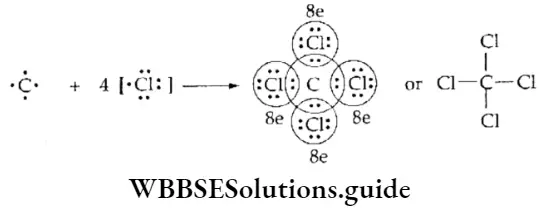
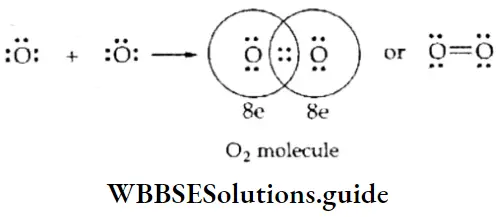
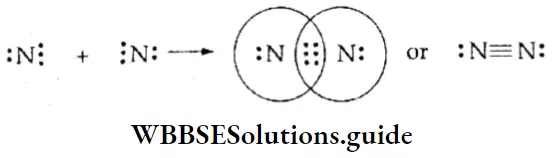
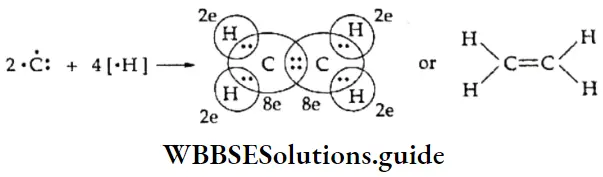
WBCHSE Class 11 Polar Covalent Bond Definition And Properties
The electronegativity of an element usually depends upon its atomic size. The smaller the size of an atom, the more it attracts the bonded electrons (the nucleus of the atom being closer), so the greater its electronegativity. The value of electronegativity also depends upon the state of hybridization (sp > sp2 > sp3).
In the periodic table, the value of electronegativity decreases down a group because the atomic size Increases. The value of electronegativity increases across a period until Group 17. The electronegativity of group 17 elements (the halogens) is the highest in every period.
- A covalent bond is formed between two atoms when they share a pair of electrons. This shared pair is under the influence of both the atoms and one might expect the two atoms to have an equal hold over the shared pair of electrons. But this is not the case always.
- How much hold each atom has over the shared pair of electrons depends on the value of its electronegativity. And when one atom has n greater hold over the shared pair of electrons and pulls it more towards itself, it acquires a partial negative charge while the other atom acquires a partial positive charge. If, on the other hand, the two atoms share the electron pair equally, no charge develops over either atom.
- Thus in molecules like HF, HBr, or KCl, the halogen atom being highly electronegative attracts the shared electron pair towards itself. On the other hand, in the case of a homonuclear diatomic molecule like H2, Br2, or N2, the electron pair is shared equally between the two atoms due to the same electronegativity.
- Thus, electronegativity is an important factor affecting the nature of covalent bonds. Another way of saying this is that a covalent bond may be polar or nonpolar, depending upon the electronegativities of the bonded atoms.
Read and Learn More WBCHSE For Class11 Basic Chemistry Notes

Nonpolar covalent bond: If two atoms of similar electronegativities form a bond by sharing a pair of electrons, the shared pair of electrons is equally attracted by the two and lies midway between the nuclei of the two atoms. Such a covalent bond is called a nonpolar covalent bond. The bonds formed between similar atoms, as in the case of H2,O2, N2, and are nonpolar covalent bonds.
| Class 11 Biology | Class 11 Chemistry |
| Class 11 Chemistry | Class 11 Physics |
| Class 11 Biology MCQs | Class 11 Physics MCQs |
| Class 11 Biology | Class 11 Physics Notes |
“WBCHSE Class 11 Chemistry, polar covalent bond, definition, properties, and examples”
Polar covalent bond: When two atoms of different electronegativities share a pair of electrons, the shared pair of electrons does not lie midway between the two bonded atoms. It shifts towards the more electronegative atom. In other words, the distribution of electrons gets distorted, or the electron cloud is displaced towards the more electronegative atom.

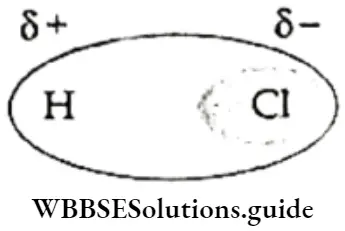
- The slightly higher electron density around the more electronegative atom makes it acquire a partial negative charge (indicated as δ-). Tine-less electronegative atom acquires a partial positive charge (δ+). Thus, positive and negative poles are developed in the molecule. This type of bond is called a polar covalent bond, i.e., a covalent bond with a partial ionic character.
- In the hydrogen chloride molecule, for example, chlorine is more electronegative than hydrogen, so the shared pair of electrons is displaced towards chlorine, i.e., the electron density is higher around the chlorine atom, and it acquires a fractional negative charge. The hydrogen atom, on the other hand, acquires an equal positive charge.
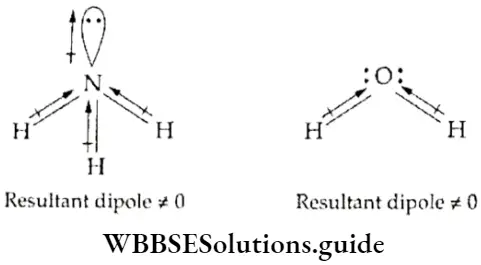
- Molecules containing polar covalent bonds are called polar molecules. A molecule may contain more than one covalent bond, for example, NH3 and H2O.

The ionic character of polar covalent bond: One can look upon an ionic bond as an extreme case of a polar covalent bond. When two atoms of different electronegativities are bonded together by a covalent bond, the bond is polar as the shared electron pair shifts slightly towards the more electronegative atom.
When the difference in the electronegativities of the two atoms is very high, the shared pair of electrons is almost in complete possession of the more electronegative atom.
- In such a case, it is more valid to say that an electron has been transferred from the less electronegative to the more electronegative atom and that a positive ion and a negative ion have been created. The two ions are then held together by mutual (electrostatic) attraction or an ionic bond.
- When two atoms are linked together by a covalent bond having different electronegativities, the bond formed is polar, or the bond is said to possess a partial ionic character. The degree of ionic character of a bond is determined by the difference in electronegativities of the combining atoms. The greater the difference in the electronegativities, the greater the ionic character.
- If the difference between the electronegativities of the two atoms is 1.9, the bond is said to have 50% ionic and 50% covalent character.
- If the difference between the electronegativities of the two atoms is more than 1.9, the partial ionic character of the bond is more than 50% and the bond is taken to be ionic.
- If the difference between the electronegativities of the two atoms is less than 1.9, the bond is predominantly covalent.
Properties And Examples Of Polar Covalent Bonds In WBCHSE Chemistry
Dipole moment: One end of a polar molecule is negative and the other is positive, so the molecule behaves like an electric dipole. Since the negative charge is always equal in magnitude to the positive charge, the molecule as a whole is electrically neutral.
The dipole moment of a molecule is a measure of its polarity. It is the product of the magnitude of the negative or the positive charge (q) and the distance (d) between the charges, i.e., internuclear distance. It is usually denoted by ji. Mathematically this can be expressed as follows.
“Polar covalent bond, WBCHSE Class 11, chemistry notes, and key characteristics”
μ = q x d.
The charge q is of the order of 10-10 esu (electrostatic unit) and the internuclear distance d is of the order of 10-8 cm (A). Therefore, the dipole moment μ is of the order of 10-10 x 10-8 = 10-18 esu cm. The dipole moment is also expressed in debye (D).
1 D = 1 x 10-18 esu cm.
In SI units, 1 esu = 3.336 x 10-10 C
and 1 cm = 10-2 m.
∴ 1D = 3.336x 10-10 x 10-2 x 10-18 = 3.336 x 10-30 Cm.
- The dipole moment is a measure of the polarity, i.e., the extent of displacement of the electron cloud. The greater the difference between the electronegativities of the bonded atoms, the higher the value of the dipole moment.
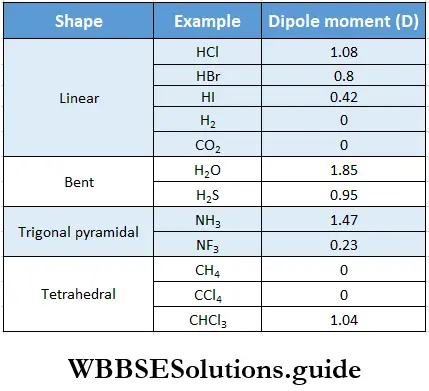
- For example, the dipole moment of HCI is greater than that of HBr though they have the same structure. This is because the difference between the electronegativities of chlorine and hydrogen is greater than the difference between the electronegativities of bromine and hydrogen.
Dipole moment and molecular structure: Dipole moment is a vector quantity, i.e., it has a magnitude and direction. It is represented by an arrow with its tail at the positive end and head pointing towards the negative end.
⇒ H-Cl
A molecule may have more than one polar bond. The polarity of a molecule with more than one polar bond is the resultant or the vector sunt of the dipole moments of all the bonded atoms, or the resultant of all the bond dipoles (the dipole moment of a bond is often referred to as the bond dipole).
“WBCHSE Class 11, chemistry notes, on polar covalent bond, and its formation”
Diatomic molecule In the case of a diatomic molecule in which the two atoms are bonded to each other by a polar covalent bond, the dipole moment of the molecule is just the dipole moment of that bond. For example, in the case of HC1, the molecular dipole moment is equal to the dipole moment of the H—Cl bond, i.e., 1.03 D.
The greater the difference between the electronegativities of the bonded atoms, the greater the dipole moment of the molecule. For example, the dipole moment of hydrogen halides are in the order,
H—F (1.91 D) > H—Cl (1.03 D) > H—Br (0.79 D) > H—I (0.38 D).
Comprehensive Guide To Polar Covalent Bonds For WBCHSE Class 11
Polyatomic molecule If a polyatomic molecule has more than one polar bond, its dipole moment is equal to the resultant of the dipole moments of all the individual bonds (or bond dipoles).
- Since dipole moment is a vector quantity, the magnitude and direction of the resultant depends not only on the values of the individual dipole moments but also on their orientation or direction.
- Thus, the dipole moment of a molecule may be zero even though it contains more than one polar bond. What the resultant of the individual dipole moments of the bonds in a molecule will be depends on the structure (spatial arrangement of atoms) of the molecule.
Dipole moments of CO2 and SO2 The carbon dioxide molecule has two C=0 polar bonds but its dipole moment is zero. This is because CO2 is a linear molecule in which the two C=O bonds are at 180° to each other, so the dipole moments are equal in magnitude and opposite in direction. The SO2 molecule has two polar bonds but unlike the CO2 molecule, its dipole moment is not zero. This is because the shape of the molecule is such that the resultant of the dipole moments of the two bonds is not zero.
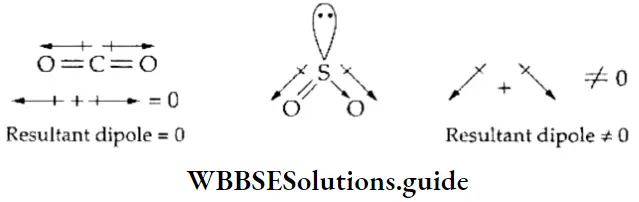
Dipole moments of BF3 and NF3 In BF3, there are three polar B—F bonds but the dipole moment of the molecule is zero because the resultant of the dipole moments of any two of the bonds is equal and opposite to the third (parallelogram law of forces).
The NF3 molecule also has three polar bonds. But because of the presence of a lone pair of electrons, the arrangement of the electron pairs is distorted tetrahedral and the orientation of the bond dipoles is such that their resultant is 0.8 x 10-30 Cm.
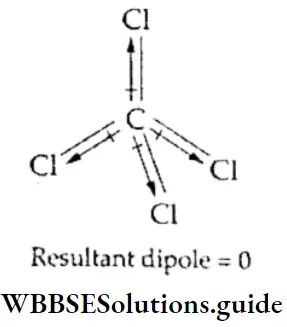
Generally, in symmetrical molecules, the resultant of all the dipole moments is zero.
- If we compare the dipole moments of NF3 and NH3 (both have pyramidal shapes with a lone pair of electrons) the dipole moment of NF3 is expected to be more as fluorine is more electronegative than hydrogen.
- But the dipole moment of NH3 (4.90 x 10-30 C m) is more than that of NF3. This is because in NH3 the dipole due to the lone pair and the resultant dipole moments of N—H bonds are in the same direction. In NF3 the direction of the dipoles is opposite.
The usefulness of dipole moment: Knowing the value of the dipole moment of a molecule is useful in many ways.
Polarity of a molecule The value of the dipole moment is an indicator of the polarity of the molecule. The higher the value of the dipole moment the greater the polarity of the molecule (the greater the polarity of the bond if the molecule contains only one polar bond). The dipole moment of a nonpolar molecule is zero. So the dipole moment can help ascertain whether a molecule is polar or nonpolar.
The ionic character of a molecule All covalent bonds have a partially ionic character. We can predict the ionic character of a molecule by considering the magnitude of its dipole moment.
Shape of molecule The dipole moment of a molecule helps to predict the shape of the molecule. For example, experiments show that the dipole moment of BeF2 is zero. This can be possible only if the resultant of the dipole moments of the two Be—F bonds is zero.
“Polar covalent bond, definition, properties, and real-world examples, WBCHSE syllabus”
In other words, the two bond dipoles must be oriented in opposite directions, which is possible only if the molecule is linear. The H2O molecule, on the other hand, has a dipole moment of 1.85 D. Since the resultant of the dipole moments of the two bonds in the H2O molecule is not zero, the molecule cannot be linear, it must be angular.
Partial covalent character of ionic bonds: Just as partial ionic character is attained in a covalent bond, similarly a partial covalent character is induced in an ionic compound. Consider a system of two ions A+ and B– together at an equilibrium distance (i.e., when the system acquires minimum energy).
The cation attracts the electron of the anion (B–) and repels the nucleus; thus the anion gets distorted or polarised. The anion being larger gets more readily polarised than the cation, and thus the effect of an anion on a cation is less pronounced.
If the extent of polarisation is large, the electrons of the anion are drawn towards the cation (this means electrons are shared by the ions), and covalent character results.
WBCHSE Class 11 Chemistry Notes: Polar Covalent Bond Properties And Examples
The extent of distortion of an anion depends on two factors:
- The polarising power of the cation
- The polarisability of the anion (ease of distortion of anion).
Generally, small cations with high positive charge have greater polarising power, and large anions with loosely held electrons are easily polarisable. The factors favoring covalency (in ionic compounds) have been summarised by Fajans and are referred to as Fajans’ rules. According to these rules, covalency is favored by:
- More charge on either ion
- Small cation
- Large anion
- Many transition metal ions and a few main group elements have high polarising power. Since a noble gas configuration (ns2np6, typical of alkali and alkaline earth metal cations) is most effective at shielding nuclear charge the ions with this configuration are not significantly polarising.
- Let us consider NaCl and AlCl3. In both cases the anion is Cl– whereas the cations are Na+ and Al3+. Al3+ (small size; high charge) has greater polarising power than Na+ (big size; small charge).
- Therefore, AlCI3 is more covalent than NaCl. Now, let us consider NaCl, NaBr, and Nal. The cation being the same, covalency depends on the aruons. In all the cases tire charge is the same, but the size follows the order: Cl– < BP– < I–. Thus polarisability also follows this order and the covalent character is maximum for Nal and least for NaCl.
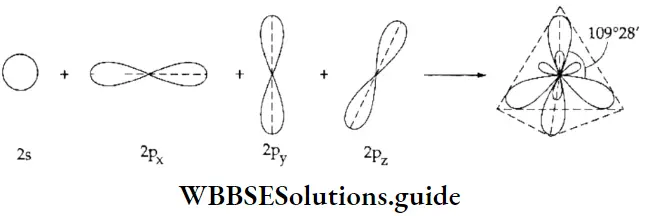
Directional properties of bonds: As you already know, different orbitals have different shapes. The shape of the s orbital is spherical so the orbital is represented as a circle.
- The p orbital consists of two lobes in contact at the origin. As you can see in the figure, p orbitals have a marked directional character, they have axes at right angles to each other. Hence they are known as px, py, and pz orbitals respectively.
- The orbitals are equivalent to each other but for the directional property. One lobe of the p orbital is marked with a positive sign and the other -with a negative sign. These signs have no physical significance, they arise from the fact that a wave function can have both positive and negative regions.
- In this context, it is important to remind you that the electron has a dual nature and it cannot be both particle and wave at the same time. According to wave mechanics, an electron is represented by a wave function \(\psi\).
- It does not matter at all which lobe of the p orbital has been given a positive or negative sign. Similarly, an s orbital can be + or -. When \(\psi\) is zero, there is no likelihood of finding an electron in the region.

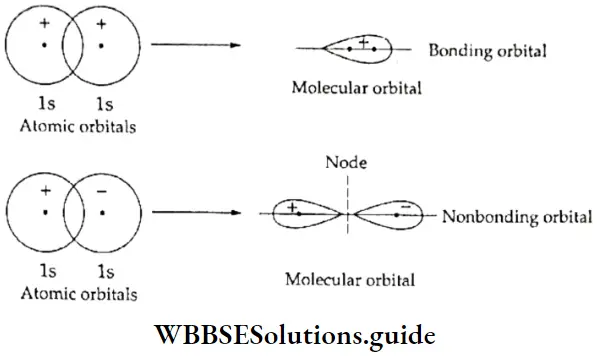
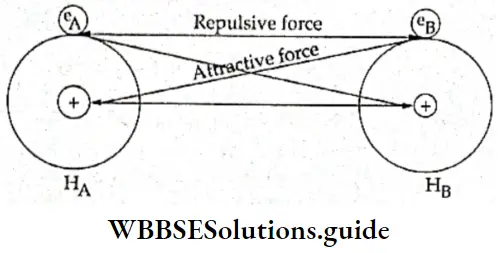
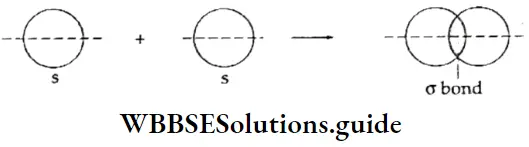
Let us apply these relative signs to understand the overlap of atomic orbitals. Consider two hydrogen atoms, A and B combining. The 1s atomic orbitals of the two atoms overlap to give rise to a molecular orbital.
The two electrons, one in each atomic orbital, can be represented by wave functions \(\psi_{\mathrm{A}} \text { and } \psi_{\mathrm{B}}\). Two combinations of the wave functions are then possible:
- Overlap of the wave functions with the same sign
- Overlap of the wave functions with opposite sign
- Wave functions with the same signs can be regarded as waves in the same phase which may add up to give a large resultant wave. On the other hand, wave functions with opposite signs cancel each other. In other words, when the overlap occurs between two regions with the same sign it results in a bonding orbital whereas in case of overlap between regions of the opposite sign, a nonbonding orbital results.
- As it is dear from the Figure, the electron charge is concentrated in the region between two nuclei in case of the bonding orbital, thereby holding the two nuclei together. On the other hand, in the nonbonding orbital, the electron charge is withdrawn from the region between the two nuclei, which results in repulsion between the two nuclei.
- Figure shows different combinations of s and p orbitals with positive, negative, and zero overlap. Overlap is said to be positive or negative if the signs of the overlapping lobes are the same or different respectively. In the case of zero overlaps, the extent of stabilization obtained by the overlap of orbitals of the same sign is equal to the extent of destabilization obtained by the overlap of orbitals of the opposite sign, so the net overlap is said to be zero.
Molecular Orbital Theory: In valence bond theory, a chemical bond is identified as a shared electron pair. Electrons in atoms are present in atomic orbitals. The atomic orbitals of different atoms overlap to give rise to a chemical bond.
This theory could not explain certain properties like relative bond strengths, and the paramagnetic and the diamagnetic characters of a molecule. Another theory that is widely used to explain bonding in molecules is referred to as molecular orbital theory.
“WBCHSE Class 11 Chemistry, polar covalent bonding, electronegativity, and dipole moment”
This theory was proposed by Hund and Mulliken (1932). Unlike the valence bond theory, the molecular orbital theory considers the valence electron to be associated with the nuclei of all combining atoms.
The main features of this theory are as follows:
- Atomic orbitals that match in energy and symmetry combine to give molecular orbitals.
- The total number of molecular orbitals formed is equal to the total number of combining atomic orbitals.
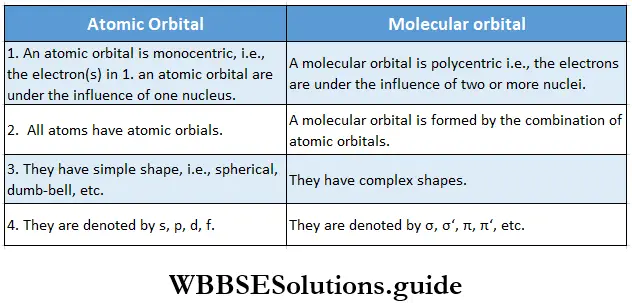
- Unlike the electron in an atomic orbital, the electron in a molecular orbital is influenced by two or more nuclei corresponding to the number of atoms in the molecule. (An atomic orbital is called monocentric, while a molecular orbital is called polycentric.)
- When two atomic orbitals combine, they form two molecular orbitals—the bonding molecular orbital which has lower energy than the atomic orbitals, and the antibonding molecular orbital which has higher energy than the atomic orbitals.
- Like atomic orbitals, molecular orbitals represent electron probability distribution.
- The filling up of molecular orbitals is done by the Aufbau principle, Pauli exclusion principle, and Hund’s rule.
- The maximum number of electrons present in a molecular orbital is 2.
Linear Combination of Atomic Orbitals (LCAO) Method: You already know that according to wave mechanics, atomic orbitals are represented by a wave function vy which is obtained by solving the Schrodinger wave equation.
It is very difficult to solve the Schrodinger wave equation for a molecule (since a molecular orbital is polycentric). Here the difficulty in finding a solution arises from the dependence of the wave function on internuclear distances or because an electron interacts simultaneously with more than one nuclei.
- Therefore, certain approximations have to be made to solve wave equations for molecules. The most common approximation is adopting the procedure of linear combination of atomic orbitals (LCAO). Why is a molecular orbital considered to be a linear combination of atomic orbitals? The answer to this lies in the fact that the electron with a larger region for movement or more freedom has lower energy.
- Now the larger region for the movement is possible only when the atomic orbitals overlap with each other. The overlap is maximum when the orbitals are in the same plane. Thus the molecular orbital is considered to be a linear combination of atomic orbitals.
Consider two atoms A and B whose atomic orbitals are represented by the wave functions \(\psi_{\mathrm{A}} \text { and } \psi_{\mathrm{B}}\) respectively. During bond formation, these orbitals combine to give molecular orbitals. According to the LCAO method, the atomic orbitals of the individual atoms may combine in two ways—the wave functions may add to or subtract from each other.
The molecular orbital obtained by the addition of the wave functions may be represented as
∴ \(\psi_{(\mathrm{MO})}=\psi_{\mathrm{A}}+\psi_{\mathrm{B}}\).
The resultant molecular orbital (\(\psi_{(\mathrm{MO})}\)) is bonding. Here signs of both the wave functions are the same, this implies that two waves are in phase. When such waves combine the resultant wave has larger amplitude (amplitudes of both waves add—constructive interference).
The molecular orbital obtained by subtracting the wave functions is represented as \(\psi_{(\mathrm{MO})}^*=\psi_{\mathrm{A}}+\left(-\psi_{\mathrm{B}}\right)\).
The resultant molecular orbital obtained by adding two wave functions of opposite signs is antibonding. This corresponds to two waves that are completely out of phase and cancel each other by destructive interference. We have already discussed the bonding and antibonding molecular orbitals taking the H2 molecule as an example, earlier in the chapter.
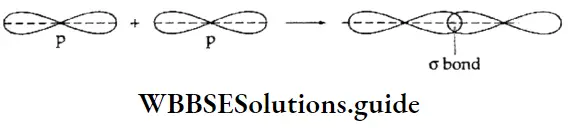
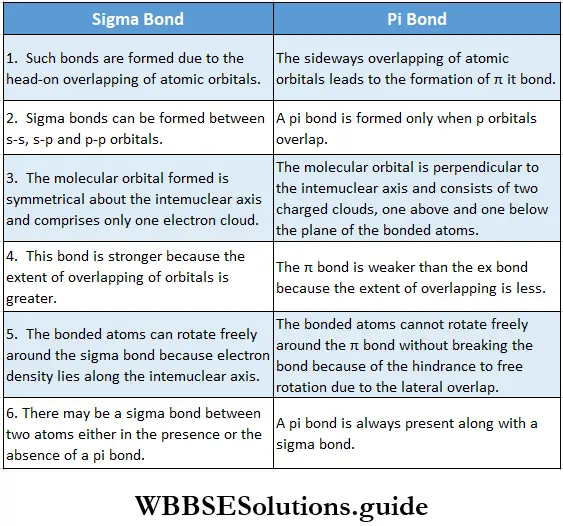
- Going back to the H2 molecule again, the bonding molecular orbital is denoted by \(\sigma_{15}\) while the antibonding molecular orbital is represented by \(\sigma_{1 s}^*\). It is called a because the overlap of atomic orbitals is along the internuclear axis and Is because the combining atomic orbitals are Is orbitals.
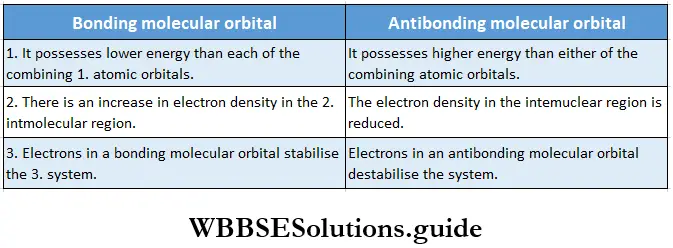
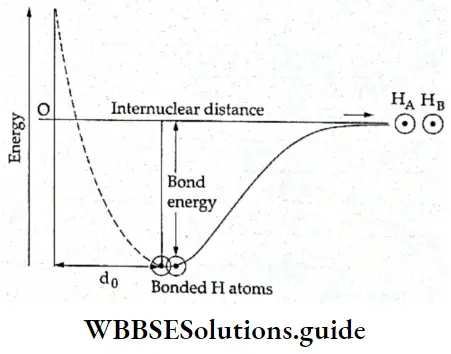
- In a bonding molecular orbital, there is an increase in electron density in the internuclear region. The nuclei are shielded from each other so that the repulsion between them is very low. This results in a lowering of energy and leads to boning.
- In the case of the antibonding molecular orbital, the electron density in the internuclear region is severely reduced. There is a nodal plane in the internuclear region. Therefore, the repulsion between the nuclei is high. The antibonding molecular orbital is higher in energy than the individual atomic orbitals.
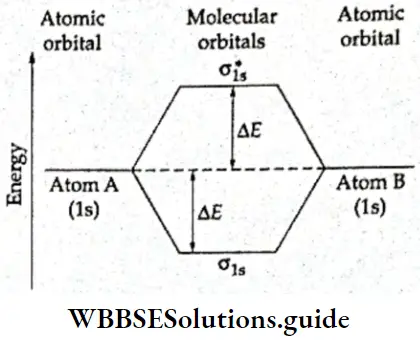
The respective energy levels of the two combining atomic orbitals (1s) and the bonding and antibonding molecular orbitals are given in Figure.
- The energy of the bonding molecular orbitals is lower than that of either of the combining atomic orbitals by an amount ΔE. The energy of the antibonding molecular orbital is more than that of the atomic orbitals by the same amount ΔE.
- In H2 molecule the two electrons are present in the bonding molecular orbital and the system is stabilized by an energy factor corresponding to 2ΔE. As already stated, the bonding molecular orbital is lower in energy than the atomic orbital by a factor ΔE, so putting an electron in a bonding molecular orbital stabilizes the system by an energy factor ΔE.
- As the hydrogen molecule has two electrons, the stabilization energy is 2ΔE. This energy parameter corresponds to the bond energy. The total energy of the two molecular orbitals is equal to the total energy of the two atomic orbitals. This is under the principle of conservation of energy.
The main differences between molecular orbitals and atomic orbitals as well as between a bonding molecular orbital and an antibonding molecular orbital are as follows.
Conditions for LCAO: The atomic orbitals which combine to form molecular orbitals should satisfy the following three conditions.
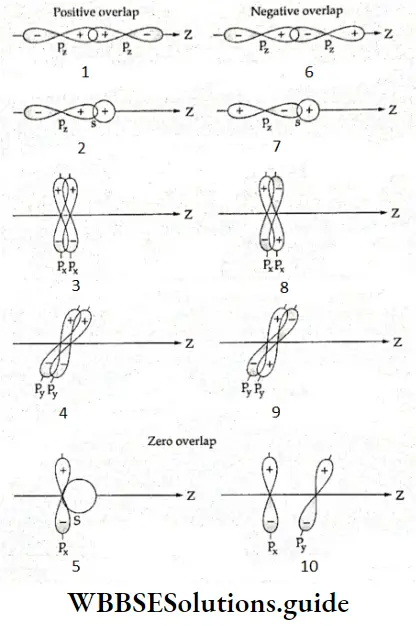
- The combining atomic orbitals should not differ significantly in energy. For example, a 1s orbital cannot overlap with a 2s orbital.
- The extent of overlap must be large, i.e., the atoms must be sufficiently close.
- The combining atomic orbitals must have the same symmetry along the intemuclear axis. By convention, the z-axis is considered to be the internuclear axis. Thus an s orbital cannot overlap with a px or py orbital, and a pz orbital cannot overlap with a px or py orbital.
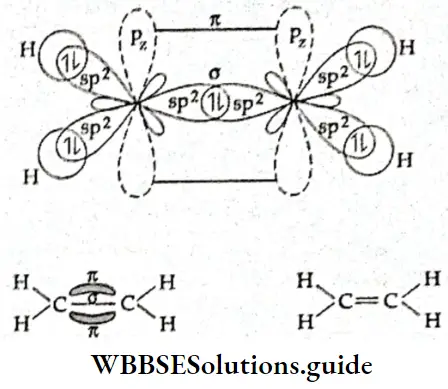
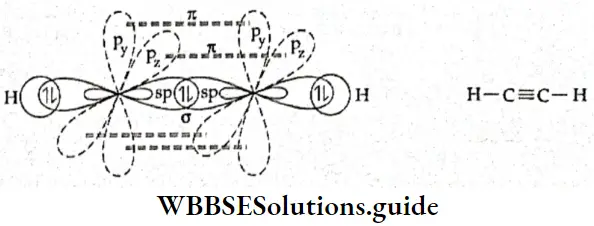
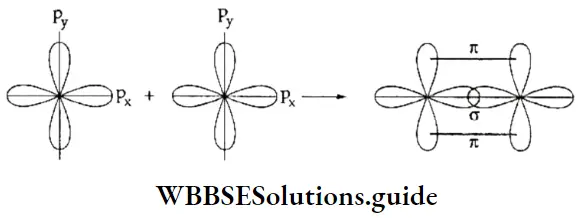
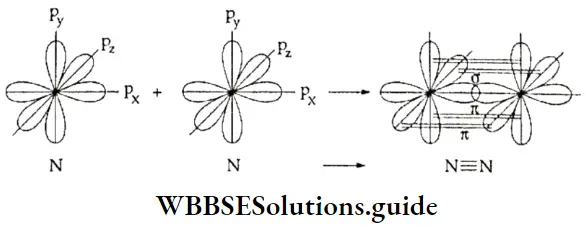
Types of molecular orbitals: Molecular orbitals of diatomic molecules are designated as σ, π, and δ. We will restrict our discussion to σ and π orbitals only. A σ orbital is formed when the overlap of atomic orbitals occurs along the intemuclear line (z-axis).
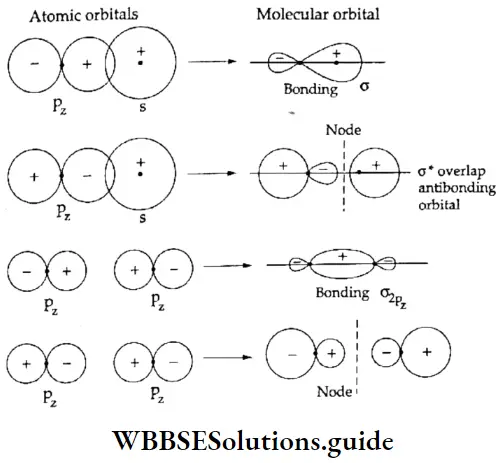
- An s orbital is spherically symmetrical, so when two s orbitals overlap a a molecular orbital is formed. A σ molecular orbital is also formed when an s orbital overlaps with a pz orbital or when two pz orbitals overlap with each other. In both these cases overlap takes place along the internuclear line. A σ molecular orbital is symmetrical about the bond axis. If the overlapping lobes have opposite signs, it gives rise to antibonding orbitals.
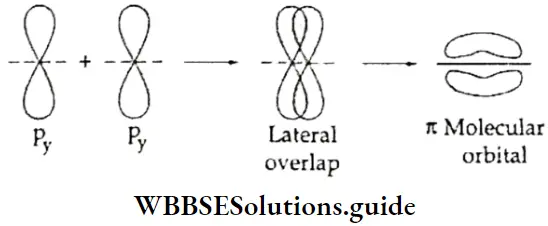
- You already know that lateral overlap of orbitals results in the formation of π bonds. Therefore, when a px orbital overlaps with another px orbital (or py with py) a π molecular orbital is formed. In this combination both the lobes of each combining orbitals are perpendicular to the internuclear line.
- The overlap does not occur along the internuclear line. In other words, a π molecular orbital is not symmetrical about the internuclear axis. In the case of π bonding molecular orbital, the electron density is large, above and below the internuclear line. The π* molecular orbital (antibonding) has a node between the two nuclei. All antibonding orbitals possess a nodal plane lying between the nuclei and perpendicular to the internuclear axis.
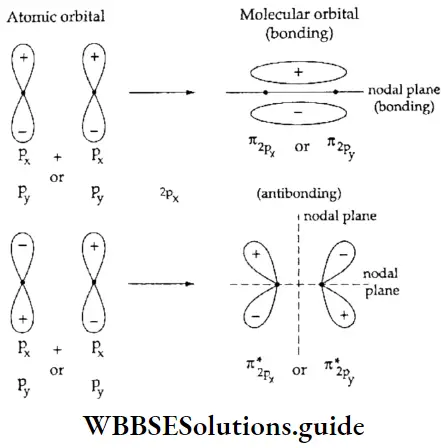
Here it should be understood that the overlap of an s orbital with px or py or a pz orbital with a px or py orbital gives a nonbonding combination. The orientation is not proper for a bond to be formed.
Order of energy of molecular orbitals: In the same way as atomic orbitals have definite energies and can be distinguished by quantum numbers, each molecular orbital has a definite energy and is specified by four quantum numbers.
- You are already familiar with the filling of atomic orbitals in accordance with the Pauli exclusion principle and Hund’s rule. The same applies to molecular orbitals. In the case of the molecular orbital method, the whole molecule is considered and the total number of electrons from all the constituent atoms are filled in the molecular orbitals.
- The order of energy of molecular orbitals has been determined experimentally by spectroscopy for the elements of the second period. The increasing order of energies of the molecular orbitals in homonuclear diatomic molecules is
⇒ \(\sigma 1 s<\sigma^* 1 s<\sigma 2 s<\sigma^* 2 s<\sigma 2 p_z<\left(\pi 2 p_x=\pi 2 p_y\right)<\left(\pi^* 2 p_x=\pi^* 2 p_y\right)<\sigma^* 2 p_z\)
“Polar covalent bond, types, properties, and differences from non-polar bonds, WBCHSE notes”
The energies of \(\pi 2 p_x \text { and } \pi 2 p_y\) molecular orbitals are the same and they form a pair of degenerate orbitals. Similarly the \(\pi^* 2 p_x \text { and } \pi^* 2 p_y\) orbitals are also degenerate. The energies of \(\sigma 2 \mathrm{p}_z \text { and } \pi 2 \mathrm{p}_x / \pi 2 \mathrm{p}_y\) molecular orbitals are very close and the order given above is correct for 02 and F2 molecules. For other lighter elements of the second period of the periodic table, there is a reversal in the order of the energy of \(\pi 2 \mathrm{p}_x / \pi 2 \mathrm{p}_y \text { and } \sigma 2 \mathrm{p}_z\) orbitals. The order now becomes
⇒ \(\sigma 1 \mathrm{~s}<\sigma^* 1 \mathrm{~s}<\sigma 2 \mathrm{~s}<\sigma^* 2 \mathrm{~s}<\left(\pi 2 \mathrm{p}_x=\pi 2 \mathrm{p}_y\right)<\sigma 2 \mathrm{p}_z<\left(\pi^* 2 \mathrm{p}_x=\pi^* 2 \mathrm{p}_y\right)<\sigma^* 2 \mathrm{p}_z\).
Class 11 WBCHSE Chemistry: Polar Covalent Bond Definition And Examples
The main difference between the two is that for the lighter elements the energy of the \(\sigma 2 \mathrm{p}_z\), molecular orbital is higher than that of the \(\pi 2 \mathrm{p}_x \text { and } \pi 2 \mathrm{p}_y\) molecular orbitals.
- While establishing molecular orbitals we deal with the concept that orbitals of atom A mix with orbitals of atom B if they are matched in energy and symmetry. However mixing occurs in all orbitals of comparable energy having proper symmetry. The energy differences between Is and 2s orbitals is large and we are justified in neglecting this mixing. The energy difference between 2s and 2p orbitals is less and varies with effective nuclear charge.
- It increases drastically from 200 kJ mol-1 for lithium to 2500 kJ mol-1 for fluorine. The 2s orbital and 2pz orbital are matched in symmetry, and thus for the lighter elements mixing occurs. This phenomenon is the equivalent of hybridisation in valence bond theory.
Molecular Orbital Treatment of Homonudear Diatomic Molecules: The distribution of electrons among various molecular orbitals of a molecule is called the electronic configuration of the molecule. The stability and magnetic behaviour of the molecule depend on its electronic configuration.
Stability: A bonding orbital is lower in energy whereas an antibonding orbital is higher in energy than either of the two atomic orbitals which combine. Therefore, an electron in a bonding orbital leads to stabilization, and conversely an electron in an antibonding orbital leads to destabilisation.
Thus, the relative number of electrons in bonding and antibonding orbitals of the molecule give an idea about its stability. If the number of electrons in bonding orbitals is given by Nb and that in antibonding orbitals by Na, then the molecule will be stable if Nb>Na.
Bond order Tire stability of a molecule is given in terms of bond order. The bond order of a molecide is expressed as half of the difference between the number of electrons present in bonding and antibonding orbitals.
Bond order = \(\frac{1}{2}\left(N_b-N_a\right)\).
A positive bond order indicates a stable molecule whereas a negative or zero bond order indicates an unstable molecule. The value of the bond order decides the nature of the bond. Integral bond order values of one, two, or three correspond to single, double, and triple bonds respectively. The bond order is a useful parameter indicating characteristics of bonds. Bond order is inversely proportional to bond length.
Magnetic behaviour: The electronic configuration of a molecule indicates the magnetic behaviour of a molecule. The macroscopic magnetic properties of a substance arise from the total of the magnetic moments of its component atoms and molecules.
If all the electrons in the orbitals are paired then the molecule is diamagnetic. But in case of unpaired electrons present in the orbitals, the molecule is paramagnetic.
In diamagnetic substances, the magnetisation is in the opposite direction to that of the applied field. In paramagnetic substances, the net spin magnetic moments due to impaired electrons in the molecule can be aligned in the direction of the applied field.
Let us now discuss bonding in a few homonuclear diatomic molecules.
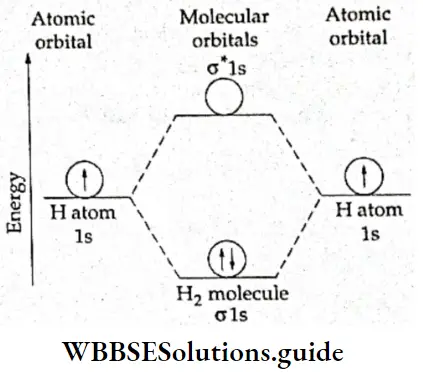
1. \(\mathbf{H}_2^{+}\) molecule ion This is the simplest diatomic molecule obtained as a transient species when hydrogen gas [H2(g)] is subjected to electric discharge at reduced pressure.
The single electron is present in the bonding, σ1s molecular orbital. The molecular orbital configuration is σ1s1. The bond order is 1/2(1- 0) = 1/2. A positive bond order implies a/ certain amount of stability. The molecule is paramagnetic in nature due to the presence of single unpaired electron.
2. H2 molecule The total number of electrons in the molecule is two (one from each hydrogen atom). The electrons are placed in the bonding molecular orbital and the electronic configuration is σ1s2.
The molecular orbital diagram is shown in Figure. The bond order of the molecule = 1/2 (2 – 0) = 1. This implies that the two atoms are held together by a single bond. Since there is no unpaired electron, the molecule is diamagnetic.
The bond order of the molecular ion, \(\mathrm{H}_2^{+} \text {is } 1 / 2\) whereas that of hydrogen molecule is 1. This shows that the H2 molecule is more stable than the \(\mathrm{H}_2^{+} ion\). This is also obvious from their bond lengths which is 106 pm for \(\mathrm{H}_2^{+} ion\) ion and 74 pm for H2 molecules.
3. He2 molecule Each He atom has two electrons. Therefore, the He2 molecule has four electrons arranged as \((\sigma 1 s)^{\frac{2}{2}}\left(\sigma^* 1 s\right)^2\). The bond order is 1/2(2 – 2) = 0, which indicates that the molecule does not exist. Similarly it can be shown that the Be2 molecule does not exist.
4. Li2 molecule The electronic configuration of Li is 1s22s1. Thus in Li2, there are six electrons and the electronic configuration of the molecule is \((\sigma 1 s)^2(\sigma 1 s)^2(\sigma 2 s)^2(a2s)2\). This is shown in Figure.
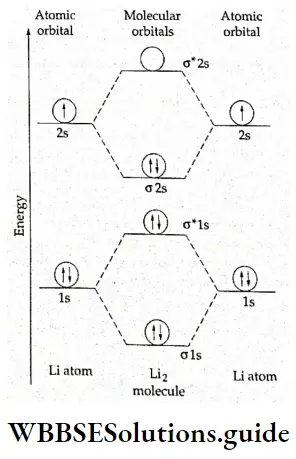
The inner shell, i.e., σ1s and \(\sigma^{\circ} 1 \mathrm{~s}\), does not contribute much to the bonding. Therefore, the electronic configuration of the molecule can be written as KK(σ2s)2. KK refers to a closed K shell which includes two molecular orbitals \((\sigma 1 s)^2 \text { and }\left(\sigma^{\circ} 1 s\right)^2\).
The bond order is 1 [1/2(4 – 2)1 which shows that the Li2 molecule is stable. In fact, diamagnetic, Li2 molecules do exist in vapour phase.
5. B2 molecule Each boron atom has five electrons and the electronic configuration is \(1 s^2 2 s^2 2 p^1\). This is the first instance in the discussion where we have come across a molecular system with an electron in a p orbital. As stated earlier for lighter molecules like B2 and C2 the doubly degenerate π2p orbitals are lower in energy than the σ2pz orbital.
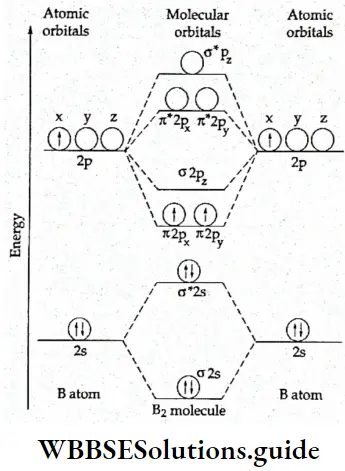
Therefore, the electronic configuration of the B2 molecule is \((\sigma 1 s)^2\left(\sigma^* 1 s\right)^2(\sigma 2 s)^2\left(\sigma^* 2 s\right)^2 \pi^1 2 p_x=\pi^1\) or \(operatorname{KK}(\sigma 2 s)^2\left(\sigma^* 2 s\right)^2 \pi^1 2 p_x=\pi^1 2 p_y\).
“WBCHSE Class 11, chemistry notes, on polar covalent bond, molecular polarity, and applications”
The molecular orbital diagram is given in Figure. The bond order is 1/2(6 – 4) = 1, which shows that the molecule is stable and, having two unpaired electrons, paramagnetic. The bond energy is 290 kJ mol-1 and the bond length is 159 pm.
6. C2 molecule Since the carbon atom has six electrons \(\left(1 s^2 2 s^2 2 p^2\right)\), the C2 molecule will have 12 electrons and the electronic configuration may be written as \(\left(\sigma_{1 s}\right)^2\left(\sigma^* 1 s\right)^2(\sigma 2 s)^2\left(\sigma^* 2 s\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\)
or \(\mathrm{KK}\left(\sigma^2\right)^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\)
The bond order is 1/2(8 – 4) = 2, which shows the presence of a double bond in the molecule. But both pairs of electrons constituting the double bond are present in the π molecular orbitals.
Therefore, the double bond comprises two π bonds and not one σ and one π bond as in other molecules. There is no unpaired electron, so that the molecule is diamagnetic and exists in the vapour phase only. The bond energy is 620 kJ mol-1 and the bond length is 131 pm.
7. N2 molecule Each nitrogen atom has seven electrons (1s2 2s2 2p3) therefore, the N2 molecule will have fourteen electrons and the electronic configuration is
⇒ \(\left(\sigma_{1 s}\right)^2\left(\sigma^* 1 s\right)^2\left(\sigma_2\right)^2\left(\sigma^* 2 s\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\left(\sigma^2 \mathrm{p}_z\right)^2\)
or \(\mathrm{KK}(\sigma 2 s)^2\left(\sigma^{\circ} 2 s\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\left(\sigma 2 \mathrm{p}_z\right)^2\).
The bond order of the N2 molecule is 1/2(10 – 4) = 3.
Therefore, in the diamagnetic nitrogen molecule, the two atoms are held by a triple bond. The bond energy is 945 kJ mol-1 and the bond length is 110 pm.
Some ionic species formed by the N2 molecule The \(\mathrm{N}_2^{+}\) ion is formed by removing an electron from the N2 molecule present in the highest occupied molecular orbital, i.e., \(\sigma 2 \mathrm{p}_{\mathrm{z}}\). The electronic configuration for the ion is
⇒ \(\mathrm{KK}(\sigma 2 \mathrm{~s})^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2 \sigma^1 2 \mathrm{p}_z\)
The presence of an unpaired electron indicates that the ion will be paramagnetic. The bond order is 2.5, suggesting that it is less stable than N2. This is expected as an electron has been removed from a bonding molecular orbital.
Comprehensive Guide To Polar Covalent Bonds For WBCHSE Class 11
The \(\mathrm{N}_2^{-}\) ion is formed by adding an electron to the lowest unoccupied molecular orbital of N2, i.e., the \(\pi^{\circ} 2 p_x\) molecular orbital. The electronic configuration of \(\mathrm{N}_2^{-}\) is
⇒ \(\mathrm{KK}(\sigma 2 \mathrm{~s})^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\left(\sigma^2 2 \mathrm{p}_z\right)\left(\pi^* 2 \mathrm{p}_x\right)^1\)
The bond order is 2.5 and the molecule is paramagnetic.
Considering the bond order values of the species N2, N+2, and N–2, they can be arranged in order of increasing stability as
\(\mathrm{N}_2, \mathrm{~N}_2^{+} \text {and } \mathrm{N}_2^{-} \text {, }\).
8. O2 molecule Each oxygen atom has eight electrons \(\left(1 s^2 2 s^2 2 p^4\right)\). Therefore the total number of electrons in the O2 molecule is 16. The electronic configuration is
⇒ \((\sigma 1 \mathrm{~s})^2\left(\sigma^* 1 \mathrm{~s}\right)^2(\sigma 2 \mathrm{~s})^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\sigma^2 \mathrm{p}_z\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\left(\pi^* 2 \mathrm{p}_x\right)^1=\left(\pi^* 2 \mathrm{p}_y\right)^1\)
or \(\mathrm{KK}(\sigma 2 \mathrm{~s})^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\sigma 2 \mathrm{p}_z\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\left(\pi 2 \mathrm{p}_x\right)^1=\left(\pi 2 \mathrm{p}_y\right)^1\).
The energy level diagram of the molecule is shown in Figure. As oxygen is a heavier molecule the energy level sequence of molecular orbitals is different from that of B2, C2, etc. The \(\sigma 2 p_z\), molecular orbital is lower in energy than the \(\pi 2 p_x\) molecular orbital.
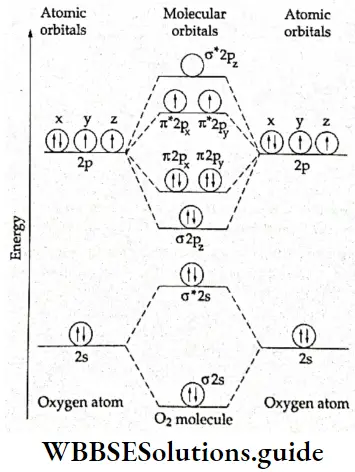
The \(\pi^{\circ} 2 p_x \text { and } \pi^* 2 p_y\) orbitals are singly occupied (by Hund’s rule). The presence of unpaired electrons gives rise to paramagnetism which is in accordance with experimental findings.
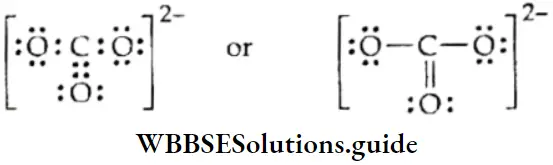
Thus, the molecular orbital theory successfully explains the paramagnetic nature of oxygen molecule, which was not explained by the valence bond depiction \((: \ddot{\mathrm{O}}=\ddot{\mathrm{O}}:)\) of the molecule. The bond order is 1/2(8-4) = 2, which corresponds to a double bond. The bond energy is 498 kJ mol-1 and the bond length is 121 pm.
Some ionic species formed by O2 molecule The \(\mathrm{O}_2^{+}\) ion contains one electron less than the O2 molecule whereas the \(\mathrm{O}_2^{-} \text {and } \mathrm{O}_2^{2-}\) ions contain one and two electrons more than the O2 molecule respectively. The electronic configuration, magnetic behaviour, and bond order of the ionic species are summarised below.

The stability of the O2 molecule and the ionic species follows the order
∴ \(\mathrm{O}_2^{+}>\mathrm{O}_2>\mathrm{O}_2^{-}>\mathrm{O}_2^{2-} \text {. }\)
Similarly, the §ond length which is inversely proportional to bond order follows the sequence
∴ \(\mathrm{O}_2^{2-}>\mathrm{O}_2^{-}>\mathrm{O}_2>\mathrm{O}_2^{+}\)
9. F2 molecule The fluorine atom has nine electrons \(\left(1 s^2 2 s^2 2 p^5\right)\). Therefore, the F2 molecule contains 18 electrons arranged in the molecular orbitals as \(\mathrm{KK}(\sigma 2 \mathrm{~s})^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\sigma 2 \mathrm{p}_z\right)^2\left(\pi 2 \mathrm{p}_x\right)^2=\left(\pi 2 \mathrm{p}_y\right)^2\left(\pi^* 2 \mathrm{p}_x\right)^2=\left(\pi^* 2 \mathrm{p}_y\right)^2\).
The bond order is one and the molecule is diamagnetic. The bond energy is 159 kJ mol-1 and the bond length is 143 pm.
10. Ne2 molecule The electronic configuration of the Ne2 molecule with 20 electrons (since each neon atom has 10 electrons) is
⇒ \(\begin{aligned}
\operatorname{KK}(\sigma 2 s)^2\left(\sigma^* 2 \mathrm{~s}\right)^2\left(\sigma 2 \mathrm{p}_z\right)^2\left(\pi 2 \mathrm{p}_x\right)^2 =\left(\pi 2 \mathrm{p}_y\right)^2 \\
\left(\pi^* 2 \mathrm{p}_x\right)^2 =\left(\pi^* 2 \mathrm{p}_y\right)^2\left(\sigma^* 2 \mathrm{p}_z\right)^2
\end{aligned}\)
The bond order is zero, which implies that the Ne2 molecule does not exist. In other words, neon is 1 monoatomic.
Example 1. Two p orbitals of one atom and two p orbitals of another atom combine to form molecular orbitals. How mam/ molecular orbitals will result from this combination? Give all the possibilities.
Solution:
Given
Two p orbitals of one atom and two p orbitals of another atom combine to form molecular orbitals.
We know that there are three degenerate p orbitals and the electronic configuration of each atom may be \(\mathrm{p}_x \mathrm{p}_y \text { or } \mathrm{p}_x \mathrm{p}_z \text { or } \mathrm{p}_y \mathrm{p}_z\).
Thus various probabilities are possible which are shown below.

The overlap of px or py with pz or px with py will give a nonbonding combination of orbitals.
Example 2. Arrange the following in order of increasing stability. \(\mathrm{H}_2, \mathrm{H}_2^{+}, \mathrm{H}_2^{-}, \mathrm{H}_2^{2-}\)
Solution:

Thus, the order of increasing stability is \(\mathrm{H}_2^{2-}<\mathrm{H}_2^{-}=\mathrm{H}_2^{+}<\mathrm{H}_2\)
“Polar covalent bond, examples, characteristics, and significance, WBCHSE Chemistry”
Example 3. Use the molecular orbital theory to explain why He2 does not exist.
Solution:
Each He atom has 2 electrons. Thus He2 has 4 electrons and the electronic configuration of the molecule is \((\sigma 1 s)^2\left(\sigma^* 1 s\right)^2\).
BO = 1/2 (2 – 2) = 0. Thus He2 does not exist.
Example 4. What is the bond order of the \(\mathbf{H}_2^{+}\) ion and the H2 molecule? Why is the bond length in H2 shorter than that in \(\mathbf{H}_2^{+}\)?
Solution:
The bond order of \(\mathbf{H}_2^{+}\) is 0.5 and that of H2 is 1. The bond order value for the H2 molecule is more than that for the \(\mathbf{H}_2^{+}\) ion. Since bond length decreases as bond order increases, the bond length of H2 is shorter.

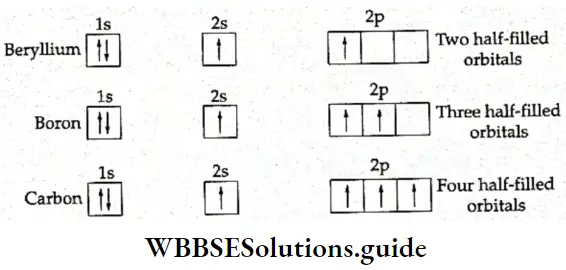

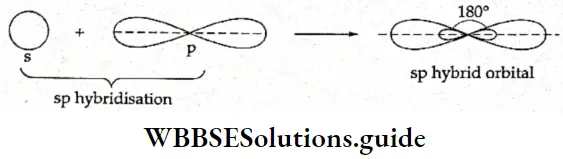

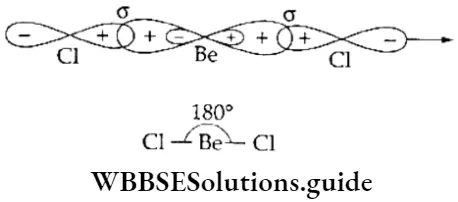
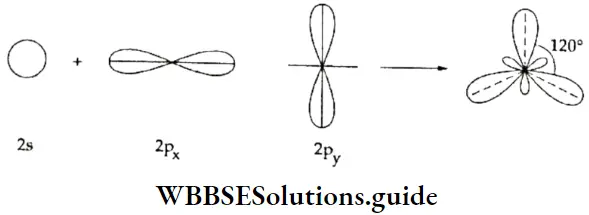
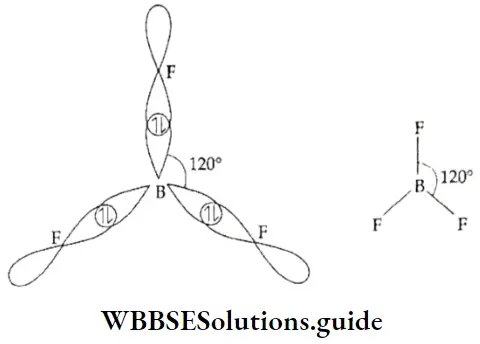
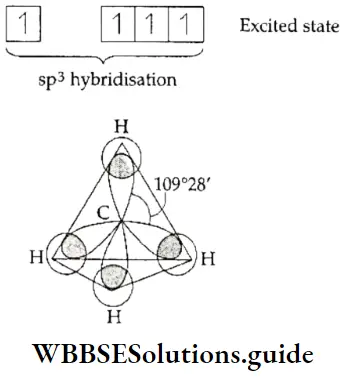
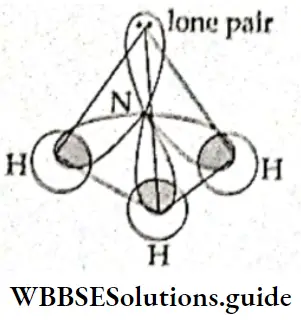
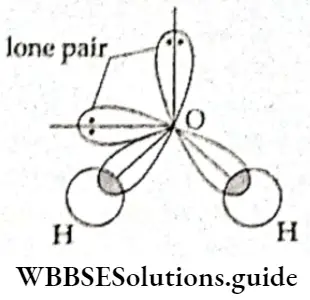
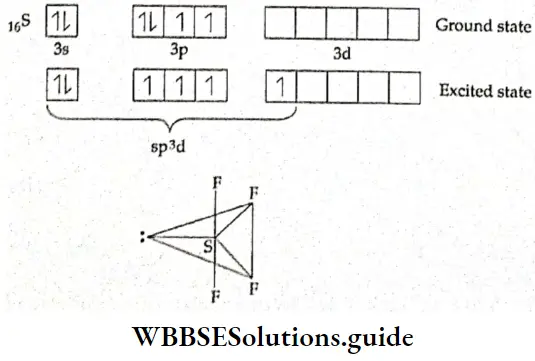
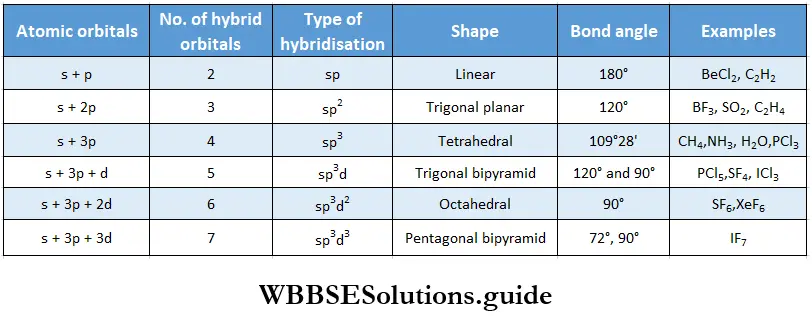


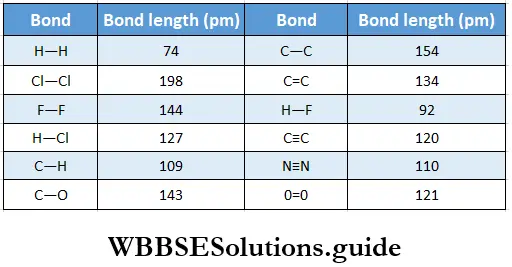
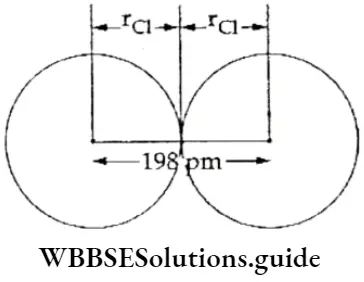
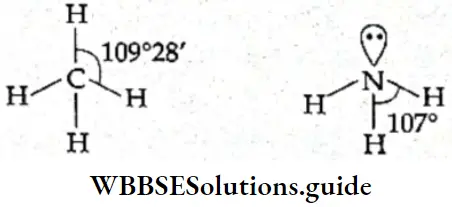


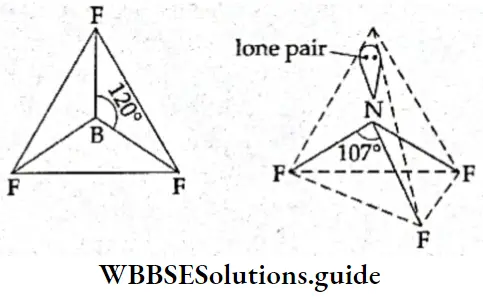
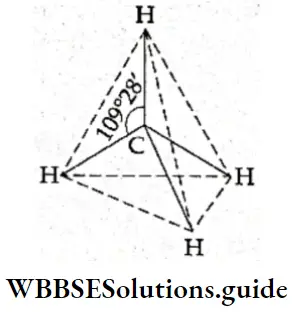
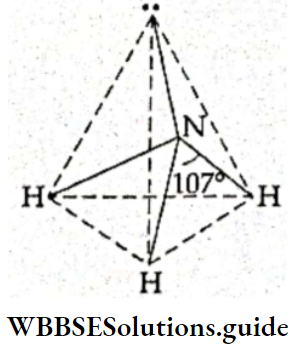
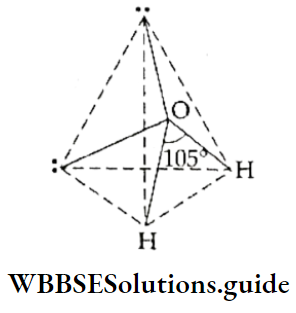
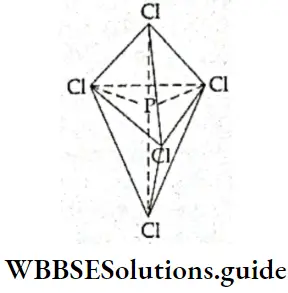
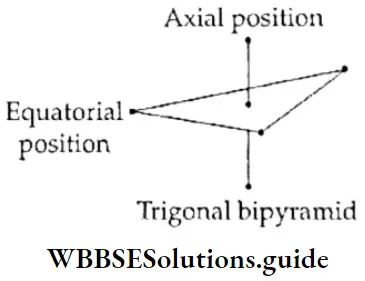
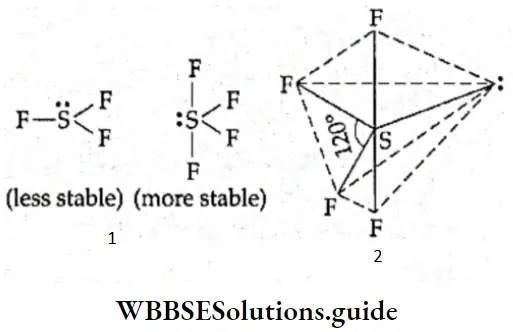
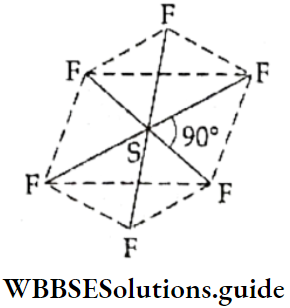
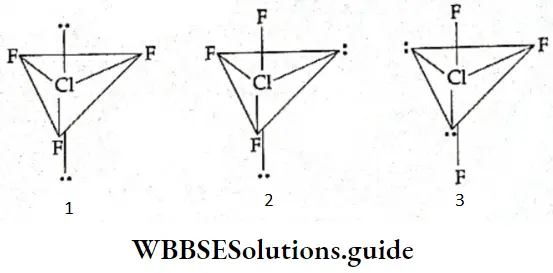
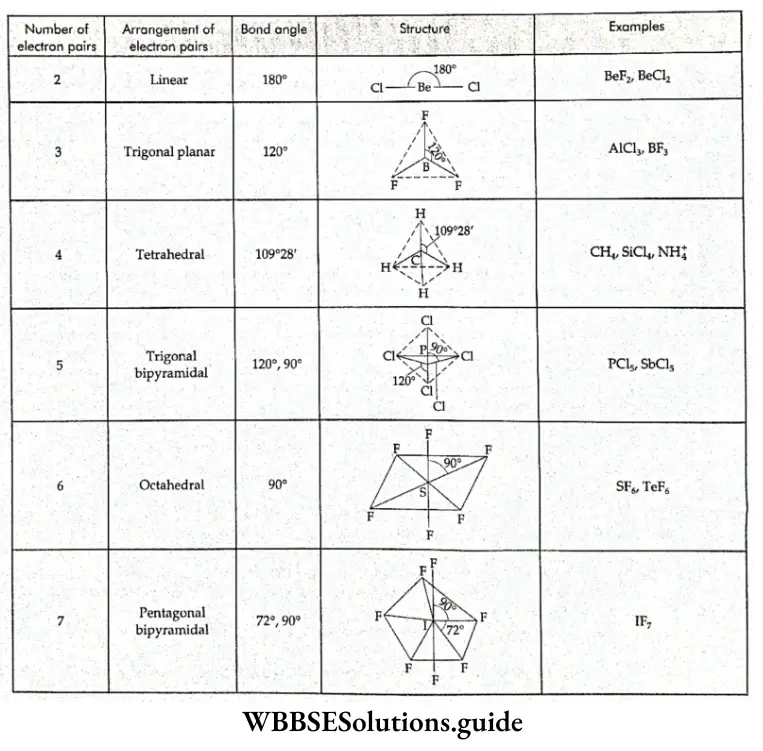 If, on the other hand, the central atom has both bonded and nonbonded electrons (lone pairs) in its valence shell, the molecule has an irregular geometry. This is because the different electron pairs do not repel each other equally.
If, on the other hand, the central atom has both bonded and nonbonded electrons (lone pairs) in its valence shell, the molecule has an irregular geometry. This is because the different electron pairs do not repel each other equally.