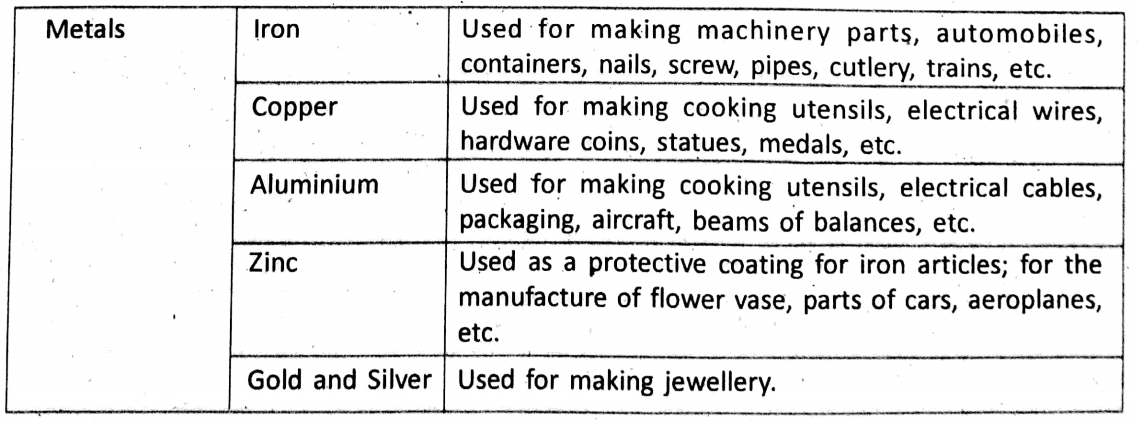
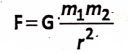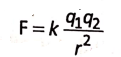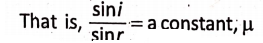Chapter 1 Physical Environment Force And Pressure
What is the exact connection between force and motion?
The exact connection between force and motion
In the previous classes, we have learned about the motion of an object (or a body) along a straight path in terms of its displacement, velocity, and acceleration. Here, we are to learn about the causes of such motion.
Read and Learn more WBBSE Notes For Class 8 General Science And Environment
Let us take some common questions from our everyday experience :
Why does o football (at rest) move when kicked? What makes a moving ball on the ground, to stop on its own? What makes a spring or a rubber band to increase or decrease in length? Why does a body accelerate or decelerate? Why does a body move in a circular path ?…. It is our common experience that an external force is to be applied in each case.
Chapter 1 Physical Environment Effects Of Force
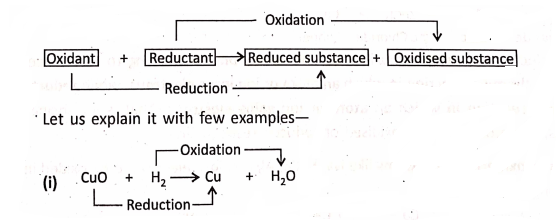
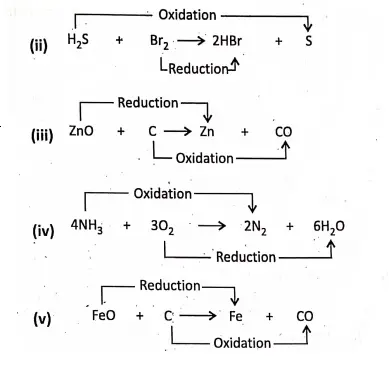
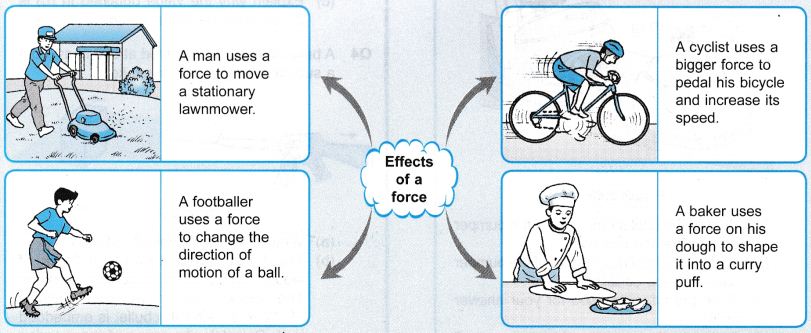
A force can be realized only by the effects that it can produce on the objects. In the effects of force are shown. Considering the effects of force can be stated as follows :
- A force can cause motion in a stationary body.
- A force can stop a moving body.
- A force can change the direction of motion of a body.
- A force can change the speed of a moving body.
- A force can change the shape and size of a body.
Thus, we can define that force is the external cause acting on a body which changes or tends to change the state of rest or of uniform motion, or the direction of motion, or the shape and size of a body.
Chapter 1 Physical Environment Newton’s Laws Of Motion
Sir Isaac Newton put forward three laws of motion related to all moving bodies in the universe in 1687. These laws are known as Newton’s laws of motion. These were published in the famous book Principia.
Basically, these laws are the foundations of Classical or Newtonian mechanics. Newton’s first law of motion gives the idea that when the net force on a body is zero, its state does not change.
That is, a body continues to be in its state of rest or of uniform motion in a straight line unless an external force is applied on it. So that force is the external cause which changes or tends to change the state of rest or of uniform motion of a body.
“WBBSE Class 8 General Science Chapter 1 notes, Physical Environment”
Thus, the first law defines the force only qualitatively. A force can change the state of motion of a body, i.e., a force can produce an acceleration in the body. Newton’s second law of motion relates force to acceleration when the net force is not zero.
That is, the second law gives the quantitative value of force. The second law of motion states that ‘the external net force acting on a body (whose mass remains unchanged) is equal to the product of its mass and acceleration and the acceleration takes place in the direction of the force applied.’
The first law and second law do not explain how does the force act on a body? This is explained in Newton’s third law of motion. The third law describes the relationship between the forces that two interacting bodies exert on each other (One of the forces is called ‘action’ and the other is called ‘reaction’).
The third law of motion states that ‘for every action, there is an equal and opposite reaction. The second law of motion describes the method to measure force, by the mathematical expression: F = ma or, force = mass x acceleration
Absolute units of force :
On this basis, the S.l. unit of force is newton (symbol N), and in. the C.G.S. system, the unit of force is dyne (symbol dyn).
Definition :
1 newton is the force which acting on a mass of 1 kg produces’ an acceleration of 1 m.s-2. That is 1N = 1 kg x 1 m.s-2.
1 dyne is the force which acting on a mass of Kg produces an acceleration of 1 cm.s-2. That is, 1 dyne = Kg x l cm.s-2
The mass of a body is the measure of its inertia :
From equation F = mo we have a = F/m. If an equal amount of force Facts on two bodies of mass mv m2 and av a2 are their respective accelerations then

That is, the acceleration produced in a body is inversely proportional to its mass. This means that a lighter body can be moved more easily than a heavier body.
Weight :
The weight of a body is the force with which the earth attracts the body toward its center. So, weight is the force of gravity. It is related to mass as follows :
W = mg or, weight = mass x acceleration due to gravity.
The value, of ‘g’, is 9.8 m.s”2 (in the S.l. system) and 980 cm.s-2 (in the C.G.S. system).
Gravitational units of force :
It is kilogram-force (symbol KGF) in S.l. system and gram force (symbol gf) in the C.G.S. system.
1 Kgf = Force of gravity on a mass of 1 kg
= 1 kg x g ms-2 = g kg.ms-2 = g newton = 9.8 newton
1 gf = Force of Gravity on a mass of 1g = 1g x g cms-2 = g g.cms-2 = g dyn = 980 dyn

Chapter 1 Physical Environment Measurement Of Force :
The force acting on a body (or the weight of a body) is measured by a spring balance. It contains a coiled spring having a hook at one end and the other end being free. The spring gets stretched when a force is applied to its free end.
The extent by which the spring gets stretched is a measure of the applied force and it is indicated by a pointer attached to the spring on a graduated scale. The reading on the scale indicates the magnitude of the force.
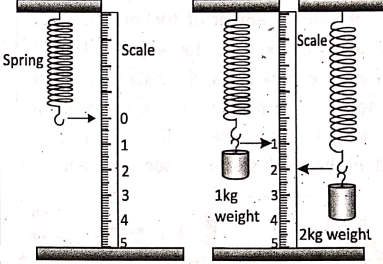
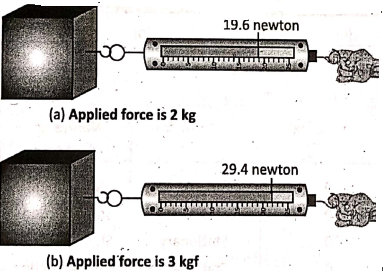
When the spring balance is held vertically it can be used to measure the weight of the body hung from its hook. While when the spring balance is held horizontally and its hook is attached to an object add pulled by hand it is said to measure the force applied.
If the reading on the scale of the spring balance is 2kg, we will mean that the applied force is 2 kgf or (2 x 9.8)N or 19.6 N. Also, for the reading 3 kg, the force is 3 kgf or (3 x 9.8) N or 29.4N.
Chapter 1 Physical Environment The Force Of Friction
It is our common experience that a ball moving on the ground gradually slows down and stops on its own after traveling a distance. According to Newton’s first law of motion, the ball after being pushed should have to continue its motion with a uniform velocity in absence of any external force.
“Class 8 General Science WBBSE notes, Physical Environment chapter”
This means the existence of an external force that is required to stop the ball. This force opposes the motion of the proving ball and occurs between the two surfaces which are in contact with each other and also act in a direction opposite to the direction of motion of the moving ball. This force is called the force of friction or simply frictional force.
Definition :
The force that always opposes the motion of an object over another object (in contact) when they slide or tend to slide over one another.
Chapter 1 Physical Environment Measurement Of Friction
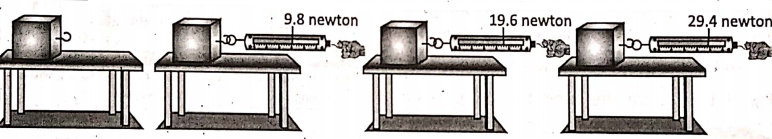
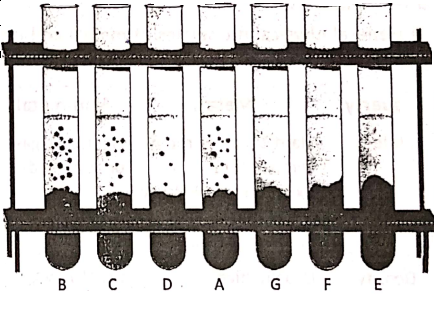
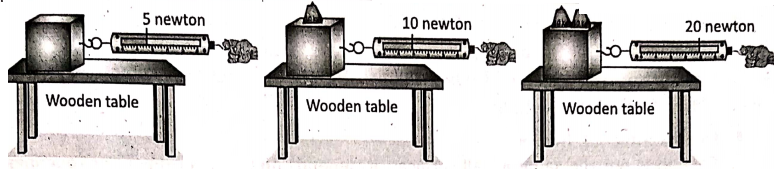
Place a wooden block on a table. Attach the hook of a spring balance with the block. Apply a pulling force to the block by hand. Note down the reading on the spring balance at each stage, as shown in Table.
| Observation No. |
The magnitude of pulling force (F) |
The magnitude of frictional force (f) |
Net force (F-f) |
State of the block |
Nature of friction |
| 1 |
0 |
0 |
0 |
Stationary |
No friction |
| 2 |
9.8N |
9.8N |
0 |
Stationary |
Static friction |
| 3 |
19.6 N |
19.6 N |
0 |
Stationary |
Static friction |
| 4 |
22.6N (say) |
22.6N (say) |
0 |
Stationary |
Limiting Friction |
| 5 |
29.4N |
20.4N (say) |
9 N (say) |
In motion |
Sliding friction |
when the block starts to slide. This reading on the spring balance gives the magnitude of the Note that in observation No. 4, for some pulling force, the frictional force becomes maximum, it is called limiting friction. Also, when the block starts to slide the magnitude of the force of friction cannot be determined directly from the reading of the spring balance.
Chapter 1 Physical Environment Different Kinds Of Friction
The frictional force offered when an object remains stationary on a surface even being pulled with a force is called static friction. The frictional force offered when sliding of an object takes place over a surface is called sliding friction.
Factors affecting friction :
1. Friction is not the same for all surfaces. It depends on the smoothness or roughness of the two surfaces which are in contact with one another. This is explained in.
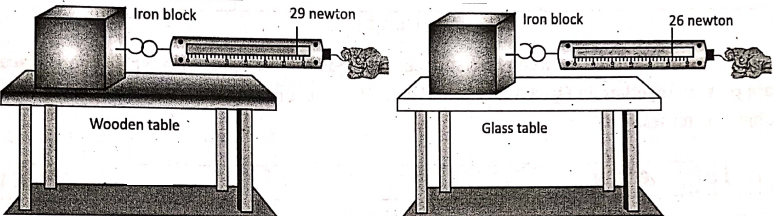
It is clear that different surfaces offer different amounts of friction to sliding. In general, rough surfaces offer more friction than smooth surfaces. As shown in suppose an iron block starts to slide over a wooden table when the pulling force is 19 newtons; while the same iron block starts to slide over a smooth glass table by a lesser amount of pulling force (say, 26 newtons).
2. The force of friction does not depend on the area of contact between two surfaces. In the frictional force remains the same for the horizontal and vertical position of a block over the wooden table. Note that the area of contact is more for the horizontal position of the block than that for. its vertical position.
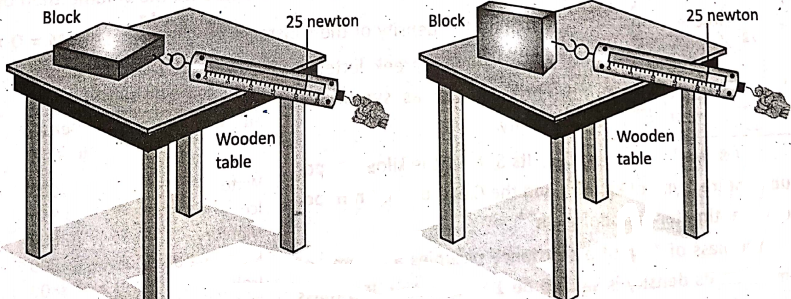
3. Frictional force between two surfaces depends on the force with which the surfaces are pressed together. In it is explained that frictional force increases as the weight of an object increases.
Chapter 1 Physical Environment Density
Let us take two identical plastic bottles of equal volume—one is completely filled with normal water and the other with kerosene oil. Hang them separately from the hook of two identical spring balances, From the reading of the spring balances, it is observed that normal water is heavier than kerosene oil, although the bottles are of equal volume.
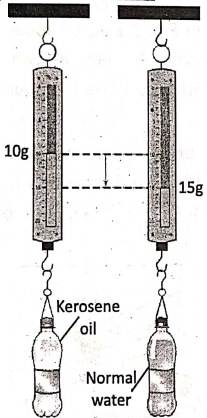
In this way, we can compare the ‘heaviness’ or ‘lightness’ of different, substances, by comparing their masses of the same volume. We do this with the help of a physical quantity, called the density of the substance.
Definition: The density of a substance is defined as its mass per unit volume.
If M be the mass of a substance and V be the volume then by definition the density of the substance is, D = M/V, i.e., M = D x V. Sometimes, greek letter P(rho) is used as a symbol of density. It’s S. I. unit is kilogram per cubic meter (symbol kg.m-3) and the C.G.S. unit is gram per cubic centimeter (symbol g.cm-3 )
| Substances |
Density in kg.m-3 |
| Water |
1000 |
| Ice |
916 |
| Mercury |
1360 |
| Kerosene oil |
800 |
| Iron |
7800 |
| Cork |
200 |
| Copper |
8900 |
| Gold |
19300 |
If a mass of 1 kg of a substance occupies a volume of 1m3 then its density is said to be 1 kgm-3. And, if a mass of 1g of a substance occupies a volume of 1 cm3 then its density is said to be 1 g.cm-3.
Relation between this two units :

“WBBSE Class 8 Environment Chapter 1, Physical Environment study guide”
Chapter 1 Physical Environment Pressure Of Liquid
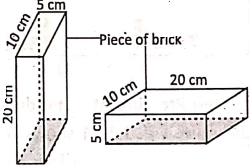
A brick exerts maximum pressure on the ground when it is kept with its longest side vertical while the pressure is minimum when the brick is placed with its shortest side vertical. So, a solid exerts pressure due to its weight only on the surface on which it is placed.
In class VI, we have learnt that pressure is the force acting normally on unit area to the surface of a substance. If F be the force acting normally on an area A, then, pressure P = normal force/area = F/A.
For example, the area of the pointed edge of a nail is too small. Hammering its head by a large force, the pressure at the edge becomes high enough and the nail can enter the surface easily.
“WBBSE Class 8 Physical Environment notes, General Science Chapter 1”
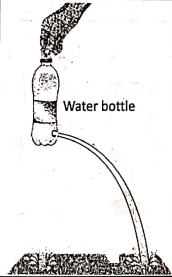
Similarly, a liquid also exerts pressure due to its weight on the bottom as well as on the walls of the container because of its tendency to flow. If a hole is made at any point on the wall of a water bottle, water spurts out normally with the wall. If we try to block the flow of water by a finger, we feel the pressure. The spurting out of water depends upon its pressure (but not on the force).
Definition :
Pressure at a point inside a liquid is the force exerted by the liquid, normally on a unit area around the point.
Features of liquid pressure :
- Liquid exerts pressure equally in all directions—upward, downward, and sideways (lateral pressure).
- Pressure at a point inside a liquid increases with the increase in its depth from its free surface.
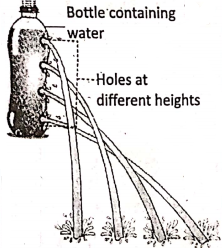
In four small holes are made on the wall of a water bottle at different heights. It is seen that as depth increases, more is the displacement of water on the ground. Here the depth of a point is measured from the free surface of the water.
Also, shows that the spurting out of water from the holes near the bottom of a wide tumbler and a big water bottle. Although the tumbler contains about 8-10 times more water than the bottle, but the depth of water from the free surface being larger in the bottle, creates more pressure to reach water a longer distance on the ground.

3. Pressure at the same depth is different in different liquids. It increases with the density of the liquid. shows that normal water exerts more pressure than kerosene oil at the same depth from the free surface.
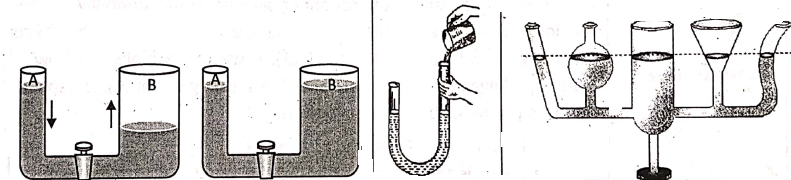
4. A liquid seeks its own level. Pressure determines the direction of the flow of a liquid. when the stopcock is opened, water flows from tube A to tube B, until water attains the same height in both tubes. water is found to attain the same height m both the limbs of a U-shaped tube. Also, shows that water attains the same height in all the containers.
Factors affecting the pressure at a point inside a liquid :
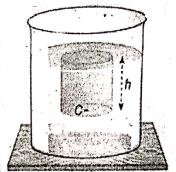
It depends directly on the following three factors :
- Depth of the point from the free surface (h).
- The density of liquid (p), and
- Acceleration due to gravity (g).
Mathematically, P = hpg = depth x density of the liquid x acceleration due to gravity.
Chapter 1 Physical Environment Air Pressure
Each and every substance has weight and anything that has weight can exert pressure. Air is light no doubt, but it is not weightless. So, air can exert pressure.
The air above the earth’s surface is considered to be divided into several air columns. Each and every air column has to bear the weights of the columns above. Hence, the weight of air column exerts a thrust (a force acting normally) on the earth’s surface, which gives rise to atmospheric pressure.
Definition :
The thrust exerted by an air column per unit area on the earth’s surface is called atmospheric pressure. The pressure at sea level is taken as the normal atmospheric pressure. It has been found by experiments that the pressure exerted by the atmosphere on a 1 cm2 area at sea level is about 1.033 kgf.
Naturally, the larger the height of a place above the sea level, the lower is the pressure. Air exerts pressure equally in all directions—upward, downward, and sideways (lateral pressure) and so we do not. feel it. ‘
Chapter 1 Physical Environment Measurement Of Air Pressure :
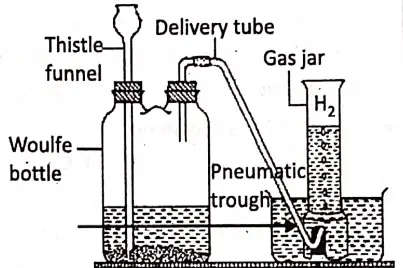
Torricelli’s experiment provides us with a method for measuring atmospheric pressure. Take a hard glass tube of about 1 meter in length closed at one end and fill it completely with pure mercury such that no air bubble remains inside. Holding open end of the tube with the thumb, the tube is inverted vertically into a trough of mercury and the thumb is then removed quickly.
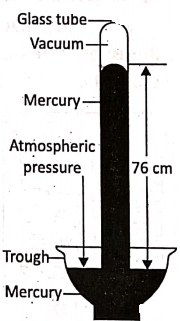
It is observed that the mercury level in the tube falls (due to gravity) till its height from the mercury level in the trough becomes 76 cm. This happens when the pressure exerted by the mercury column in the tube becomes equal to the atmospheric pressure on mercury in the trough. Thus, the mercury column of height of 76 cm is a measure of atmospheric pressure.
“Class 8 WBBSE General Science Chapter 1, Physical Environment summary”
As the density of water is low (1000 kgm-3), Torricelli argued that air pressure would push water up to a height equal to about 10.34 m. So, air pressure forces water up to a height of about 10.34 m and mercury up to a height of 76 cm. That is why a lift pump can not made to lift water beyond a height of 10.34 m.
Chapter 1 Physical Environment Buoyancy, Immersion, And Archimedes’ Principle
If a piece of cork or an empty can is pushed into the water, we feel an upward force acting on the object so that we find it difficult to push it further into the water. This upward force is buoyant force and the property of liquid to exert such a force is called buoyancy.
Definition:
When a body is partially or completely immersed inside a liquid, it experiences an upward force. This upward force is known as upthrust or buoyant force. It is due to the buoyant force that a body partially or completely in a liquid, appears to be lighter than its true weight.
Factors on which the buoyant force depends on:
- The volume of the body immersed and
- The density of the liquid.
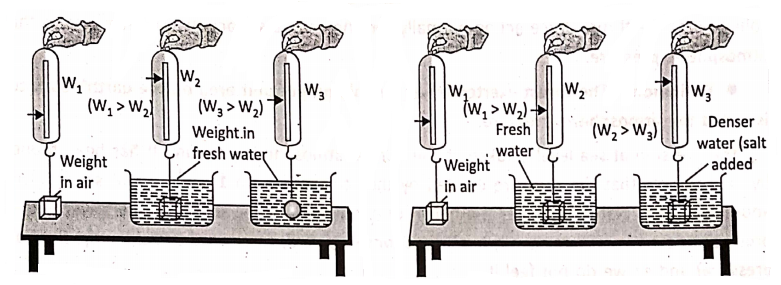
More volume means more upthrust shows that the more is the volume of the body immersed in a liquid more is the upthrust as more amount of liquid is displaced. Denser the liquid, more is the upthrust indicates that the denser is liquid, the more is the upthrust.
Chapter 1 Physical Environment Archimedes’ Principle
When a body is partially or completely immersed in a liquid (or a fluid), it experiences an apparent loss in weight (due to upthrust) and this apparent loss in weight is equal to the weight of the liquid displaced by the immersed portion of the body.
Sinking and flotation of a body inside a liquid:
When a body is immersed in a liquid, two forces act on it:
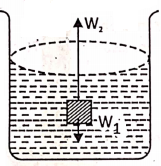
The forces are :
- Its weight, W1 acts vertically downward. This force has a tendency to sink the body.
- The upthrust or buoyant force, W2, is applied by the liquid on the body, acting vertically upwards. This force has a tendency to move the body upwards.
Depending upon the values of W1 and W2, the following three situations may arise :
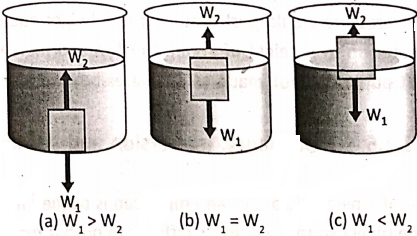
Case-1: W1 > W2, i.e., the weight of the body is greater than the buoyant force.
In this case, the net force is (W1– W2), which acts on the body vertically downward and the body will sink by the action of net force. Such a situation arises when the density of the body is nearly more than that of the liquid.
Case-2: W1= W2, i.e., the weight of the body is equal to the buoyant force.
In this case, the net force acting on the body is zero and the body floats just under the liquid surface. Such a situation arises when the density of the body is nearly equal to the density of the liquid.
Case-3: W1= W2, i.e., the weight of the body is less than the buoyant force.
In this case, the net force is (W2-W1), which acts on the body vertically upward and the body will remain in a floating condition with some portion above the liquid surface. Such a condition happens when the density of the body is less than the density of the liquid.
Examples of immersion and flotation :
- An iron nail sinks in water, but a ship made of iron floats. This is because the weight of iron nail is greater than the upthrust of water on it (as the density of iron is greater than that of water).On the other hand, a ship having a larger surface area displaces much more amount of water by its submerged part such that the weight of the displaced water becomes equal to the total weight of the ship and thus it floats.
- An egg sinks in freshwater but floats in a strong salt solution.
- Icebergs being lighter than water floats in the sea.
- An iron nail floats on mercury. This is so because the density of iron is less than that of mercury.
- It is easier for a man to swim in seawater than in river water. Due to the presence of salt and other minerals, the density of seawater is much more than that of river water. So, when a part of the body is submerged in seawater, it experiences a larger amount of upthrust than the upthrust experienced in river water. Thus, a man swims in sea water more easily.
- A ship submerges more as it sails from seawater to river water.
Chapter 1 Physical Environment Forces Active Without Contact
Non-Contact force
A force that can be exerted by an object over another object even from a distance without coming in physical contact with each other is known as a non-contact force. Examples of non-contact forces are :
- Gravitational force,
- Electrostatic force and
- Magnetic force.
Features of non-contact forces :
The magnitude of the non-contact forces depends upon
1. The distance between the objects and
2. Nature of the medium between two objects (except for gravitational force).
In general, the non-contact force is inversely proportional to the square of the distance between two objects.
Chapter 1 Physical Environment Gravity and Gravitation :
If we drop a stone from a height, it falls down towards the earth. Also, a stone thrown upwards falls back to the earth. Similarly, a leaf falls down from a tree, a fruit also falls down, rain falls down, and water in a river flows downwards. Also, we feel a downward pull on keeping a stone on one palm.
This is because the earth exerts a gravitational force of attraction on all the bodies and pulls them downwards. The force with which the earth pushes the objects towards its center is called the force of gravity (or simply gravity).
“WBBSE Class 8 Science Chapter 1 notes, Physical Environment PDF”
Isaac Newton made the bold statement that every object in the universe pulls every other object with a certain force, similar to the force that pulls a body falling towards the earth.
The force of attraction (or pull) exerted between any two objects in the universe possessing mass is called gravitation or the gravitational force, In this sense, the gravitation between the earth and any other object is called gravity.
Chapter 1 Physical Environment Newton’s Law of Gravitation :
In 1687 Isaac Newton published his work on the law of gravitation in Mathematical Principles of Natural Philosophy. The law is applicable only for two-point masses. Although the earth, sun, moon, and other planets are very large in size, but their total mass is considered to be concentrated at their centers.
Statement:
Every particle in the universe (small or big) attracts every other particle along the line joining their centers with a force that is directly proportional to the product of their masses and inversely proportional to the square of the distance between their centers.
Newton introduced a mathematical equation to calculate gravitational force :
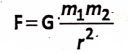
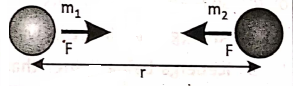
where two particles of masses m1 and m2 are separated by a distance r between their centers; G is a constant called the universal gravitational constant. In S.l. system, the vajue G is 6.67 x 10-11 N.m2 . kg-2. The values of ‘G’ is the same everywhere in the universe. The above equation is applicable for all bodies having a spherical shape placed anywhere in the universe even in space.
However, if r increases, the magnitude of F decreases. Theoretically, the effect of gravitation on any object happens to exist up to an infinite distance. Actually, after a certain distance, such an effect becomes negligible. The gravitational force between two stones is so weak that it cannot be detected easily.
Acceleration due to gravity ort the surface of the earth: It is the acceleration produced in a body due to the force of gravity.
Suppose a body of mass m is placed on the surface of the earth. If M be the mass and R be the radius of the earth, then by Newton’s law of gravitation, the force of attraction between these two bodies is

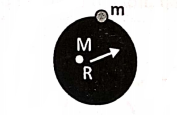
It is evident from this relation that the value of g is
- Maximum at the surface of the earth,
- More at the poles than at the equator,
- Decreases with a height from the surface of the earth and
- Decreases with depth from the surface of the earth.
The direction of gravity :
The direction of gravity is always toward the earth. However, when an object is thrown upwards in any direction (making an angle with the horizontal plane) [like a cricket ball, javelin, discus throw], the object reaches the ground following a curved path. This is so because the speed of the moving object continuously changes from point to point due to the earth’s gravity.
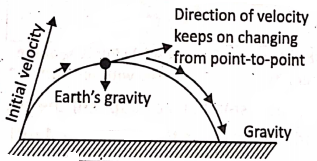
Motion due to gravity :
What we see if a stone and a piece, of paper (or a feather), are allowed to fall at the same time from the top of a tower ?—We see the stone reaches earlier than the piece of paper. Galileo proved the fact that while falling, the objects are to overcome air resistance.
The size and shape of the piece of paper being larger, the air resistance is larger for it. If there is no air, the two objects reach the ground at the same time. Galileo proved the fact from his famous Guinea-Feather Experiment.
Laws of falling bodies: There are three laws:
- All freely falling bodies whether heavy or light fall with the same rapidity.
- The velocity attained by a freely falling body at a particular instant of time is directly proportional to the time of fall.
- The height traversed by a freely falling body at a particular instant of time is directly proportional to the square of the time of fall.
Chapter 1 Physical Environment Concept Of Electrostatic Force And Charge
If we bring a plastic comb near small bits of paper, the comb does not attract them. But, when the same comb is rubbed on dry hair or on nylon or woolen substance and brought near the paper bits, it attracts them. If the hair is not dry, then rub.
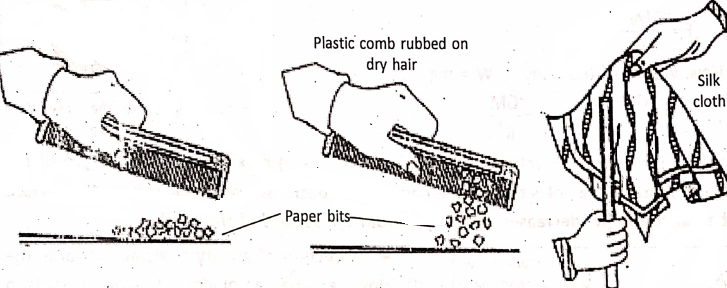
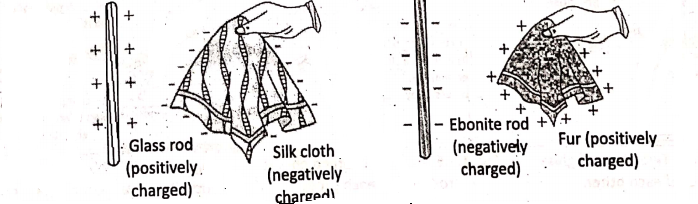
Similarly, an ordinary glass rod rubbed with silk cloth, attracts small bits of paper. If the silk cloth is brought near the paper bits, it is noticed the silk cloth also attracts them. In a similar way, a balloon rubbed with a woolen substance sticks with a wall, which falls down if not rubbed so. Also, two inflated balloons rubbed with the same woolen substance repel each other.
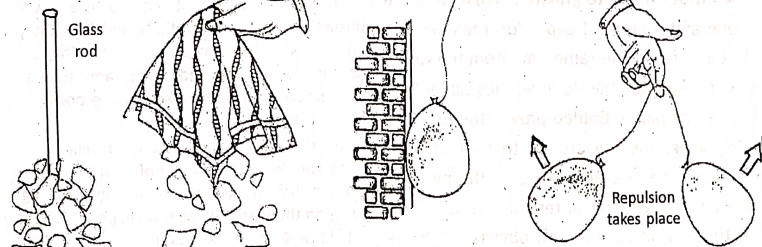
To explain the reason of such facts, English scientist William Gilbert stated that the materials get charged on rubbing. That’s why the materials attract the bits of paper. It is not an isolated property of any matter, but much general property and holds good for glass, silk, fur (cat skin), ebonite, sealing wax, cotton, resin, amber, etc.
Thus, an attractive property (or attractive force) is developed in the materials due to rubbing. The force is called the electrostatic force. In the above examples, the comb, glass rod, silk cloth, and balloon are said to be electrically charged on rubbing.
“Class 8 General Science Physical Environment notes, WBBSE syllabus”
Charging a material by cubbing it with another material is called charging by friction. And, the charges that remain bound to the surface of a material and do not flow are called static electricity.
Chapter 1 Physical Environment Different Types Of Electric Charges :
Highlights the fact that there seems to be possible the existence of two different types of electric charges. Benjamin Franklin referred to them as negative charge and positive charge.
Conventionally, we use Electric Series to determine which material will acquire which type of charge when rubbed with another material. In general, if anyone is rubbed with any other higher-up in the series, the first one gets negatively charged and if rubbed with a lower one in the series, then it gets positively charged.
Example :
If a glass rod is rubbed with silk cloth then the glass rod gets positively charged, while a glass rod rubbed with fur gets negatively charged. Similarly, when one ebonite rod is rubbed with fur, the ebonite rod gets negatively charged, while fur gets positively charged.
To show the existence of two types of electric charges :
If two glass rods being rubbed with silk cloth are brought near each other, it is observed that the glass rods repel each other Similarly, two ebonite rods being rubbed with fur also repel each other when they are brought near each other. But, when a glass rod being rubbed with a silk cloth and an ebonite rod being rubbed with fur are brought closer, it is observed that both the rods attract each other.
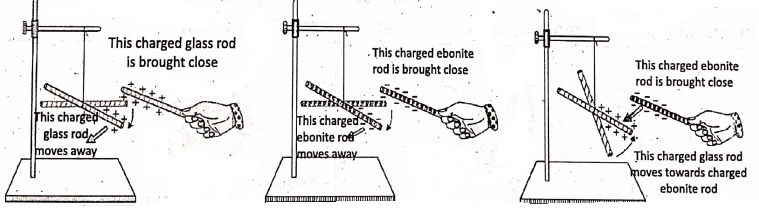
So, we can conclude :
- There are two types of electric charges called positive charge and negative charge.
- Like charges repel each other.
- Unlike charges attract each other.
Explanation of charging on rubbing by the simple orbital model of an atom :
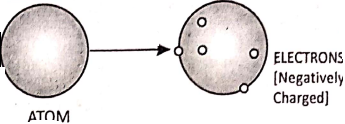
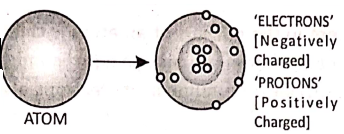
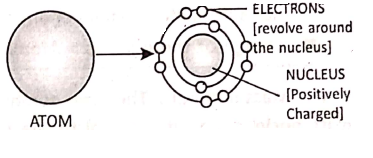
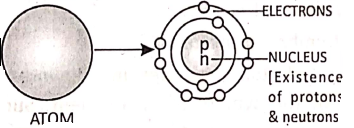
All matter is made up of very small entities called atoms. An atom consists of a central positively charged core called the nucleus, containing still smaller particles protons, and neutrons. A proton is a positively charged particle while a neutron has no charge. An atom on the whole is electrically neutral.
The electrons revolve around the nucleus in certain permitted orbits. An electron is a negatively charged particle. In the normal state, the total negative charge of the electrons in an atom is equal to total positive charge of the protons.
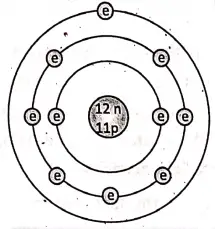
When two bodies are rubbed together then due to heat provided by friction, electrons are torn off from the body in which they are less tightly bound and transferred to the body .in which they are more tightly bound. Thus, the body which gains free electrons becomes negatively charged while the body which loses free electrons becomes positively charged.
For example, when a glass rod is rubbed with a silk cloth, some electrons are transferred from glass to silk as the electrons are less tightly bound in glass than in silk. Due to such a deficit of electrons, the glass rod becomes positively charged while due to the gain of an equal number of electrons, the silk cloth becomes negatively charged.
Similarly, on rubbing an ebonite rod with fur, the electrons from the fur are transferred to’ an ebonite rod as the electrons in fur are less tightly bound than in ebonite. Thus, the ebonite rod acquires a negative charge while fur acquires an equal but opposite amount of positive charge.
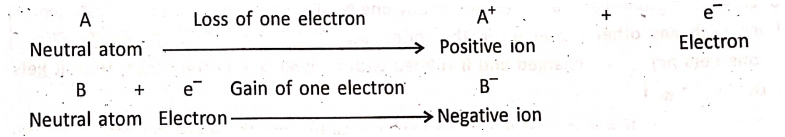
So, we can conclude :
- On rubbing, the body gains electrons. becomes negatively charged and the body which loses electrons becomes positively charged,
- Charges developed on two bodies are equal in magnitude but opposite in nature.
Chapter 1 Physical Environment Measurement of Electrostatic Force: Coulomb’s Law :
French physicist Charles Augustin de Coulomb derived a formula to calculate the (attractive, or repulsive) force acting between two point charges.
According to Coulomb’s law :
The magnitude of the force between two charged particles is directly proportional to the product of the magnitude of the charges and inversely proportional to the square of the distance between them.

If q1 and q2 are the magnitudes electrostatic of two charges and r is the distance between them, then the force is
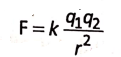
where k is the constant of proportionality, the value of which depends upon the nature of the medium separating two charges and the system of units in which various quantities in the above expression are expressed. Thus, the value of k is different for different media in different systems of units. For example, in C.G.S. system k = 1 for vacuum and in S.l. system k = 9 x 109 newtom meter2 coulomb-2 (symbol N.m2 .C-2)
It is evident from the above expression that
- If the magnitude of the charges are doubled and tripled respectively then the force becomes (2 x 3) or 6 times
- If the distance between the charges is halved, then the force becomes 1/(½)2 or 4 times, and.
- If the distance between the charges is halved, then the force becomes 1/(2)2 or 1/4 th of the previous force.
Chapter 1 Physical Environment Motion Due To Electrostatic Force :
The rotation of an electron around the nucleus of an atom requires a centripetal force to be acted on the electron towards the center of the circular orbital path.
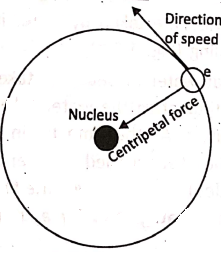
This force is supplied by the electrostatic force between the electron and the nucleus. The fact is similar to the planetary motion around the sun, where the required centripetal force js supplied by the gravitational force between the sun and the planet.
Chapter 1 Physical Environment Heat
The direction of flow of heat
When two bodies at different temperatures are placed near one another or kept in contact, heat flows from the body at a higher temperature to the body at a lower temperature. Even if different parts of a body differ in their temperatures, then also heat flows from its higher temperature portion to the lower temperature portion.
The flow of heat continues until the two bodies attain at the same temperature. When such a state is reached, the two bodies are said to be in a state of thermal equilibrium, and their common temperature is called neutral, temperature.
Chapter 1 Physical Environment Factors Affecting The Heat Energy Gained Or Lost By A Body
Measurement of heat :
The amount of heat gained or lost by a body is found to depend on three factors :
- Mass of the body,
- Change in its temperature (i.e. either increase or decrease in temperature), and
- Nature of material of the body.
Let us now perform the following activities one by one in order to study the effect of these three factors.
Chapter 1 Physical Environment Activity 1:
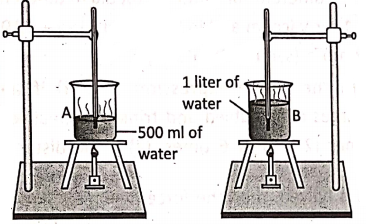
Let us take two identical beakers A and B. Pour 500 ml of water in beaker A and 1 liter of water into beaker B. Now, put the beakers on two identical separate stands and heat them by two identical bunsen burners. Let us note down the time taken by water in each beaker for boiling.
We will note that water in beaker B takes almost double the time than the water taken in beaker A for boiling. As the time taken is almost double, so the heat supplied in water in beaker B is almost double. This proves the fact that the amount of heat gained by a body depends on its mass.
Chapter 1 Physical Environment Activity 2:
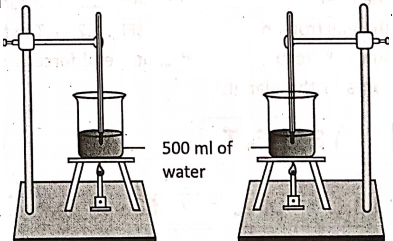
Let us take a beaker and pour 500 ml of water into it. Record the temperature of this water by a thermometer. Let it be 20°C. Now, put the beaker on a tripod stand and heat it by a bunsen burner.
Note the time taken to raise the temperature of the water up to a certain temperature, say 50°C [temperature rise being (50-20)°C = 30°C]. Again, note the time taken to raise the temperature of the same quantity of water to another temperature, say 80°C [temperature rise being (80° – 20)°C = 60°C).
“WBBSE Class 8 Science Chapter 1, Physical Environment important questions”
We will note that the time taken in the second case is almost double that in the first case. This proves the fact that the amount of heat gained by a body depends upon the rise in its temperature.
Chapter 1 Physical Environment Activity 3:
Let us take two identical beakers A and B. Pour 500 ml of water in beaker A and 500 ml of mustard oil in beaker B. Put a thermometer in each beaker. Let the initial temperature of water and mustard oil is 20°C.
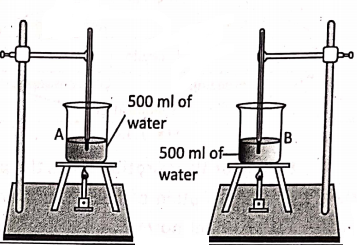
Now, heat both beakers by two separate identical bunsen burners simultaneously for the same duration of time. We will notice that the rise in temperature in the two thermometers is different. This proves the fact that the amount of heat gained by a body depends upon the nature of its material.
If Q be the amount of heat gained by a body of mass m to rise its temperature to t° then according to the above activities we can write: Q ∝ m and Q ∝ t
Combining these two, we can write Q mt or, Q = mst. where s is the constant of proportionality and it is called the specific heat capacity of the material of the body.
- Thus, heat gained by a body = mass x specific heat capacity x rise in its temperature. Similarly, if the temperature of a body falls.
- Then the heat lost by the body = mass x specific heat capacity x fall in its temperature.
Units of heat :
In C.G.S; system, the amount of heat is measured in the unit of calories (cal). One calorie is the amount of heat required to raise the temperature of 1 g of pure water by 1°C (actually from 14-5°C to 15-5°C).
A calorie is a small unit, so a bigger unit kilocalorie (kcal) is often used. 1 kcal = 1000 cal. Since heat is a form of energy, so the unit of heat is the same as that of energy. The S.l. unit of heat is the joule (abbreviated as J). The units calorie and joule are related as 1 cal = 4.186 J (or nearly 4.2 J)
Change of state
A material can exist in any of the three states namely solid, liquid, and gas under different conditions of temperature and pressure. The process of change of a substance from one state to another at a constant temperature is called its change of state. shows a complete cycle of change of state.
Melting or Fusion :
It is the process during which a solid change to its liquid state by the absorption of heat. The fixed temperature at which a solid melt to its liquid state under normal atmospheric pressure is called the melting point of the solid.
For example, ice melts into the water at a temperature of 0°C under normal pressure.
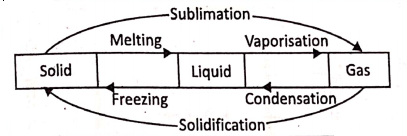
Boiling or Vaporisation :
It is the process during which a liquid changes to its gaseous state by the absorption of heat. The fixed temperature at which a liquid changes into its gaseous state under normal atmospheric pressure is called the boiling point of the liquid.
For Example, water boils or vaporizes to steam at a temperature of 100°C under normal pressure.
Condensation :
It is the process during which the vapor of a substance changes to its liquid state by releasing heat. The fixed temperature at which the vapour of a substance changes to its liquid state under normal atmospheric pressure is called the condensation point of the vapor.
For Example, the steam condenses to water at 100°C under normal pressure.
Freezing :
It is the process during which a liquid changes into its solid state by releasing heat. The fixed temperature at which freezing occurs under normal atmospheric pressure is called the freezing point.
For Example, water freezes into ice at 0°C under normal pressure.
Sublimation :
It is the process in which a solid directly changes into its gaseous state at all temperatures without being liquified.
For Example camphor, naphthalene, iodine, ammonium chloride, etc. directly perform the act of sublimating from solid state to gaseous state.
Solidification :
It is the process in which a substance directly changes from a gaseous state to its solid state.
For Example, carbon dioxide gas solidifies into dry ice at a very low temperature.
Chapter 1 Physical Environment Change Of Volume During Melting And Freezing
In general, the solids increase in volume during melting and the liquids decrease in volume during freezing. But there are some exceptions. There are some solids like ice, cast iron, antimony, bismuth, etc. which contract during melting and expand during freezing.
For example, 11 cm3 of water solidifies to form 12 cm3 of ice.
Examples :
- Water pipelines, rocks, etc. are found to burst during very cold winter nights. This is due to the fact that water expands on freezing, such that it exerts a larger amount of pressure on the pipes, rocks, etc. So, they burst.
- In cold countries, when the atmospheric temperature falls below 0°C, the water at the surface of a pond contracts into ice. Now, ice being lighter than water, ice floats on the upper surface of the water. As a result, the aqua life of fish and other animals is possible in the water of a pond, although the water at the surface has been frozen into ice.
- Iron, type metal, etc. are used for casting press letters. Because these metals expand on cooling and form sharp casting.
Chapter 1 Physical Environment Factors Affecting Melting Point
Two factors affect the melting point of a solid.
The factors are :
- Pressure and
- Impurities.
Effect of pressure on melting point :
Pressure plays an important role as far as the process of melting is concerned.
- For substances which contract on cooling (like wax), the melting point increases with the increase in pressure.
- For substances which expand on cooling (like ice), the melting point decreases with the increase in pressure.
Regelation :
If two pieces of ice are pressed together tightly for a few seconds, and then released,.they are found to join to form a single piece. When pressure is applied, the ice at the interface of two pieces melts at a temperature below 0°C and forms water.
As the pressure is released, the melting point is raised to 0°C. So that the water re-freezes thereby joining the two pieces. The fact.is known as regelation.
Definition :
The process of melting of ice by applying pressure and re-freezing of molten ice by releasing pressure is called regelation.
Effect of impurities on melting point :
In general, impurities lower the melting point of a solid substance. If common salt is mixed with ice (in ration 1: 3), the temperature falls below its melting point (0°C) to about -23°C.
Also, a mixture of anhydrous CaCI2 and ice (in the ratio 3 : 2) produces a temperature of about -50°C. Such a mixture which can produce a lower temperature is called a freezing mixture.
Uses of freezing mixture :
It is usually used
- For making ice creams and kulphies,
- To retain the freshness of fish, meat, etc. from being damaged with contact in air, and
- In several practical purposes to maintain lower temperatures in the laboratories.
Chapter 1 Physical Environment Concept Of Latent Heat
It is found from experiments that the temperature of a substance does not rise or fall, as long as the change of state takes place, although heat is supplied or removed in the process. Where does the heat go away?
In fact, heat is utilized to convert changes of the physical state of a substance. It appears to be ‘hidden’ or ‘concealed’ in the substance. The heat is known as ‘hidden heat’ or ‘latent heat’.
Chapter 1 Physical Environment Activity 1 :
Let us take some ice cubes in a beaker and put a thermometer into it so that its bulb is completely covered with ice. The thermometer reads CTC. Now, heat the beaker gently over a flame. We will notice that on heating, ice starts melting into water, but the thermometer reading remains at 0°C till whole of the ice melts.

On further heating, the temperature of water starts rising till it attains a temperature of 100°C. We will notice that water starts boiling at 100°C and the thermometer reading remains the same till whole of the water changes into steam.
Definition :
The amount of heat absorbed or released by a unit mass of a substance to change its state without any change in temperature is called the latent heat of the substance of that particular change of state.
It is usually denoted by T. So, by definition, if Q. be the amount of heat absorbed or released by a substance of mass m, then
Units :
The S.l. The unit of latent heat is joule per kg or J. kg-1 and the C.G.S. unit is calorie per gram or cal. g-1 .
Different kinds of latent heat:
Experimentally it is found that the melting point of ice and the freezing point of water are the same (0°C). So, the values of latent heat of fusion (melting) of ice and freezing of water are the same.
The value is 80 cal. g-1 . This means that the amount of heat required to change 1g of ice at 0°C into lg of water, at the same temperature is 80 cal. OR, the amount of heat released by 1g of water at 0°C to be changed into 1g of ice at the same temperature is 80 cal.
Likewise, the latent heat of the vaporization of water and that of condensation of water vapour are the same and it is 537 cal. g”1. This means that the amount of heat required to change 1g of water at 100°C into 1g of water vapour at the same temperature is 537 cal. OR, the amount of heat released by 1g of water vapour at 100°C to be condensed into 1g of water at 100°C is 537 cal.
Ice at 0°C is colder than water at 0°C. This is so because that there is a lack of heat of amount 80 calorie per gram in ice at 0°C than in water at 0°C.
Water at 100°C is less harmful than steam at 100°C. Because there is an excess of the heat of amount 537 calories per gram in steam at 100°C than in water at 100°C.
Suppose in a big ice block, there is a hole which is filled with water at 10°C. What will happen ?—Initially, the water at 10°C gives out heat as it is in contact with the ice block.
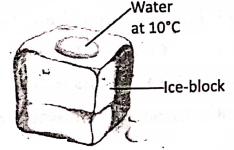
At a particular stage, both the ice block and the water inside the hole acquire the same temperature (0°C). Thereby, the exchange of heat stops, and the water can no longer give out 80 calories of heat per gram to be converted into ice.
Vaporization
There are two ways through which a liquid can be changed into its vapour state, namely,
- Evaporation and
- Boiling.
1. Evaporation :
If we take a drop of ether on our palm, we notice that ether disappears immediately and the palm feels cold. This happens due to the evaporation of ether.
Evaporation takes place at the free surface of a liquid, at all temperatures (below the boiling point). It is a slow process but the rate of evaporation increases with an increase in temperature and also, with an increase in the surface area of the liquid.
For Example wet clothes dry rapidly when spread in the sunshine.
Definition :
The (slow) process of conversion of a liquid into its vapor at all temperatures (below its boiling point) from its free surface is called evaporation.
Evaporation causes cooling :
When a liquid evaporates, it requires the latent heat of vaporization. If the heat is not supplied from outside, then the liquid takes the required heat. from its own body and its surroundings causing a fall of temperature.
A few examples of this are :
1. In summer, water gets coolecUivan earthen pitcher:
The pitcher being made of clay has a large number of pores through which water comes out and evaporates. The required latent heat for evaporation is taken from the water and the pitcher, causing a fall of temperature. But the water kept in a metal pot is not cooled, as it has no pores.
2. Wet cloth is put on the forehead of a fevered person:
The water from the wet cloth evaporates taking the latent heat of vaporization from the body of the patient. So, the body temperature of the patient is lowered.
3. Dogs hang out their tongues in summer:
They do not perspire through their skin. The saliva, as well as, sweat from their tongues evaporates taking latent heat from their body. This gives a comfortable sensation of cooling to the dogs.
2. Boiling :
proves the constancy of the temperature during boiling. It takes place at a fixed temperature which is the boiling point of the liquid> over its entire surface area. Boiling is a rapid and violent process.
Definition :
The (rapid) process of conversion of a liquid into its vapour at a fixed temperature over its entire surface area is called boiling.
The distinction between evaporation and boiling :
| Evaporation |
Boiling |
| (1) Takes place at all temperatures. |
(1) Takes place at a fixed temperature. |
| (2) Takes place only from the exposed surface area of the liquid. |
(2) Takes place from all parts of the liquid. |
| (3) Slow and silent process. |
(3) Rapid and violent process. |
| (4) Evaporation causes cooling. |
(4) Boiling causes heating. |
Chapter 1 Physical Environment Factors Affecting Boiling Point :
1. Effect of pressure :
Water boils at a temperature higher than 100°C if the atmospheric pressure is more than the normal pressure (76 cm of Hg) and vice versa. Thus, the boiling point of a liquid increases with the increase in pressure. Cooking in a pressure cooker is such an application.
A pressure cooker consists of an aluminum container, to which a lid is fitted. The lid is made air-tight with a rubber pad. At the center of the lid, there is a heavy safety pin valve. A handle of non-conducting material is attached to the cooker.

As long as the wafer boils inside the cooker, the steam so-produced cannot escape, so that the pressure inside the cooker increases more and more. Due to it, the boiling point of water increases from 100°C to about 120°C. This makes cooking much easier and quicker.
2. Effect of impurities:
It is seen that when a little amount of common salt is added with water, the water boils at a temperature of about 109°C. Thus, the boiling point of a liquid increase by the presence of impurities in it.
Condensation
There are a few natural examples of condensation of water vapour present in the atmosphere such as dew, fog, cloud, etc.
Formation of dew :
At night, the earth radiates heat to be cool down. So, the air in contact with the surface of the earth cools down very rapidly by radiating heat. In this way, the temperature of the air comes down gradually.
Especially in winter, the temperature of air quickly reaches the dew point. With the further lowering of temperature, some amount of water vapor present in the atmosphere condenses into small droplets and deposits as dew on thin, exposed objects like grass, and leaves near the ground mainly in the morning or evening.
“Physical Environment WBBSE Class 8 notes, General Science Chapter 1”

Formation of fog:
If the temperature over a wide region of the atmosphere near the surface of the earth comes closer to the dew-point or sometimes falls below the dew-point, the water vapor present in the atmosphere condenses into small droplets of water which float in air on the dust particles, smoke particles, coal dust, etc.
This is known as fog or mist. Fog is very often seen in industrial areas where there are plenty of pollutant particles on which fog can deposit. Fot reduces the range of normal visibility. It disappears at noon.
Formation of the cloud :
Air containing water vapor becomes lighter, and goes high up, where due to very low pressure, the air expands in volume. This makes the air to be cooling. Due to such successive coolings, at one stage, the temperature of air falls below the dew point.
Then, the moisture present in the air are condensed into small droplets on the suspended dust particles. The layer of such droplets is known as a cloud.
Chapter 1 Physical Environment Transfer of heat
So far we have studied that heat can flow from higher temperatures to lower temperatures of different bodies or different parts of the same body. This transfer of heat occurs through three different distinct processes called
- Conduction.
- Convection and
- Radiation.
If we place one end of a metal spoon in the fire holding its other end by hand, within a very short time, it is seen that the other end becomes too hot to be held by hand. This shows the conduction of heat in metals.
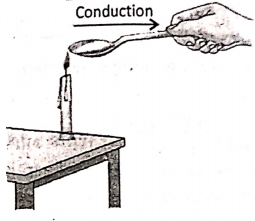
Illustrates heating by conduction, convection, and radiation. In the hand is placed in three different positions near the fire.
Position-1:
One end of a metal rod is placed in the fire. After a very short time, the other end of the rod will become so hot by conduction that it would be held by a gloved hand (shown at the upper right).
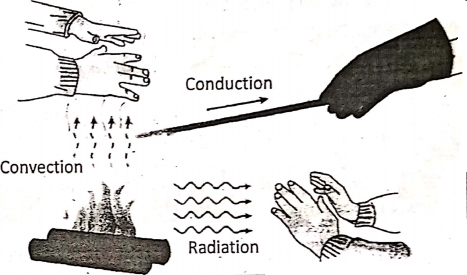
Position-2 :
The hands at the left (above the fire) are becoming hot by the convection of rising hot air.
Position-3 :
The hands at the lower right feel hot by radiation. The process of conduction is prominent in solids, while the process of convection occurs in liquids and gases, and radiation occurs with no need of substance to be present in the space between the bodies. Perhaps, radiation is common in all three states of matter. Now, let us discuss the processes one by one in more detail.
Chapter 1 Physical Environment Conduction
Definition :
Conduction is the process of transfer of heat from the hotter end to the colder end of a solid without any actual movement of the particles of the medium.
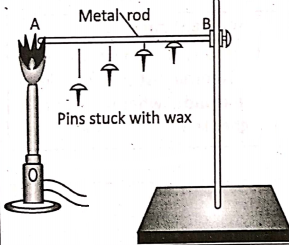
Activity :
Take an iron rod or a knitting needle AB. Fix four board pins in a row along its length using a few drops of candle wax . Clamp the end B of the rod on a vertical stand and heat the end A. We will observe that after some time, the board pins start falling down one by one starting from the end A. This implies the fact that heat travels along the length of the rod from its hotter end to the colder end.
Good and bad conductors of heat:
Materials which conduct heat readily are called good conductors of heat. Most metals like silver, copper, aluminium, iron, nickel, etc. and their alloys like brass, bronze and stainless steel are good conductors of heat. However, different metals conduct heat differently.
Materials that do not conduct heat readily are called insulators or bad conductors of heat. Glass, wood, paper, plastic, wool, porcelain, stone, water and air are bad conductors of heat. All liquids (except mercury) and gases are insulators of heat.
Activity : (Ingen Hauz’s experiment) :
Take a metal trough with five holes drilled in one of its sides near the lower edge. Take five rods of different metals, but exactly of the same size and shape.
Insert them into the holes such that an equal portion of each rod remains outside. The outside portion of each rod is coated with a uniform layer of candle wax. Now, fill the trough with boiling hot water. Wait for 5-10 minutes.
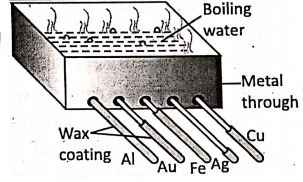
It is observed that the wax has melted up to different lengths in different rods. It shows that different metals conduct heat differently. Out of the metals, silver, and copper are the best conductors of heat.
Chapter 1 Physical Environment Activity :
Wrap a strip of wire gauze around a piece of ice cube and drop it in a test tube that is filled with water. Heat the surface of the water over a burner. Continue heating till the water at the surface starts boiling.
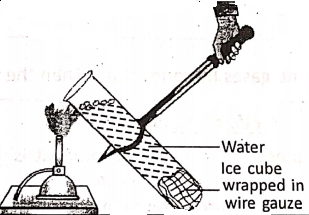
It is observed that the water at the surface starts boiling, but the ice cube at the bottom of the test tube does not melt. This proves that water is a bad conductor of heat, otherwise ice would melt.
Uses off good and bad conductors of heat:
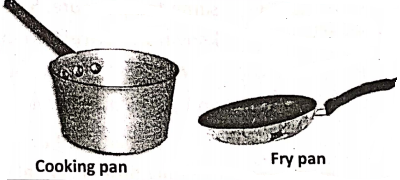
- Cooking utensils are made up mostly of metals and some alloys like copper, aluminum, brass, and stainless steel as they are good conductors of heat. Heat quickly reaches the food for easily being cooked. But, their handles are made of insulators like wood, plastic, and bakelite. Being insulators, they do not become hot with the utensils and we can ‘ easily hold the handles with our hands. Similarly, we can use hand gloves! or any other insulator to hold a hot dish out of an oven.
2. Cooling coils of air conditioners and refrigerators are made of copper. Because copper is a better conductor of heat than most other metals.
3. Slabs of ice are usually covered with a thermally insulating material like sawdust. This prevents outside heat from melting the ice.
4. Mercury is a good conductor of heat. So, mercury is used as a thermometric liquid and it acquires the temperature of the object very quickly, without making an abrupt change in the temperature of the object.
5. Woolen clothes have fine pores filled with air. Both the wool and air are bad conductors of heat. In winter, they do not allow the heat of the body to flow outwards and keep our body warm.
6. A newly made quilt is warmer than an old one. Because a more amount of air is trapped in a newly made quilt compared to that in an old one. When we use quilt in winter, the air trapped in the quilt does not allow the heat to flow outwards from our body.
7. Eskimos make their houses (called igloos) out of snow. Because the large amount of air trapped in snow acts as an insulator. So that the heat within the igloo does not flow out and thus, it. keeps the inside warm.

Chapter 1 Physical Environment Convection
We’ve learned that heat is not transferred through liquids and gases by conduction. Then the question arises, how the heat is transmitted through them ?—Actually, when a liquid or a gas is heated, the molecules near the source of heat, absorb heat and expand, ultimately becoming lighter and rising up while the colder molecules move downward to take their place.
“WBBSE Class 8 Chapter 1 General Science, Physical Environment solutions”
Now, these molecules also get heated and rise up. This process (convection) continues till the entire liquid or gas acquires the same temperature. Such a circulatory movement set up in the medium is known as a convection current.
Definition :
Convection is the process of transfer of heat in liquids and gases due to the actual movement of the medium particles.
Chapter 1 Physical Environment Activity 1:
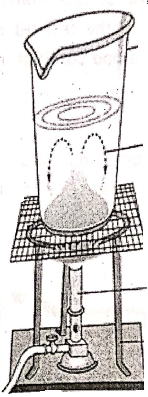
Fill a glass beaker three-fourths with water and carefully drop a crystal of potassium permanganate into the beaker. Heat the beaker using a bunsen burner. It is seen that the violet color of potassium permanganate starts moving upwards. This is actually the convection currents of water molecules.
Chapter 1 Physical Environment Activity 2:
Take a rectangular hollow glass box with two glass chimneys A and B on its topside as shown in. Light a candle and place it under chimney A and hold a smoldering agarbatti (incense stick) just above the mouth of chimney B.
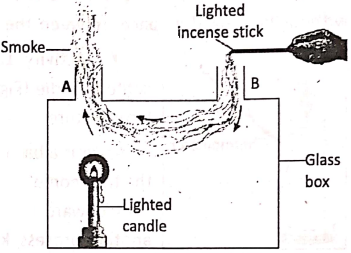
Now, watch the direction of flow of smoke.’ It is seen that the smoke entering through the chimney B, comes out of the chimney A . It shows that convection currents are set up in the air inside the hollow box. In factories the smoke and hot gases rise up due to convection currents and escape out through the chimneys.
Chapter 1 Physical Environment Application of convection :
1. Sea breeze and Land breeze :
During the daytime, the land gets heated more rapidly than the seawater. So, the air above the land becomes warmer and lighter, hence rises upwards. In turn, cooler air above the seawater blows in to fill up the air towards the land that rises up, thereby giving rise to sea breeze.

After sunset, the land cools down faster than the seawater. So, the air above the seawater remains warmer than the air above the land and rises upwards. This creates a current of air to blow from land towards the sea, giving rise to the land breeze.
2. Ventilation :
The air, we breathe out, is warmer and lighter than ordinary air. It passes out through the ventilators (openings near the roof) provided in our houses. In turn, fresh air being heavier enters the house through the doors and windows.
Chapter 1 Notes For Class 8 Physical Environment Radiation
Radiation is the process of transfer of heat from a hot body to a cold body directly, without heating the space between the bodies.
Chapter 1 Physical Environment Activity 1:
Hold a thermometer below the flame of a lighted candle. The thermometer will show an increase in temperature. As air is a bad conductor of heat, so heat could neither have reached the thermometer by conduction nor by convection as convection current rises upward, rather than sideways.
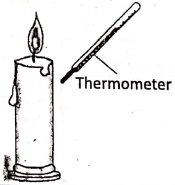
Therefore, the bulb gets heat by another process, known as radiation. Furthermore, if a thin cardboard or any other insulator is placed between the flame and the thermometer, it no longer becomes hot. This proves that air does not get heated by radiation.
All hot bodies—solids, liquids, and gases emit heat radiation, called radiant heat. For example, we feel hot in front of a fire, electric heater, electric iron or electric bulb, or from any other hot body.
Radiation requires no medium. For example, the heat of the sun reaches us by radiation without heating the space (the vast space between the sun and the earth is a vacuum).
Absorption and reflection of radiant heat by a body :
When radiant heat is incident on an object, a part of it is reflected and the remaining part is absorbed by the object. The amount of heat radiated by an object depends on its temperature, colour, and nature (dull or shiny). It has been proved that
- Black bodies are good absorbers, as well as, good radiators (i.e., bad reflectors) of heat and
- Shining bodies are poor absorbers and poor radiators, and good reflectors of heat.
Chapter 1 Notes For Class 8 Physical Environment Activity :
Take two identical cans having small holes in their lids. Paste black paper over one and keep the other remain as it is. Pass two similar thermometers through the holes into the cans. Place the cans in the sun for about an hour.
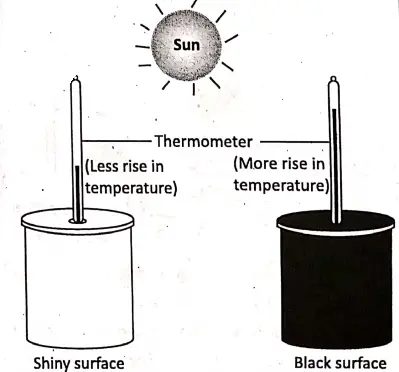
After some time we will notice that the thermometer in the can with black surface shows more rise in temperature than the other. This proves that a black surface is a good absorber of heat than the white surface.
Now, take the cans in a dark room. We will notice that the temperature drop in the thermometer of the can with black surface becomes more than the other. It follows that a black surface is a good radiator while a shiny surface is a poor radiator.
Applications of radiation :
- We wear white and light-colored clothes in summer because they absorb the minimum amount of heat from the surroundings and keep us cool. However, we wear black and dark-colored clothes in winter because they absorb the maximum amount of heat from the surroundings, and keep us warm.
- The outside bottom of cooking utensils is painted black while the other outer sides are kept sparkling bright. This is done so that the bottom portion can absorb the maximum amount of heat from the source of heat and gets heated quickly. But, to keep the food contents warmer for a longer time, the minimum amount of heat is radiated from the polished outer sides.
- A teapot is brightly polished so that a little amount of heat can be radiated from hot tea and it remains warm for a longer time.
- The radiators of automobiles and air conditioners are often painted black to radiate away the heat easily.
Chapter 1 Notes For Class 8 Physical Environment Thermos (Or Vacuum) Flask
A thermos flask, also called Dewar flask after its inventor Sir James Dewar, is a special type of insulated flask that can store liquids with very little changes in temperatures over a long time. That is, a thermos flask keeps a hot liquid hot and a cold liquid cold for few hours.
The flask consists of a double-walled glass vessel with both the inner and outer surfaces of the walls being silvered, like a mirror. The space between the walls is completely evacuated and the opening through which air is evacuated is sealed. The mouth of the flask is closed with a stopper (cork or plastic).
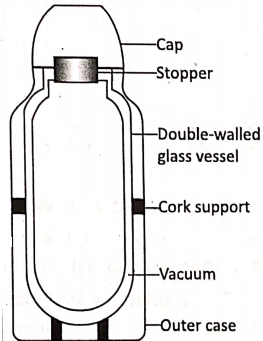
The double-walled glass vessel is fitted securely inside a plastic or metal case and is supported on corks at the sides and at the bottom. The thermos flask stops the transfer of heat by conduction, convection, and radiation so that a liquid in the thermos flask retains its temperature for a long time.
- As glass and cork are poor conductors of heat, so heat loss by conduction is minimized.
- The vacuum in between the walls prevents heat loss by conduction and convection (as conduction and convection require a medium).
- The silvered walls reflect all radiations. This prevents the heat loss by radiation.
The distinction between Conduction, Convection, and Radiation
| Conduction |
Convection |
Radiation |
| (1) Conduction requires a medium for heat transfer, without actual movement of the medium particles. |
(1) Convection requires a medium for heat transfer and it occurs through the actual movement of the medium particles. |
(1) Radiation does not require a medium. |
| (2) It mainly occurs in metallic solids. |
(2) It mainly occurs in liquids and gases. |
(2) It occurs without heating the medium. |
| (3) It is a slow process. |
(3) It is faster than conduction. |
(3) It is a fast process. It happens at the speed of light. |
| (4) Transfer of heat may take place in any direction. |
(4) Heat is transferred mainly in a vertically upward direction only. ‘ |
(4) Heat travels in straight lines. |
Chapter 1 Notes For Class 8 Physical Environment Light
Reflection
When, a beam of light traveling through a medium (like air) strikes a surface (a wall, a floor, a mirror or anything around us), some of the incident light bounces back or travels back into the first medium. We say this the reflection of light. It can be compared with the bouncing of a ball from the ground.

It is a matter of fact that all surfaces reflect light. That is how we see the world around us. A plane mirror is an ideal reflector and is used in the study of reflection in plane surfaces.
Definition :
When a ray of light traveling in a medium strikes a surface leading into another medium, a part of the incident ray bounces back into the first medium. This bouncing back of light in the same medium is termed as a reflection of light.
Regular and irregular reflection :
Usually, a reflection of light is of two kinds depending on the nature of the reflecting surfaces-
- Regular reflection and
- Irregular or diffuse reflection.
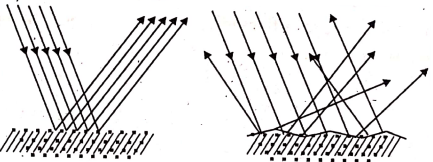
- When a beam of light falls rays become parallel to each other. This type of reflection is known as regular reflection.
- When a beam of light falls on’ a rough, uneven or irregular surface, the reflected rays of light are scattered in all directions. This type of reflection is known as irregular reflection.
Terms related to reflection :
In, MM’ is a plane reflecting surface (like a plane mirror), AO the incident ray, OB the on a smooth and very well polished surface, the reflected ray, 0 the point of incidence of the incident ray AO, ON the normal drawn on the surface MM’ at o, ∠AON = i = angle of incidence and ∠Bon = r = angle of reflection.
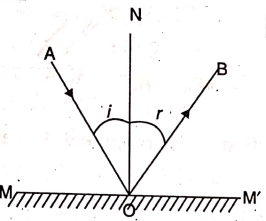
laws of reflection of light are as follows :
- The incident ray, the reflected ray, and the normal drawn at the point of incidence all lie in the same plane.
- The angle of incidence (i) is equal to the angle of reflection (r).
Formation of image By a plane mirror :
In our daily experience/ when looking through a plane mirror, we see our own image appear to form behind the mirror. But, light rays cannot pass through the mirror and go beyond it.
How then the image can be formed? :
Actually, light from our body is reflected by the mirror and enters our eyes such the reflected rays appear to come from an image behind the mirror. However, if a screen is placed at the position from where the reflected rays appear to be coming, we will not be able to see an image. Such an image that cannot be formed on a screen is called a virtual image.
“WBBSE General Science Class 8 Physical Environment, Chapter 1 explanations”
The position of the image formed by a plane or mirror can be located by making a ray diagram, as shown in.
When the object is very small, like a point:
Suppose 0 be the point object in front of a plane mirror M1 M2. Consider two rays OA and OB from the object that fall at A and B on the mirror. They will be reflected along AC and BD respectively, with the angle of incidence equal to the angle of reflection at each point. These rays do not converge at a point.
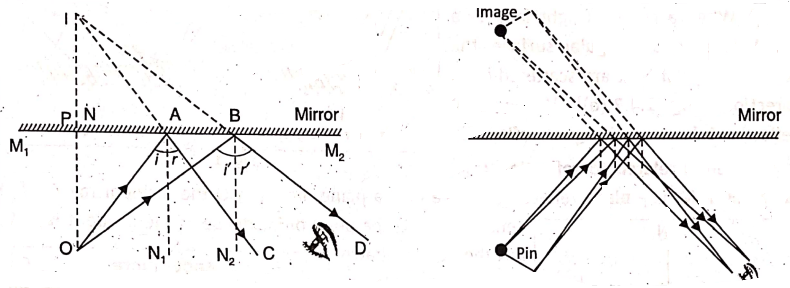
If we extend the reflected rays AC and BD backward, they meet at the point I behind the mirror, from which the reflected rays appear to be coming from. Therefore, I represents the virtual image of object O. On measuring the distances of the object 0 and its image I from the mirror, the distances are found to be equal. That.is, OP = IP.
Chapter 1 Notes For Class 8 Physical Environment The Image Formed By An Extended Object:
An extended object is a combination of several point objects. To draw the ray diagram, consider the rays emerging from the upper and lower ends of the object to fall on the mirror and the rays being reflected by the mirror appear to come from its virtual (extended) image.
Formation of real image :
Images formed by a camera or on the retina of the human eye are real images. Also, the pictures projected on the screen in the cinema hall are real images. We are able to see the rays of light coming from the projector and falling on the screen.
Thus, the rays of light often being reflected by a mirror, actually meet at a point to form an image, called a real image. Such as image can be formed on a screen.
Characteristics of an image formed by a plane mirror :
The image formed by a plane mirror is
- Virtual and erect (upright);
- Of the same size as that of the object;
- As far behind the mirror as is the object ‘ in front of the mirror; and
- Laterally inverted (if the object is not symmetrical).
What is a lateral inversion of the image ?—Standing in front of a plane mirror, we see our left hand appears right of the image and vice versa. Such interchange of the left and right sides in the (nonsymmetrical) object and its image is due to reflection. of light by a plane mirror is called lateral inversion of image.

Note that the lateral inversion of the image is not noticeable for the objects which are symmetrical like the alphabets A, H, I, M, V, W, X, Y; while lateral inversion is noticeable for the objects which are not symmetrical like the alphabets B, C, D, E, F, G, J, K, L, P7 Q, R, S, Z.
This is due to the fact the word Ambulance’ is printed laterally inverted in front of an ambulance—so that it may appear accurate in the rear-view mirror of the driver in front.
Formation of the image by a pair of plane mirrors :
When two plane mirrors are placed at an angle, reflecting surfaces facing each other, and an object is placed between them, multiple images are formed. This is due to the fact that multiple reflections occur at the mirrors such that the image formed in one mirror acts as an object for the other mirror. The number of images formed depends on the angle between the mirrors. Mathematically, it is proved that, if 0 be the angle between two mirrors, then the number of images formed is
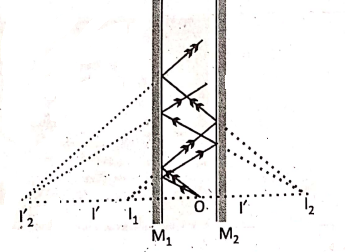
Case-1: When 0 = 0°, i.e., the two mirrors are placed parallel to each other, then an infinite number (theoretically) of images are formed.
0 is the position of an object. The rays from 0 produce an image l1 on the mirror M1 such that OM1 = l1 M1. Now, l1 acts as a virtual object for the mirror M2 and by further reflection at M2 it produces the image l2, such that l1 M2 = l2 M2.
“WBBSE Class 8 General Science Physical Environment notes, easy explanation”
The next image l2 is formed by M1, such that = M1l2 = M1 l2. In this way several successive reflections may occur in the two mirrors and the brightness of the remote images keeps on decreasing due to the loss of energy by successive reflections.
In actual practice (e.g., in. a showroom or in a barber’s shop) only the first few images can be seen clearly.
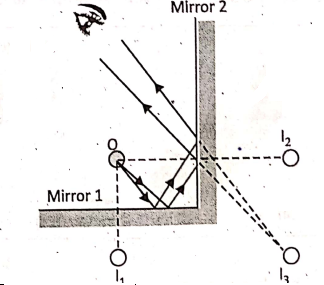
Case 2: When 0 = 90°, then n = 360°/90° = 4-1 = 3, i.e., when the two mirrors are placed perpendicular to each other, four images are formed.
0 is the position of an object. Each mirror forms an image. The images l1 and l2 are formed after reflection at the two mirrors 1 and 2 respectively. A third image is formed behind the line where the mirrors meet.
Chapter 1 Notes For Class 8 Physical Environment Applications of Plane Mirrors :
We find wide applications of plane mirrors in our daily life. Some of these are :
- As a looking mirror in the bedroom or washroom.
- In the showroom or optician’s room to increase the effective length of the room.
- In the barber’s shop for looking the hairs at the back of my head.
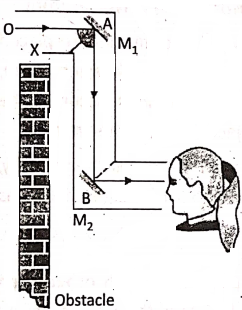
4. In a periscope, two parallel plane mirrors M1 and M2 are inclined at an angle of 45° with The vertical sides.of.a long tube at two opposite ends with two openings X and Y . A periscope enables us to see objects over an obstacle.
A ray of light OA coming from a distant object in front of the opening X falls on the mirror at an angle of incidence of ‘ 1 45° and is reflected vertically downward making an angle of reflection of 45°, and finally being reflected by mirror M2 reaches at the eye through the opening Y. The observer then sees the object above the obstacle.
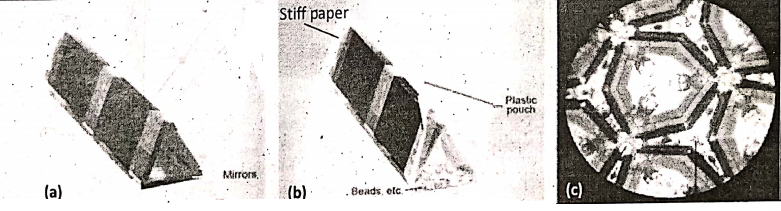
5. In a kaleidoscope, three strips of plane mirrors of equal size, inclined at an angle of 60° with each other, are used. A simple kaleidoscope is shown the three mirrors are taped with each other to form a triangular tube, Tape a small polythene pouch to two sides of the triangular tube and put some pieces of colored beads, and bangles into the pouch. Then, another end of the tube is covered with stiff paper and makes a hole in it. In this way, a kaleidoscope is formed. If we look through the hole and rotate the kaleidoscope, multicolored images are seen.
Chapter 1 Notes For Class 8 Physical Environment Refraction
Refraction is the phenomenon by means of which a ray of light deviates from its original path while passing obliquely from one transparent homogeneous optical medium to another transparent homogenous optical medium. It is essentially a surface phenomenon.
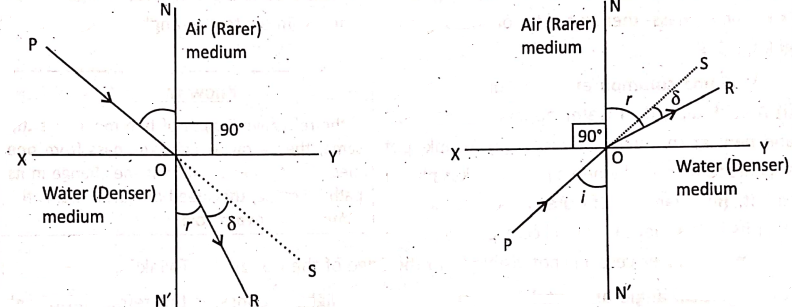
Light travels with different speeds in different media. The speed of light in a vacuum is 3 x 108 ms-1. Light travels faster in air (speed being 3 x 108ms-1) than in water (speed being 2.25 x 108 ms-1) or glass (speed being 2 x 108ms-1). Here, the air is the optically rarer* medium while water or glass are the optically denser** media.
When light travels
- From a rarer medium to a denser medium, it bends toward the normal.
- From a denser medium to a rarer medium it bends away from the normal.
PO is the incident ray; OR, the refracted ray; NON’, the normal drawn to the surface XY at the point of incidence 0; ∠PON, the angle of incidence (i); and ∠RON, the angle of refraction (r). The deviation of the refracted ray is, δ = (i – r), as i> r; and the deviation is, δ = (r- i), as i < r.
Chapter 1 Notes For Class 8 Physical Environment Laws of Refraction
Refraction of light at the boundary of separation of two media obeys the following two laws :
- The incident ray, the refracted ray and the normal drawn at the point of incidence on the boundary of separation of two media, all lie on the same plane.
- For a given pair of media, the ratio of the sine of the angle of incidence (i) to the sine of the angle of refraction (r) is constant.
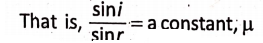
This constant (μ) is known as the refractive index of the second medium with respect to the • first medium. The second law is known as Snell’s law. For example, the refractive index of glass is 1.5. This means that—if a ray of light travels from air to glass, then the ratio of the angle of incidence in the air to the angle of refraction in glass is 1-5.
Some common examples of refraction :
- A stick dipped in water appears to be short and bent at the water’s surface,
- An ink spot appears to be raised when a glass block is placed over it.
- A tank full of water appears shallower than its actual depth,
- A coin placed at the bottom of a water vessel is not visible from the edge of the vessel,
- The twinkling of stars, etc.
Chapter 1 Notes For Class 8 Physical Environment Critical Angle And Total Internal Reflection Of Light
Consider the refraction of light from a denser medium (say, glass) to, a rarer medium (say, air). Starting with a small angle of incidence, the ray is bent away from the’ normal where i < r. As the angle of incidence in the denser medium increases, the angle of refraction in the rarer medium also increases. Finally, at a certain angle of incidence i(=θc ), the angle of refraction becomes equal to 90°. This angle ‘θc‘ is termed as the critical angle for the pair of media.
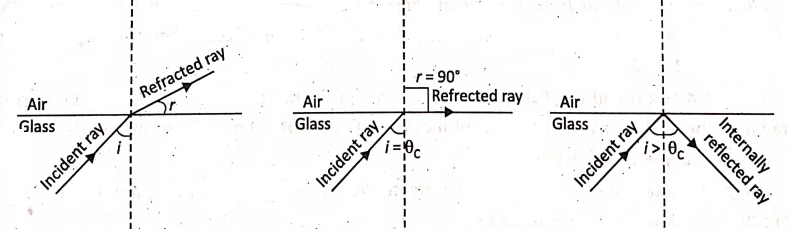
However, if the angle of incidence is increased beyond θc, the ray is not refracted at all, but it gets reflected internally into the denser medium. (Since it is not possible to get an angle of refraction more than 90°). This phenomenon is known as the total internal reflection of light.
Definition :
The critical angle is the angle of incidence in the denser medium for which the angle of refraction in the rarer medium is 90°, while a ray of light passes from a denser medium to a rarer medium.
When a ray of light traveling in a denser medium is incident at the surface of a rarer medium at an angle of incidence greater than the critical angle for the pair of media, the ray gets totally reflected back into the denser medium. The phenomenon due to which this happens is called total internal reflection of light.
Essential conditions for total internal reflection of light :
- The light must travel from a denser medium to a rarer medium.
- The angle of incidence in the denser medium must be greater than the critical angle for the given pair of media.
The distinction between ordinary reflection and total internal reflection :
- For ordinary reflection, light may incident from any medium to any other medium at any angle of incidence. But, for total internal reflection, light must travel from a denser medium to the interface of a rarer medium, at an angle of incidence greater than the critical angle for the given pair of media.
- In ordinary reflection, the incident light is partially reflected, while in total internal reflection, the entire amount of the incident light is reflected.
Chapter 1 Notes For Class 8 Physical Environment Some Facts Of Total Internal Reflection
1. Small beads of water on lotus leaves appear shiny. Because a layer of air sticks to the drops of water within the wax-like coating on lotus leaves.
When light rays passing through water strike the water-air interface at an angle more than the critical ‘ angle between the pair of media, concerned, the light rays suffer total internal reflection and finally enter in to our eyes.
2. In general, the critical angle of precious stones are small. For example, it is about 24-5° for diamonds. A piece 0f diamond is cut in such a fashion that the rays of light pass through the diamond at angles more than the critical angles, and suffer repeated total internal reflection through its different faces and finally emerge out to reach into our eyes. Thus, a piece of diamond appears shining.
3. A crack in a glass window pane appears silvery. This happens due to the occurrence of total internal reflection from the layer of air between the two glass surfaces.
4. Formation of mirage :
The mirage is a natural phenomenon, and it occurs due to the occurrence of total internal reflection of light at the surface separating two layers of air of different densities. In the desert, the sand becomes excessively hot during the daytime and heats the layer of air in contact with it, causing a lowering in density. But, layers of air in the upper side are comparatively cold, i.e., denser. Thus, successive layers of air going upwards are denser than the layers below them.
Light rays emerging from a distant tree are refracted away from the normal while coming from an optically denser medium to an optically rarer medium. This continues till a stage comes when the angle of incidence becomes more than the critical angle between the interface of two layers.
“Class 8 WBBSE Physical Environment Chapter 1, General Science revision”
Then total internal reflection takes place and the rays reach into the eye of a person such that the rays appear to come from an inverted image of the tree . Moreover, due to continuous changes of temperature and convection currents in the air, the image of the tree shimmers, and it adds to the illusion of the presence of water (virtual image). This is known as the mirage in the desert or inferior image.

In cold countries, the layers of air near the ground are colder and denser than the layers above it. So, the rays of light passing upwards from an object near the ground are totally reflected downwards when the angle of incidence becomes more than the critical angle between the interface of two layers of air and produces an inverted image hanging in the air. This is known as a mirage in cold countries or a superior image.
5. On hot sunny days, similar reflections occur on tarred roads.











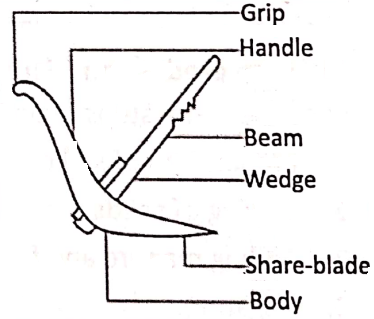


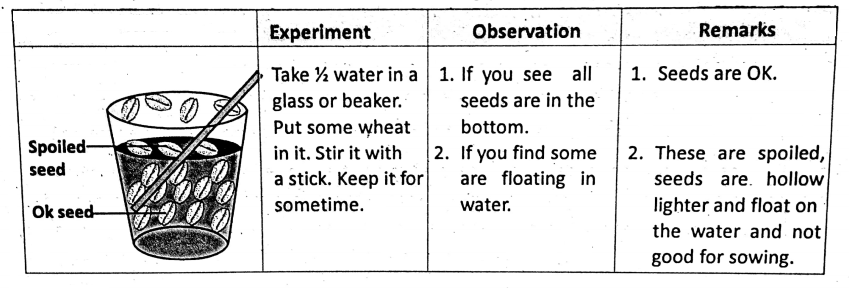








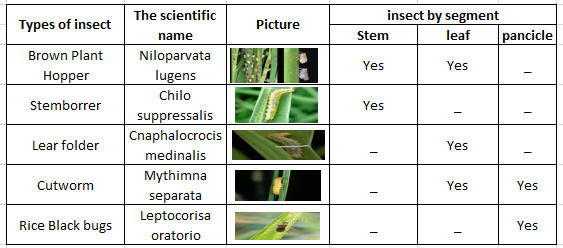






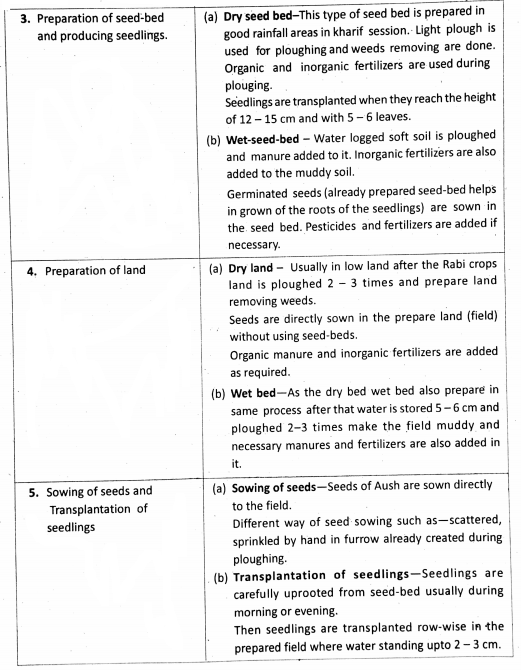




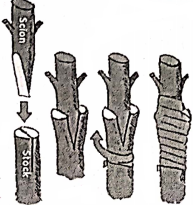






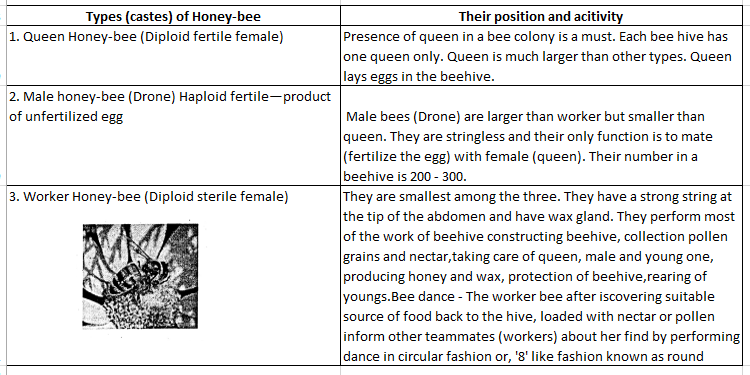
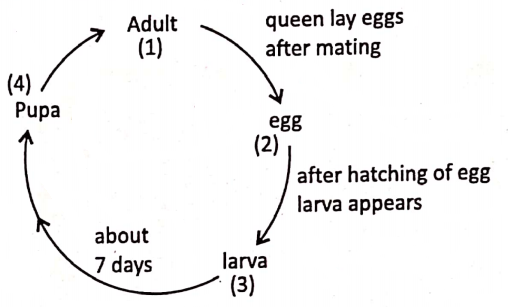





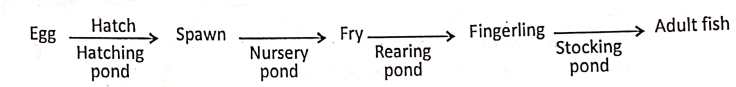



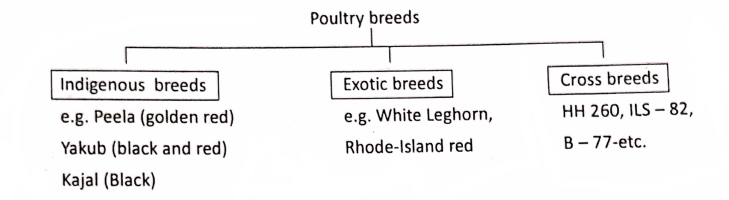
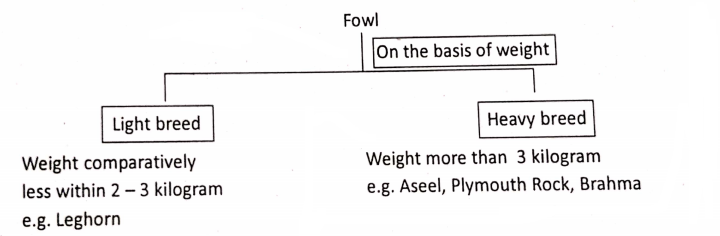















 Usage :
Usage :







 Triphala :
Triphala :



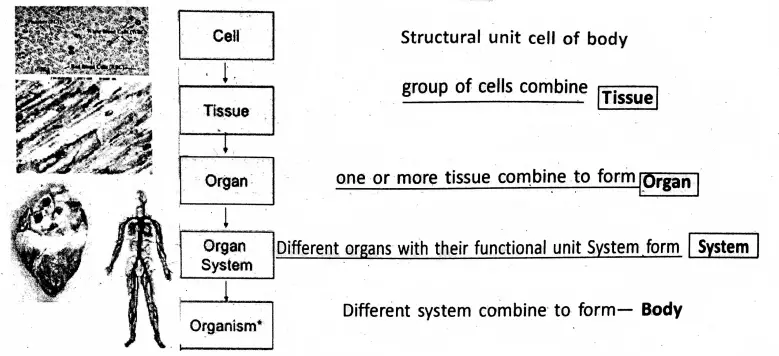
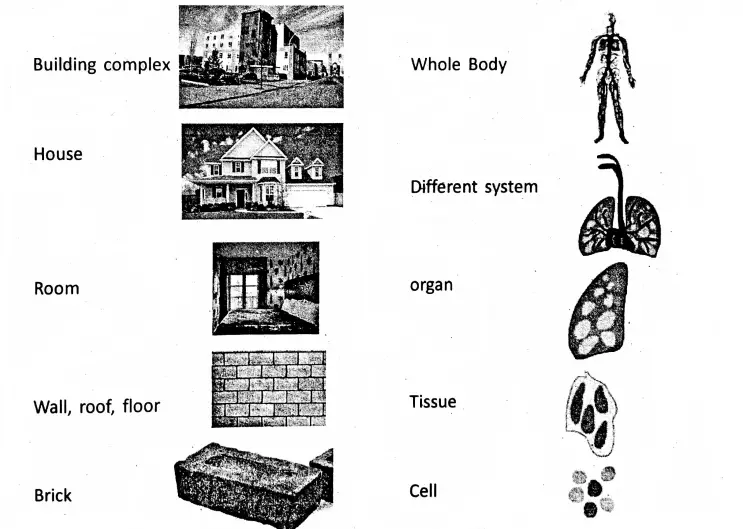




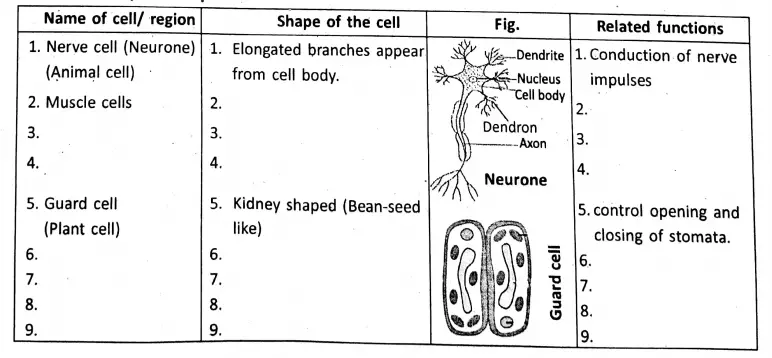
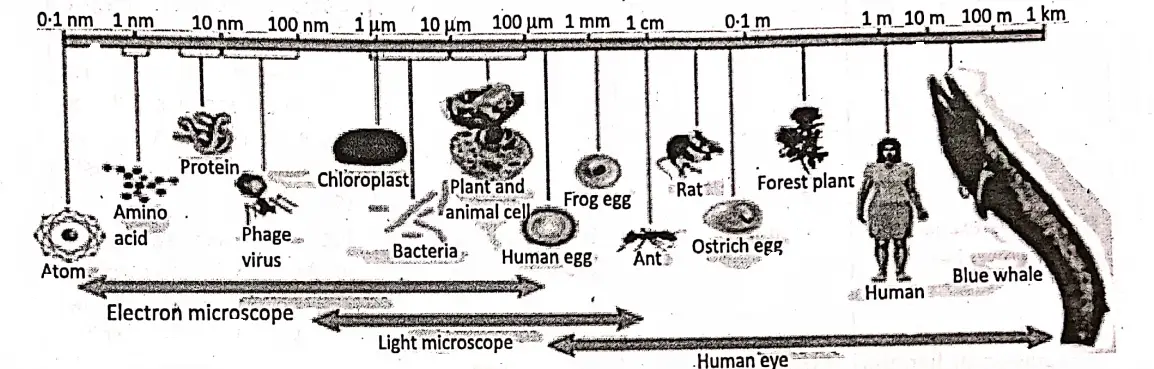
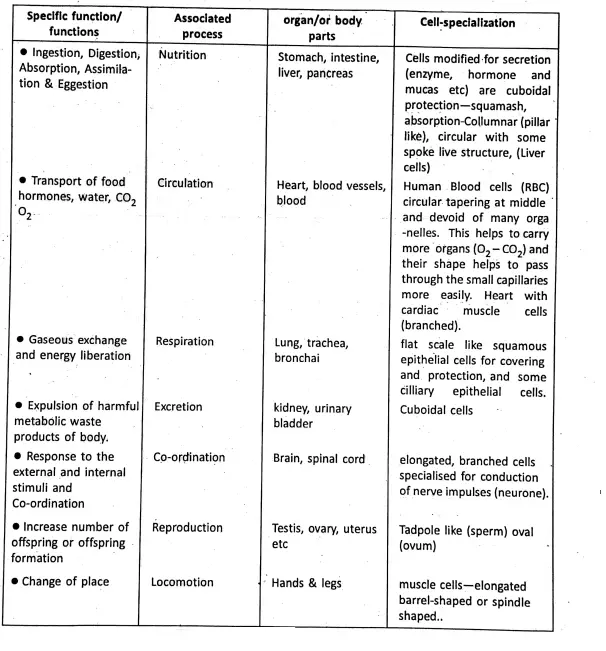 Typical cell range from 5-50 micrometres
Typical cell range from 5-50 micrometres
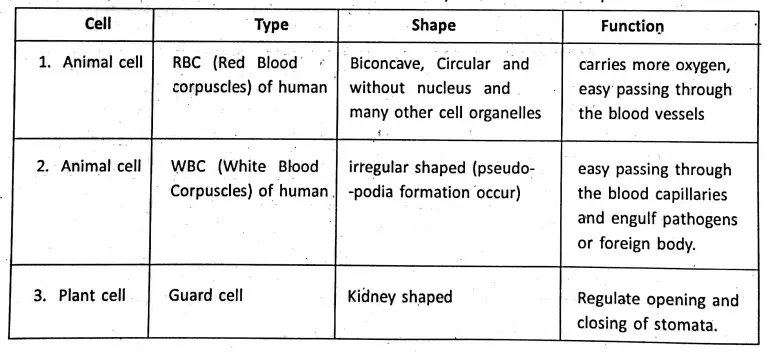 Now just think of your body which is consists of many organs such as the heart, liver, lung, ovary/ testis etc. which are the part of different systems such as circulatory, digestive, respiratory, reproductive etc.
Now just think of your body which is consists of many organs such as the heart, liver, lung, ovary/ testis etc. which are the part of different systems such as circulatory, digestive, respiratory, reproductive etc.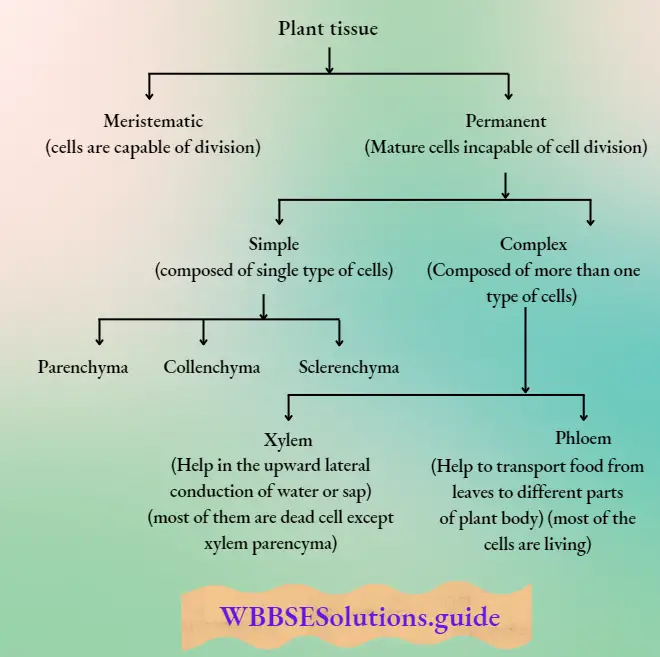

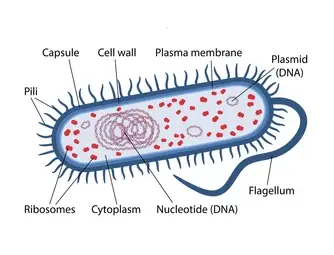 Function :
Function :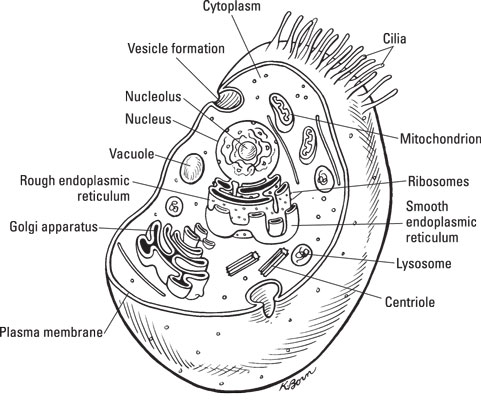
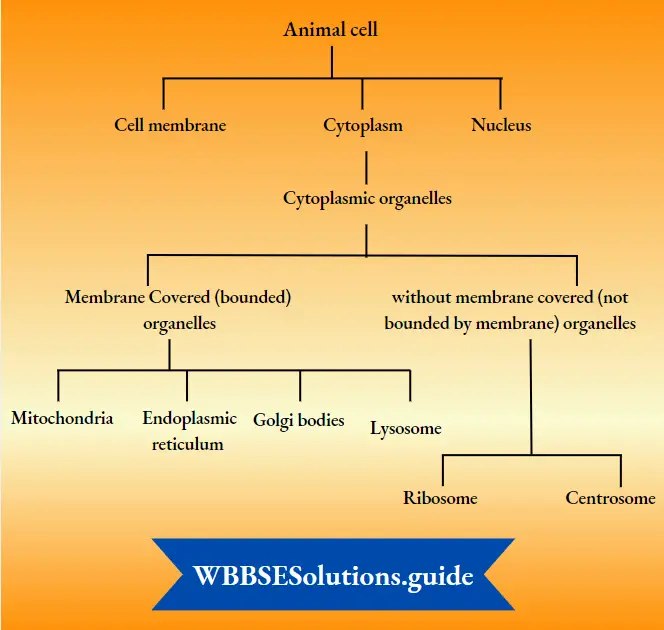
 The function of the Nucleus :
The function of the Nucleus :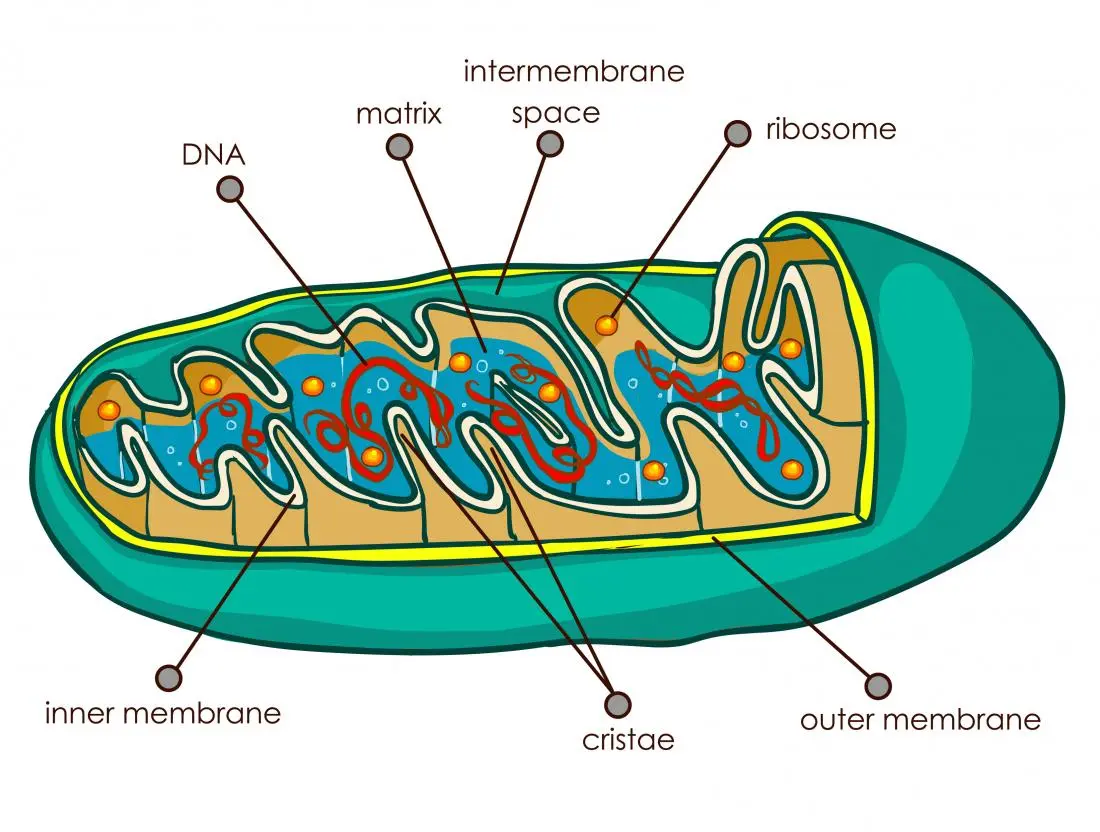



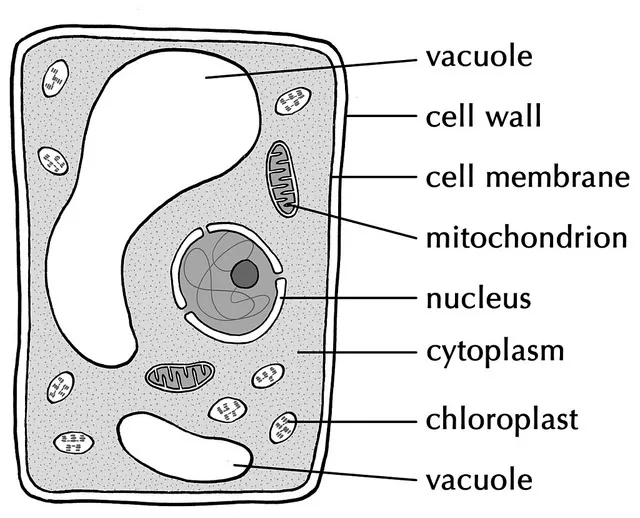
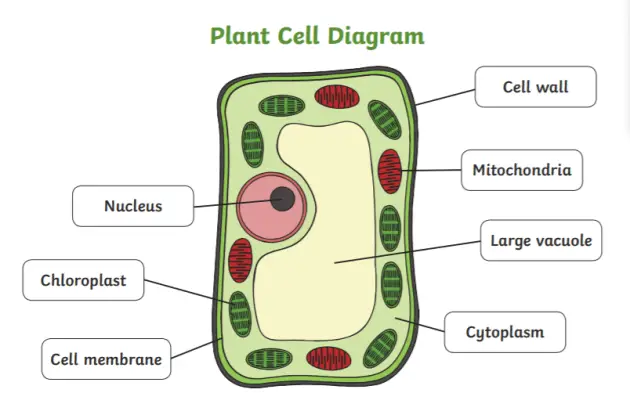
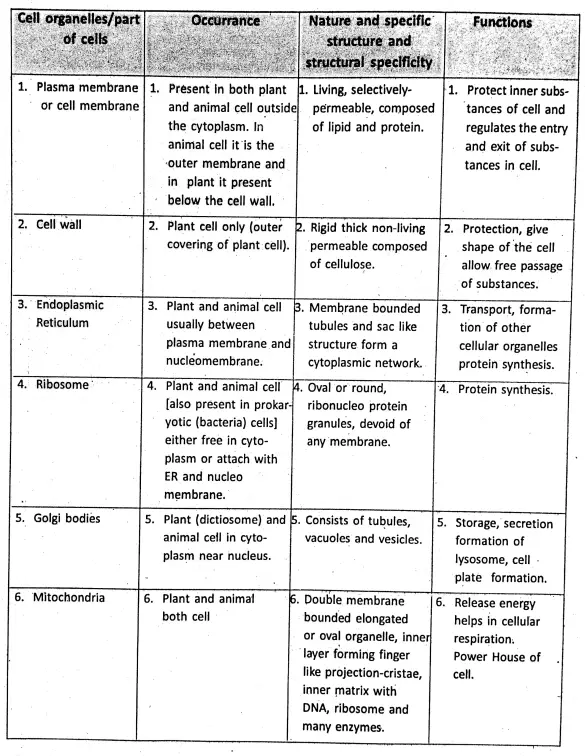
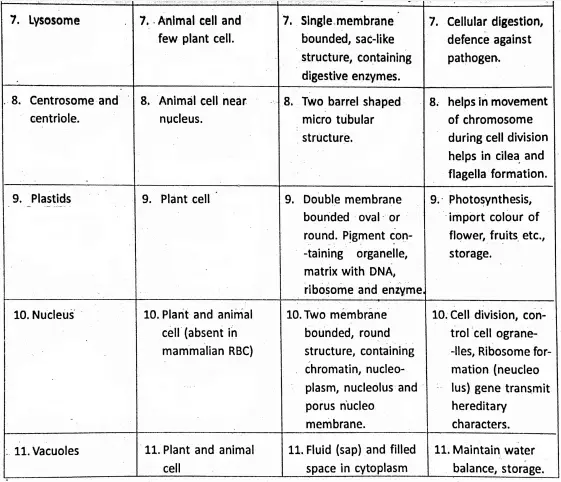
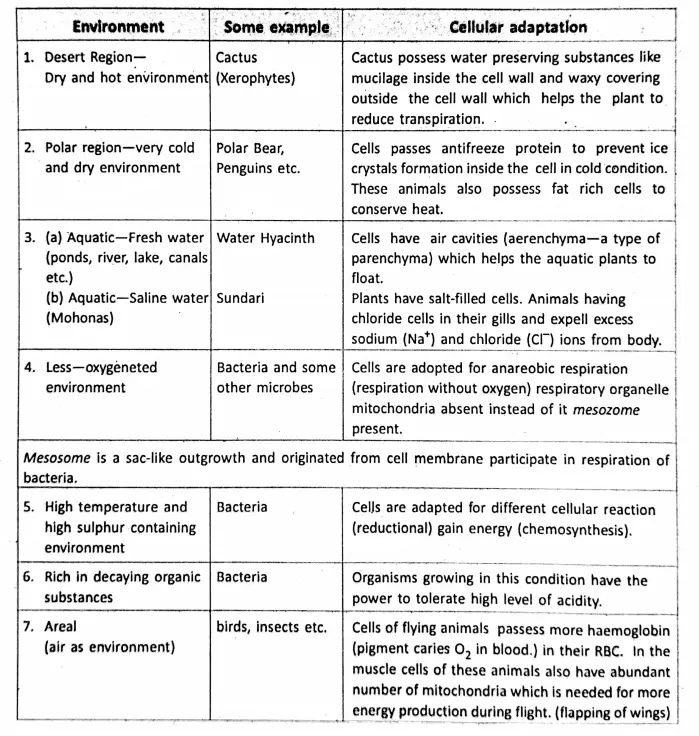
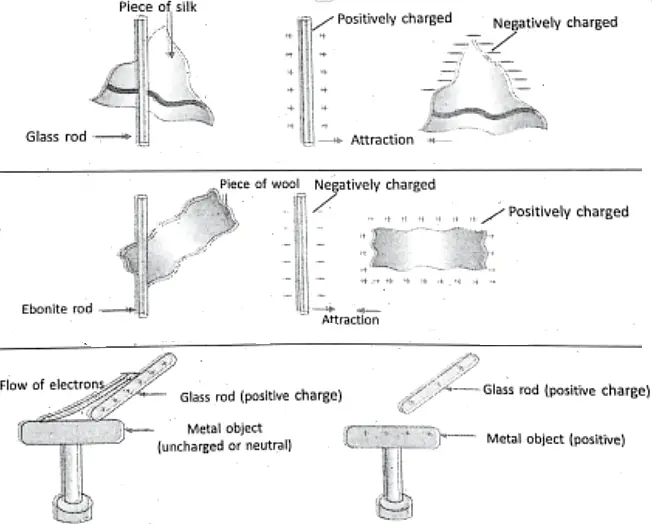


















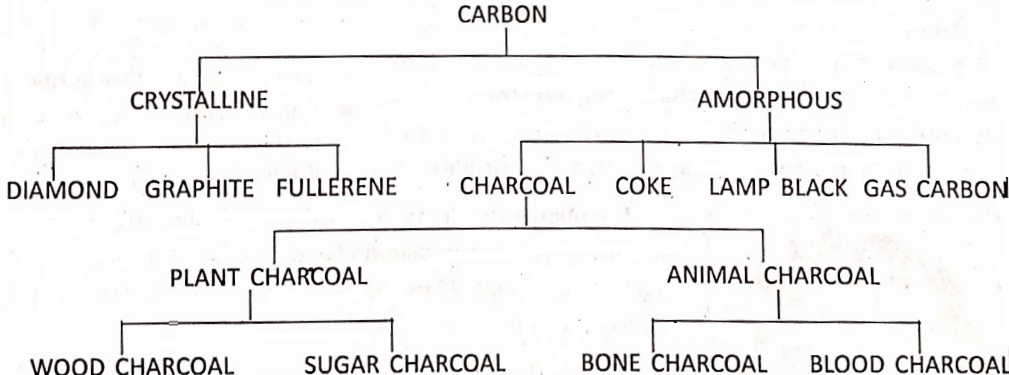




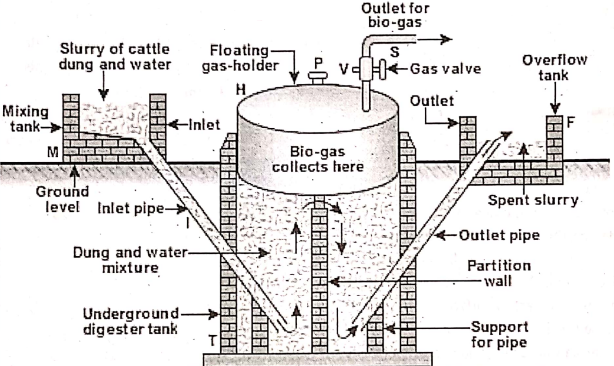


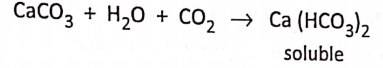




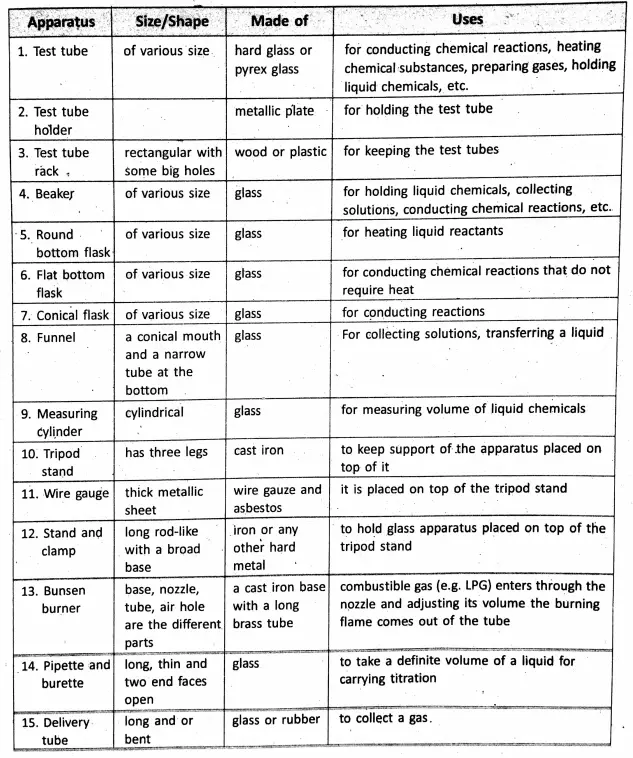


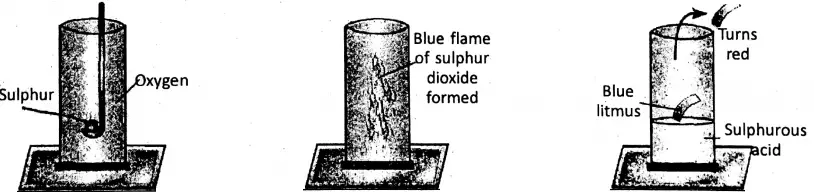
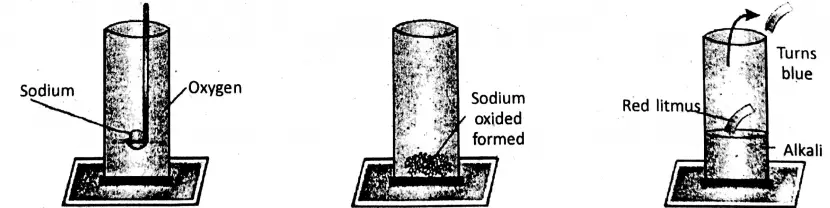
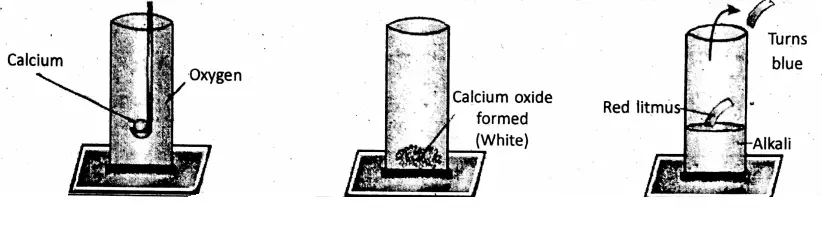

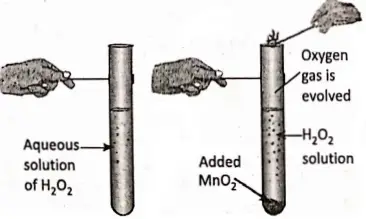
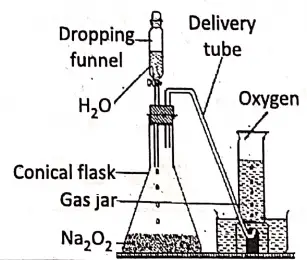
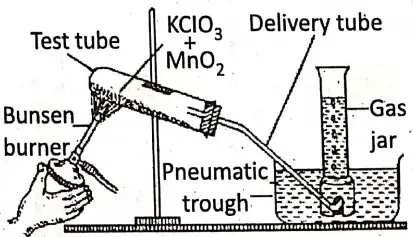
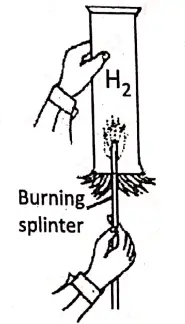
 3. Reaction with metals :
3. Reaction with metals :