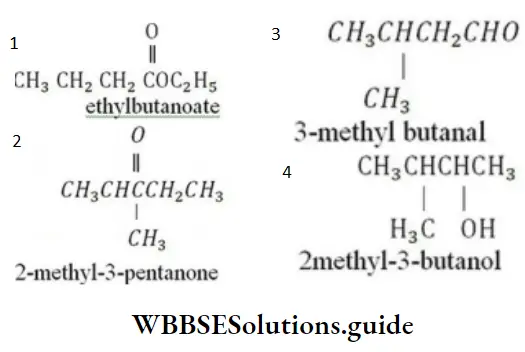
Answer: 4
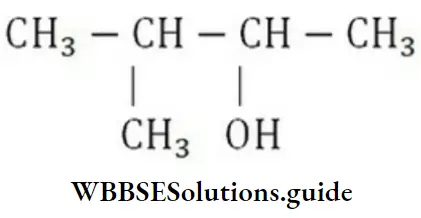
Question 81. The IUPAC name of the compound is
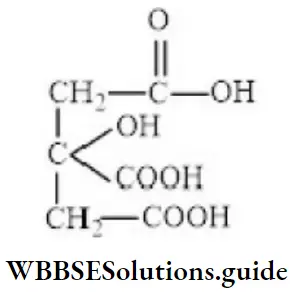
- 1,2,3-tricarboxylic-2,1-propane
- 3-carboxy-3-hydroxy-1,5-pentanoic acid
- 3-hydroxy-3-carboxy-1,5-pentanoic acid
- None of the above
Answer: 2. 3-carboxy-3-hydroxy-1,5-pentanoic acid
Solution: Follow IUPAC rules.
Question 82. IUPAC name of  is
is
- Bicyclo (2,1,0) pentane
- 1,2-cyclopropyl Cyclobutane
- Cyclopentane (4,3) annulene
- 1,2-methylene Cyclobutane
Answer: 1. Bicyclo (2,1,0) pentane
Solution: Follow IUPAC rules.
Question 83. The IUPAC name of neopentane is:
- 2,2-dimethylpropane
- 2-methylpropane
- 2,2-dimethylbutane
- 2-methyl butane
Answer: 1. 2,2-dimethylpropane
Solution: Follow IUPAC rules.
Question 84. The IUPAC name of compound shown below is
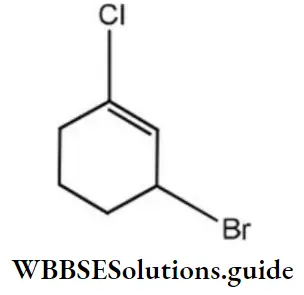
- 2-bromo-6- chlorocyclohex-1-ene
- 6-bromo-2-chlorocyclohexene
- 3-bromo-1-chlorocyclohexene
- 1-bromo-3-chlorocyclohexene
Answer: 3. 3-bromo-1-chlorocyclohexene
Solution:
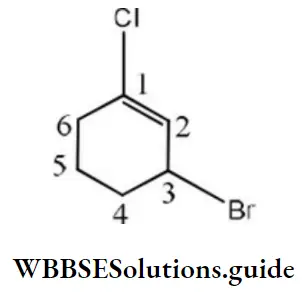
Unsaturation (double bond) is given priority over halogen, then the lowest set of locants. So, the correct IUPAC name is 3-bromo-1-chlorocyclohexene.
NEET important nomenclature problems with step-by-step solutions
Question 85. The IUPAC name of
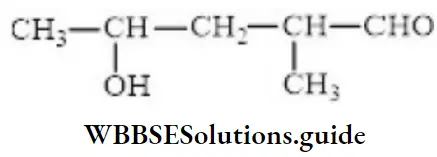
- 4-hydroxy-1-methylpentanal
- 4-hydroxy-4-methyl pent-2-en-1-al
- 2-hydroxy-4-methyl pent-2-en-5-al
- 2-hydroxy-3-methyl pent-2-en-5-al
Answer: 2. 4-hydroxy-4-methyl pent-2-en-1-al
Solution: Follow IUPAC rules.
Question 86. IUPAC’s name of is
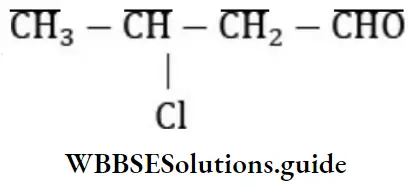
- 3-chlorobutanol
- 3-chlorobutanaldehyde
- 3-chlorobutanol
- 2-chlorobutanol
Answer: 3. 3-chlorobutanol
Solution:
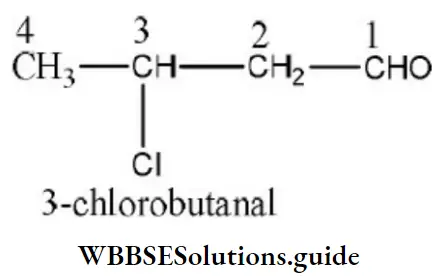
The order is priority is -CHO > -Cl
Question 87. The IUPAC name of is
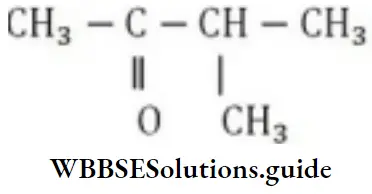
- 2-methyl-3-butanone
- 3-methyl-butan-2-one
- 3-methyl butanone
- None of these
Answer: 2. 3-methyl-butane-2-one
Solution: Follow IUPAC rules.
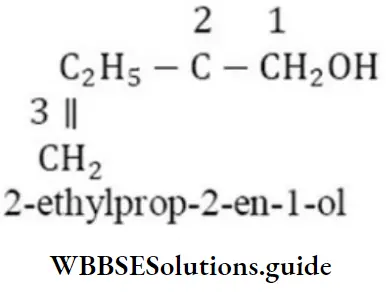
Question 88. The IUPAC name of compound is 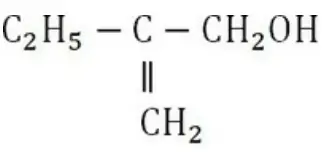
- 2-ethyl prop-2-en-1-ol
- 2-hydroxymethylbutan -1-ol
- 2-methylenebutan-1-ol
- 2-ethyl-3-hydroxypropyl-1-ene
Answer: 1. 2-ethyl prop-2-en-1-ol
Solution:
Question 89. Which nomenclature is not according to the IUPAC system?
Answer: 3
Solution: CH2=CH-CH2-Br is allyl bromide. Its IUPAC name is 3-bromopropene or 3-bromoprop-1-ene. Because here double bond is a functional group while halogen is a substituent. Double bond is given more priority and hence numbering starts from left.
Question 90. Acetonitrile is
- CH3CN
- CH3COCN
- C2H5CN
- C6H5CN
Answer: 1. CH3CN
Solution: CH3 C=N is known as acetonitrile or methyl cyanide.
Question 91. Isobutyl chloride is:
- CH3CH2CH2CH2Cl
- (CH3)2CHCH2 Cl
- CH3CH2CHClCH3
- (CH3)3C-Cl
Answer: 2. (CH3)2CHCH2 Cl
Solution: It should contain (CH3)2CH- group to be named as iso.
Question 92. The given compound in IUPAC is called as
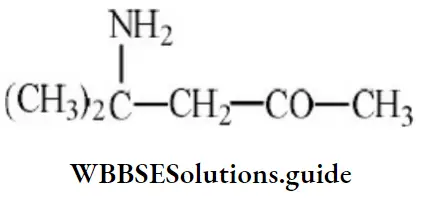
- Diacetone
- Acetone amine
- Diacetone amine
- 4-amino-4-methyl pentane-2-one
Answer: 4. 4-amino-4-methyl pentane-2-one
Solution: Follow IUPAC rules.
Question 93. The IUPAC name of compound is
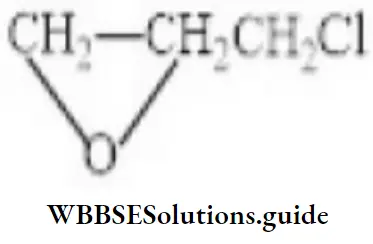
- 1-choloro-2,3-epoxypropane
- 3-chloro-1,2-epoxypropane
- 1-chloroethoxymethane
- None of the above
Answer: 2. 3-chloro-1,2-epoxypropane
Solution: Follow IUPAC rules.
Question 94. Neo-heptyl alcohol is correctly represented as:
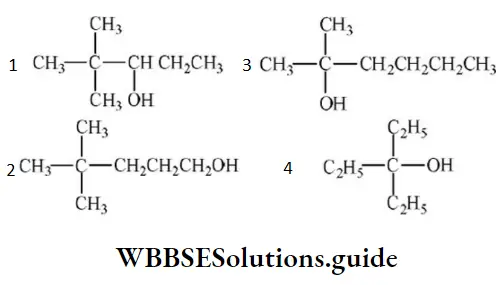
Answer: 3
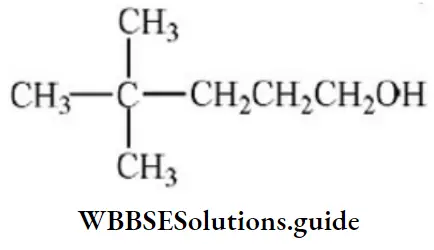
Solution: Follow IUPAC rules.
Question 95. IUPAC name of the compound is CH3 CH2 C(Br)=CH-Cl
- 2-bromo-1-chloro butene-1
- 1-chloro-2-bromo butene-1
- 3-chloro-2-bromo butene-2
- None of the above
Answer: 1. 2-bromo-1-chloro butene-1
Solution: Follow IUPAC rules.
Question 96. The IUPAC name of CH3-CH2-CHO is
- Propanal-1
- 2-methyl butanal
- Butanal-1
- Pentanal-1
Answer: 1. Propanal-1
Solution: IUPAC name of CH3 CH2 CHO is propan-1-al.
Question 97. The IUPAC name of is
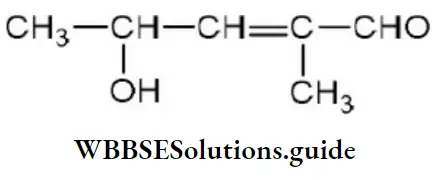
- 4-hydroxy-1-methylpentanal
- 4-hydroxy-2-methylpentanal
- 3-hydroxy-2-methylpentanal
- 3-hydroxy-3-methylpentanal
Answer: 2. 4-hydroxy-2-methylpentanal
Solution: Follow IUPAC rules.
Question 98. Select the correct statement:
- The prefixes are written before the name of compound
- The suffixes are written after the name of compound
- The IUPAC name of a compound is always written as one word
- All of the above
Answer: 4. All of the above
Solution: These are the IUPAC rules.
Question 99. Pick out the correct statement from the following and choose the correct answer from the codes given below
- Hexa-1, 5-diene is a conjugated diene
- Prop-1, 2-diene is conjugated diene
- Hexa-1, 3-diene is a conjugated diene
- Buta-1, 3-diene is an isolated diene
- Prop-1, 2-diene is a cumulative diene
- 1, 2
- 2, 3
- 4, 5
- 2, 5
Answer: 3. 4, 5
Solution: In conjugated diene alternate single and double bonds are present while in cumulative diene, double bonds are present at adjacent positions.

Hence, statements 3 and 5 are correct.
Question 100. The IUPAC name for
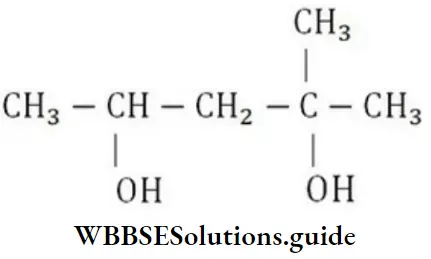
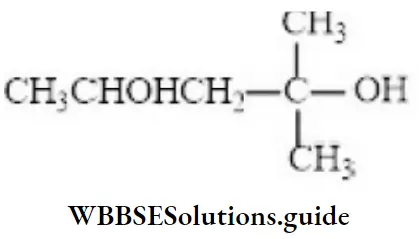
- 1,1-dimethyl-1,2-butanediol
- 2-methyl-2,4-pentanediol
- 4-methyl-2,4-pentanediol
- 1,3,3-dimethyl-1,3-propanediol
Answer: 2. 2-methyl-2,4-pentanediol
Solution: -do-
Question 101. Formulae of phenyl carbinol and chloral are respectively:
- C6 H5.CH2 CH2 OH and CHCl2 CHO
- C6 H5 CH2 OH and CCl3 CHO
- C6 H5 OH and CH2 Cl.CHO
- C6 H5 CHO and CHCl2 CHO
Answer: 2. C6 H5 CH2 OH and CCl3 CHO
Solution: Carbinol is trivial name for HCH2 OH. Thus, C6 H5 CH2 OH is phenyl carbinol and chloral is CCl3 CHO.
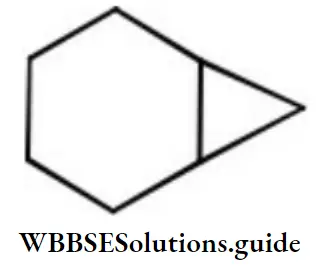
Question 102. The correct name for the following hydrocarbon is
- Tricycle [4.1.0] heptane
- Bicyclo [5.2.1] heptane
- Bicyclo [4.1.0] heptane
- Bicyclo [4.1.0] hexane
Answer: 3. Bicyclo [4.1.0] heptane
Solution:
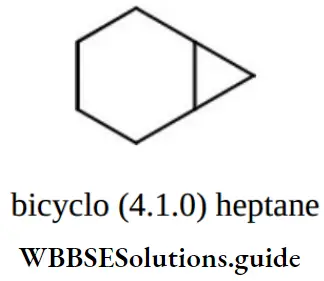
This compound contains 7 carbon atoms, so the corresponding alkane is heptane. Two bridges contain 4 and 1 carbon atom respectively and one bridge does not contain any carbon atom. So, the name of the compound is Bicyclo (4,1,0) heptane.
Question 103. The IUPAC name of the compound is:
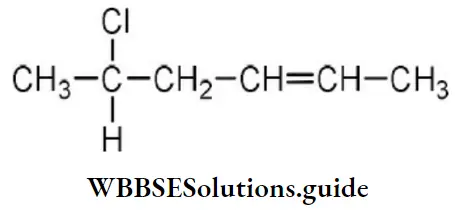
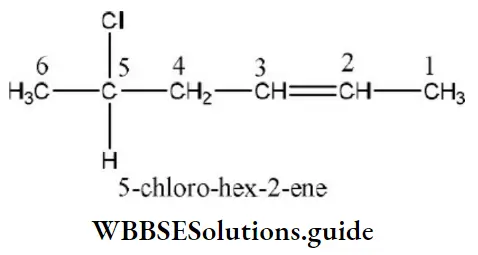
- 5-chloro-hex-2-ene
- 2-chloro-hex-5-ene
- l-chloro-1-methyl-pent-3-ene
- 5-chloro-5-methyl-pent-2-ene
Answer: 1. 5-chloro-hex-2-ene
Solution: First the longest continuous chain of carbon atoms is selected. Number the chain from the side containing senior functional group (i.e., the group placed above in the seniority table).
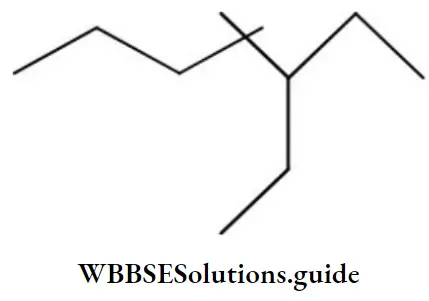
Question 104. The IUPAC name of the compound is:
- 1,1-diethyl1-2, 2-dimethyl pentane
- 4,4-dimethyl-5,5-diethyl pentane
- 5, 5-diethyl-4,4-dimethyl pentane
- 3-ethyl-4,4-dimethyl heptane
Answer: 4. 3-ethyl-4,4-dimethyl heptane
Solution: -do-
Question 105. 2, 3-dimethyl hexane contains _______ tertiary ____ secondary and ______ primary carbon atoms, respectively
- 2, 2, 4
- 2, 4, 3
- 4, 3, 2
- 3, 2, 4
Answer: 1. 2, 2, 4
Solution: The structure of 2, 3-dimethyl hexane is
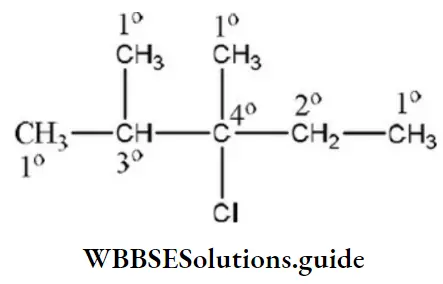
So, the number of tertiary carbon atoms = 2
The number of secondary carbon atoms = 2
The number of primary carbon atoms = 4
Question 106. IUPAC name of following compound is:
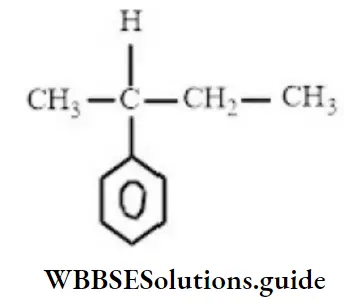
- 2-cyclohexyl butane
- 2-phenylbutazone
- 3-cyclohexyl butane
- 3-phenylbutazone
Answer: 2. 2-phenylbutazone
Solution: Follow IUPAC rules.
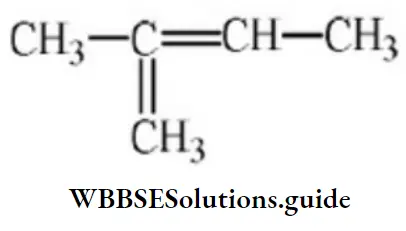
Question 107. 2-methyl-2-butene will be represented as:
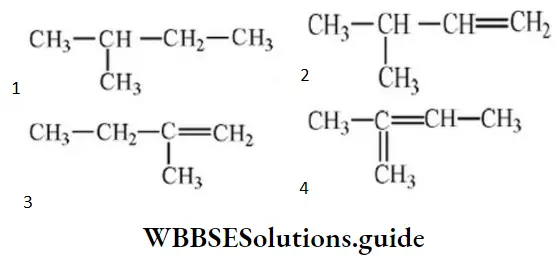
Answer: 4
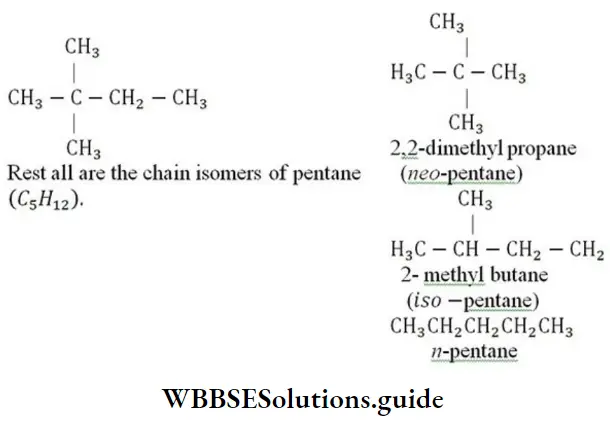
Question 108. Pick out the alkane which differs from the other members of the group
- 2,2-dimethyl propane
- Pentane
- 2-methyl butane
- 2, 2-dimethyl butane
Answer: 4. 2, 2-dimethyl butane
Solution: 2, 2-dimethyl butane is 6-carbon hydrocarbon (C6 H14)
Question 109. In IUPAC suffix name of —COX is:
- Oyl halide
- Halo carbonyl
- Carbamoyl
- None of these
Answer: 1. Oyl halide
Solution: Follow IUPAC rules.
Question 110. IUPAC name of following compound is:
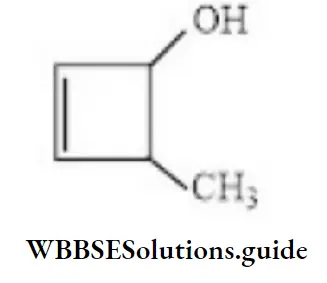
- 3-methyl cyclo-1-buten-2-ol
- 4-methyl cyclo-2-buten-1-ol
- 4-methyl cyclo-1-buten-3-ol
- 2-methyl cyclo-3-buten-1-ol
Answer: 2. 4-methyl cyclo-2-buten-1-ol
Solution: Follow IUPAC rules.
Question 111. The correct IUPAC name of the acid is
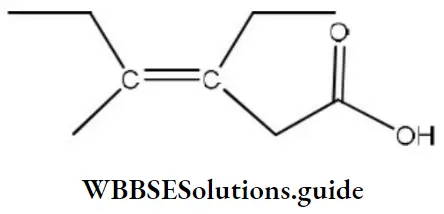
- Z-3-ethyl-4-methyl hex-3-en-1-oic acid
- Z-3-ethyl-4-methyl hexanoic acid
- Z-3, 4-diethyl pent-3-en-1-oic acid
- E-3-ethyl-4-methylhex-4-en-1-oic acid
Answer: 3. Z-3, 4-diethyl pent-3-en-1-oic acid
Solution:
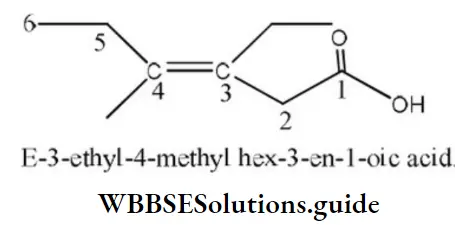
[The configuration of this compound is E because bulkier groups are present opposite of the double bond.]
Question 112. The IUPAC name of aldehyde is
- Prop-2-en-1-al
- Propenyl aldehyde
- But-2-en-1-al
- Propenal
Answer: 1. Prop-2-en-1-al
Solution: CH2=CH-CHO (Prop -2-en-1-al)
Question 113. Write the IUPAC name of the below compound.
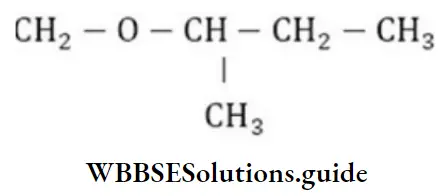
- 3-methoxy butane
- 2-methoxy butane
- 3-methyl-3-methoxy propane
- Butoxy methane
Answer: 2. 2-methoxy butane
Solution: According to the IUPAC system ethers are named as alkoxy alkanes. The larger alkyl group forms the parent chain while the lower alkyl group is taken with the ethereal oxygen and forms a part of the alkoxy group.
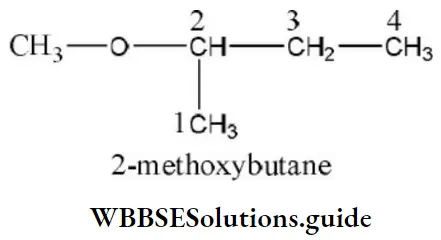
Question 114. IUPAC name of the compound is
- 4-butyl-2,5-hexadien-l-al
- 5-vinyloct-3-en-l-al
- 5-vinyloct-5-en-8-al
- 3-butyl-1,4-hexadien-6-al
Answer: 1. 4-butyl-2,5-hexadien-l-al
Solution: Follow IUPAC rules.
Question 115. The IUPAC name of the compound is
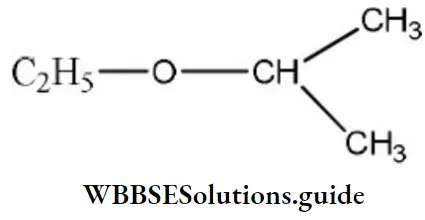
- Ethoxy propane
- 1, 1-dimethyl ether
- 2-ethoxy zso-propane
- 2-ethoxy propane
Answer: 4. 2-ethoxy propane
Solution:
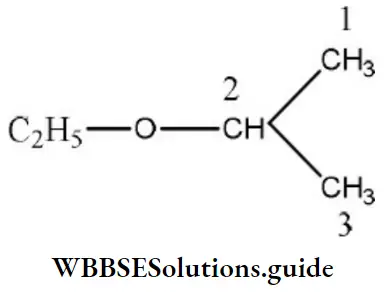
The above compound is an ether and its name is written as alkoxy alkane. Oxy is attached to the lower group. Hence, the IUPAC name of the above compound is 2-ethoxy propane.
Question 116. The IUPAC name of the compound
- Ethane nitrile
- Methane isonitrile
- Ethane isonitrile
- None of these
Answer: 2. Methane isonitrile
Solution: CH3NC is methaneisonitrile.
Question 117. The structural formula of methyl aminomethane is:
- (CH3)2 CHNH2
- (CH3)3 N
- (CH3)2 NH
- CH3 NH2
Answer: 3. (CH3)2 NH
Solution: IUPAC name is N-methyl methanamine.
Question 118. The IUPAC name of CH3-CCH(CH3)2 is
- 4-methyl-2-pentyne
- 4, 4-dimethyl-2-butyne
- methyl isopropyl acetylene
- 2-methyl-4-pentyne
Answer: 1. 4-methyl-2-pentyne
Solution: Follow IUPAC rules.
Question 119. Which one of the following is the correct formula for dichlorodiphenyltrichloroethane?
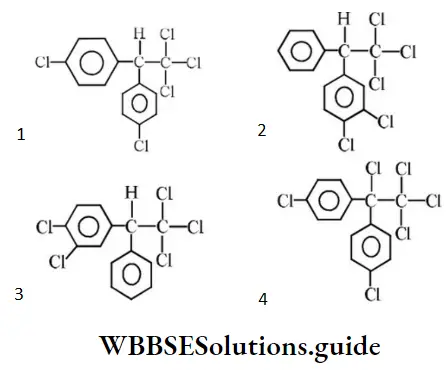
Answer: 1
Solution: Follow IUPAC rules.
Question 120. IUPAC name of (CH3)2N-C2H5 is:
- Dimethyl ethyl amine
- Dimethyl aminomethane
- Dimethyl aminoethane
- N, N-dimethylethanolamine
Answer: 4. N, N-dimethylethanolamine
Question 121. 3-methyl penta-1,3-diene is:
- CH2=CH(CH2)2CH3
- CH2=CHCH(CH3)CH2CH3
- CH3CH=C(CH3)CH=CH2
- CH3-CH=CH(CH3)2
Answer: 3. CH3CH=C(CH3)CH=CH2
Solution: Follow IUPAC rules.
Question 122. The IUPAC name of the compound is
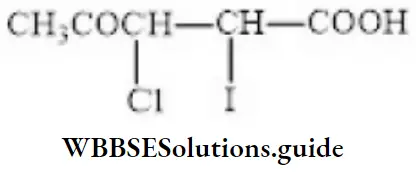
- 2-iodo-3-chloro-4-pentanoic acid
- 4-oxo-3-chloro-2-iodo Pentanoic acid
- 4-carboxy-4,3-chloro-2-butanone
- 3-chloro-2-iodo-4-oxo-pentanoic acid
Answer: 4. 3-chloro-2-iodo-4-oxo-pentanoic acid
Solution: Report prefixes in alphabetical order.
Question 123. The IUPAC name for CH3COCH(CH3)2 is:
- 4-methyl isopropyl ketone
- 3-methyl-2-butanone
- Isopropyl methyl ketone
- 2-methyl-3-butanone
Answer: 2. 3-methyl-2-butanone
Question 124. Give the IUPAC name of the compound
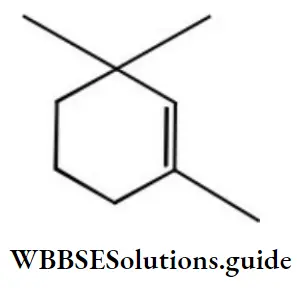
- 1,1,3-trimethyl cyclo hex-2-ene
- 1,3,3-trimethyl cyclo hex-l-ene
- 1,1,5-trimethyl cyclo hex-5-ene
- 2,6,6-trimethyl cyclo hex-l-ene
Answer: 2. 1,3,3-trimethyl cyclo hex-l-ene
Solution: Follow IUPAC rules.
Question 125. The name of the compound is:
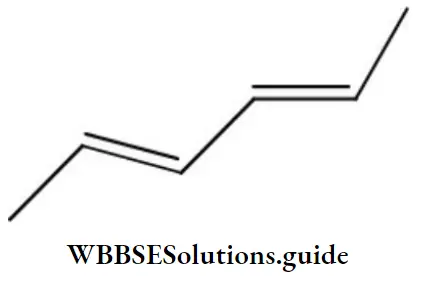
- (2Z,4Z)-2, 4-hexadiene
- (2Z-, 4E)-2, 4-hexadiene
- (2E, 4Z)-2, 4-hexadiene
- (4E, 4Z)-2, 4-hexadiene
Answer: 1. (2Z,4Z)-2, 4-hexadiene
Solution: The name of the compound is (2Z,4Z)-2, 4-hexadiene.
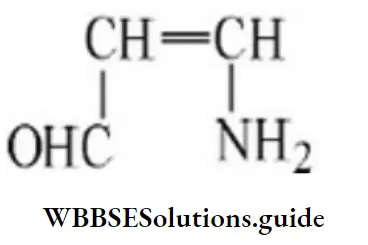
Question 126. The IUPAC name of the compound is
- 2-amino-3-hydroxy propanoic acid
- l-hydroxy-2-amino propan-3-oic acid
- l-amino-2-hydroxy propanoic acid
- 3-hydroxy-2-amino propanoic acid
Answer: 1. 2-amino-3-hydroxy propanoic acid
Solution: Follow IUPAC rules.
Question 127. What is the formula of tertiary butyl alcohol?
- CH3-CH(CH3)-CH2-OH
- CH3-(CH2)2OH
- CH3-CH(OH)-CH2-CH3
Answer: 4. CH3-CH(OH)-CH2-CH3
Solution:
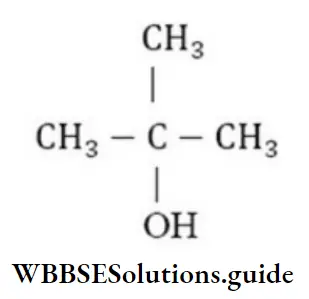
is the formula of tertiary butyl alcohol as in it – OH group is attached to tertiary carbon.
Question 128. The IUPAC name of the compound is

- 2-ethoxy pentane
- 4-ethoxy pentane
- Pentyl-ethyl ether
- 2-pentoxy ethane
Answer: 1. 2-ethoxy pentane
Solution: Follow IUPAC rules.
Question 129. The IUPAC name of the compound is
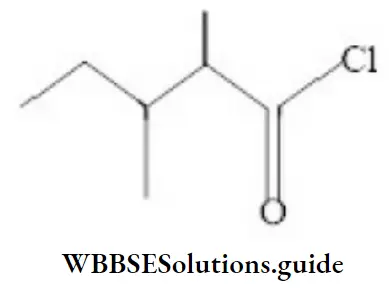
- 2-ethyl-3-methyl butanol chloride
- 2,3-dimethyl pentanol chloride
- 3,4-dimethyl pentanol chloride
- l-chloro-l-oxo-2,3-dimethyl pentane
Answer: 2. 2,3-dimethyl pentanol chloride
Question 130. The IUPAC name of the compound is
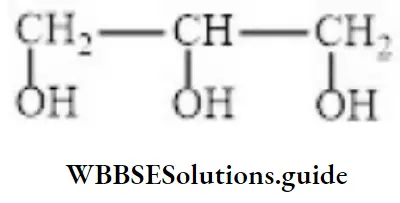
- 1,2,3-trihydrosypropane
- 3-hydroxy pentane-l,5-diol
- 1,2,3-hydroxy-propane
- Propane-1,2,3-triol
Answer: 4. Propane-1,2,3-triol
Solution: Follow IUPAC rules.
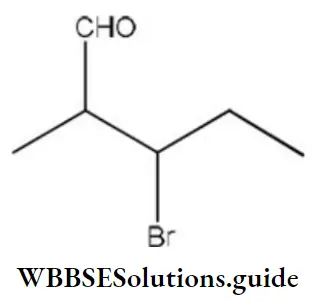
Question 131. The IUPAC name of is
- 2-methyl-3-bromohexanal
- 3-bromo-2-methylbutanal
- 2-bromo-3-bromobutanal
- 3-bromo-2-methylpentanal
Answer: 4. 3-bromo-2-methylpentanal
Solution: Follow IUPAC rules.
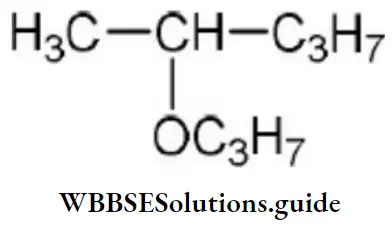
Question 132. The IUPAC name of
- 4-propoxy pentane
- Pentyl-propyl ether
- 2-propoxy pentane
- 2-pentoxy propane
Answer: 3. 2-propoxy pentane
Solution: Follow IUPAC rules.
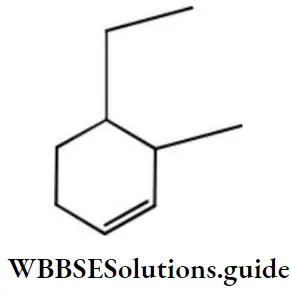
Question 133. The systematic (IUPAC) name of the compound with the following structural formula shall be
- 1-ethyl-2-methyl cyclohexene
- 2-methyl-l-ethyl cyclohexene
- 3-ethyl-2-methyl cyclohexene
- 4-ethyl-3-methyl cyclohexene
Answer: 4. 4-ethyl-3-methyl cyclohexene
Solution: Follow IUPAC rules.
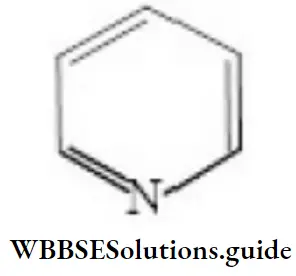
Question 134. Pyridine is:
- An aromatic compound and a primary base
- A heterocyclic amino compound and a tertiary base
- An aromatic amino compound forms salts
- A cyano derivative of benzene and a secondary base
Answer: 2. A heterocyclic amino compound and a tertiary base
Solution: Pyridine is a heterocyclic compound having six carbon-membered ring formed by C and N atoms
Question 135. The IUPAC name of (CH3)3C-CH=CH2 is
- 1, 1, 1-trimethyl-2-propene
- 3, 3, 3-trimethyl-2-propene
- 2, 2-dimethyl-3-butene
- 3, 3-dimethyl-1-butene
Answer: 4. 3, 3-dimethyl-1-butene
Solution:
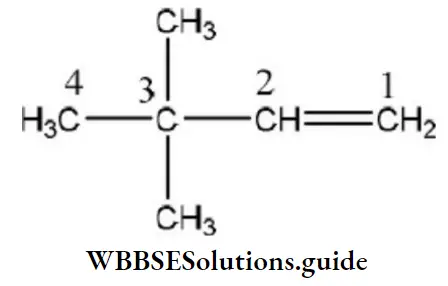
IUPAC name=3, 3-dimethyl-l-butene.
Question 136. The name of the compound is:

- 2-pentanone
- Pentanone-2
- Pentan-2-one
- All are correct
Answer: 4. All are correct
Question 137. Write the IUPAC name of
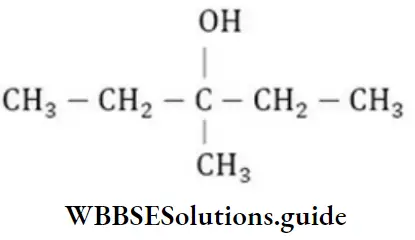
- 3-methyl pentane-3-ol
- 3-hydroxy hexane
- 3-hydroxy-3-methyl pentane
- All of the above
Answer: 1. 3-methyl pentane-3-ol