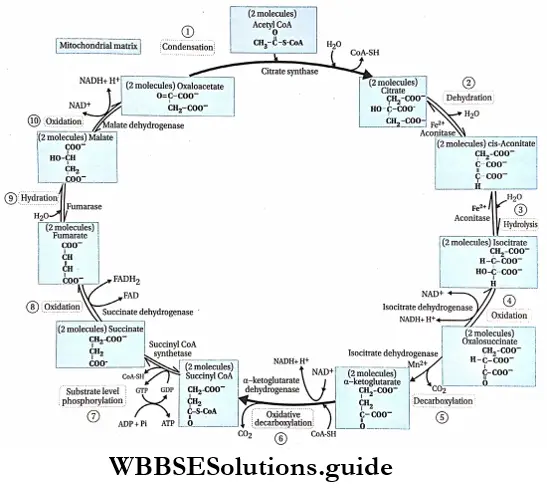
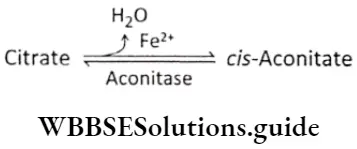
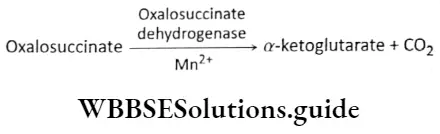
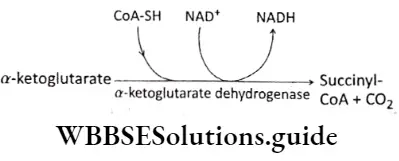
Plant Growth And Development Introduction
On your way to school or home, you see different kinds of trees and plants growing along the road. If you observe them carefully, you will find that they maintain a seasonal pattern in their growth such as the production of new leaves in springtime.
We also see a small bud blossom into a flower or a fruit develops from a flower. All these changes occur through growth and development.
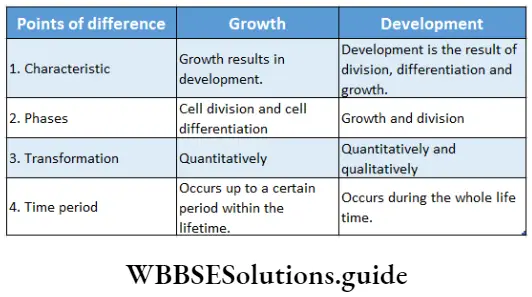
Growth is one of the important characteristics of all living organisms. Plant growth is unique as they have unlimited or indefinite growth throughout their life.
“plant growth and development notes for class 11”
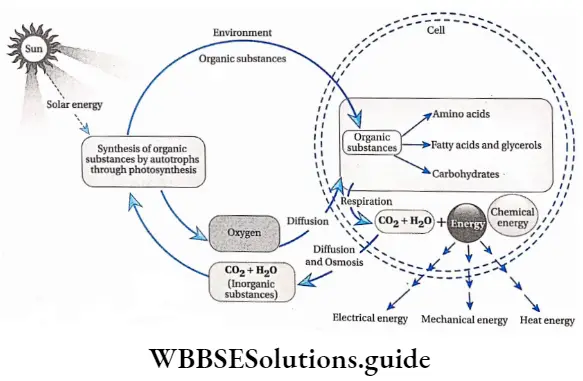
The development of a plant is a highly complex phenomenon. A zygote in the embryo sac starts dividing mitotically by utilizing available nutrients. In this way, it develops into a young sapling, that gradually develops into an adult plant.
Growth in plants includes an irreversible increase in size and in dry weight. It also leads to an increase in the amount of protoplasm. When the rate of anabolism is greater than the rate of catabolism, protoplasm in the cells increases. Increased amount of protoplasm in the cell leads to cell division which is followed by cell growth and differentiation.
Different parts of a plant are made of different tissues. These tissues are formed by the growth and differentiation of various kinds of cells. Hence, the development of the plant body occurs by growth and differentiation.
Growth is an irreversible or permanent increase in size, shape, volume, and dry weight of an organism or its parts or even that of an individual cell caused by the synthesis of new protoplasmic materials.
Generally, growth is accompanied by metabolic processes (both anabolic and catabolic), that occur at the expense of energy.

Seed Germination
Germination starts when a seed is provided with the appropriate amount of water, oxygen, etc.
Seed Germination Definition:
The process by which a resting embryo grows out of the seed coat as a seedling under suitable conditions is known as germination.
“detailed notes on plant growth and development”
Germination of a seed depends on several conditions, such as temperature, light, availability of water, oxygen, and nutrients. Plant hormones play a great role in germination.
Types Of Germination
There are mainly three main types of germination found in angiosperms. They are—
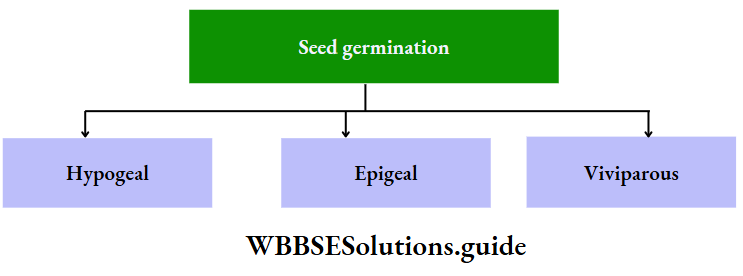
Hypogeal germination
Hypogeal germination Definition:
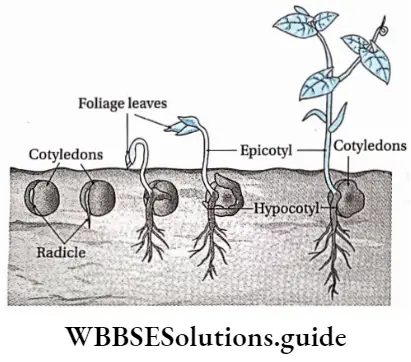
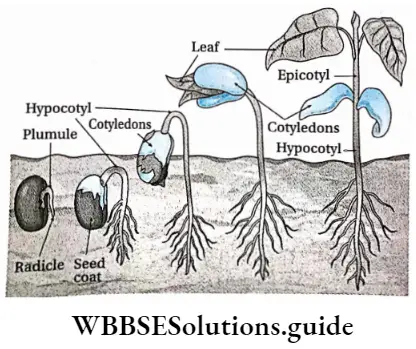
The germination where the cotyledons remain under the soil and hypocotyl does not elongate is known as hypogeal germination.
“plant growth and development NEET notes”
Explanation: During hypogeal germination, the epicotyl elongates rapidly. This helps the plumule to push upward and to emerge above the ground. The cotyledons and other parts of the seed remain beneath the soil. example Monocotyledonous seed— rice, wheat, maize, etc.; dicotyledonous seed—gram, pea, etc.
Epigeal germination
Definition: The germination where the cotyledons are raised above the ground due to the elongation of hypocotyl is known as epigeal germination.
Explanation: In this type of germination, the hypocotyl elongates more rapidly than epicotyl to form an arch. Thus, the hypocotyl comes out of the soil, pulling the cotyledon and the enclosed plumule through the ground. The cotyledons are projected out into the air and turn green.
These cotyledons act like the first pair of leaves and provide nutritive support to the growing plants. As the seedling starts to grow leaves, the cotyledons detach and fall to the ground, Examole Dicotyledonous seed—pumpkin, tamarind; monocotyledonous seed—onion.
plant growth and development short notes
Viviparous germination
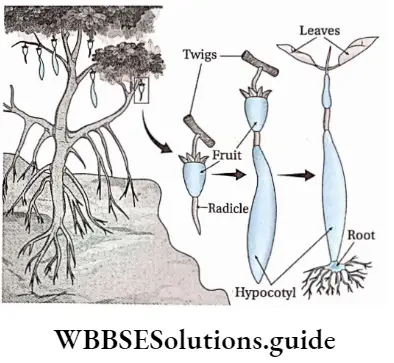
Definition: The germination in which the seed germinates before detachment of the fruit from the parent plant is called viviparous germination.
Explanation: This type of germination is found in mangroves. The radicle starts growing rapidly inside the fruit without any resting period. Thus, radicle comes out of the fruit. The radicle gradually increases in size.
After some time this structure falls off vertically from the plant on the salty mud and gets implanted in it. Then the whole structure starts growing as an independent plant.
In this way, neither the cotyledons nor the young twig comes in contact with the salty water or mud. example Rhizophora, Ceriops sp., Excoecaria agallocha, etc.
Phases Of Plant Growth And Plant Groth Rate
Generally, plant growth continues throughout their life. Primarily growth in plants is limited to the root and shoot apex. This growth occurs in the meristematic tissues present in those regions.
This type of growth is known as primary growth or apical growth. Growth in plants occurs at different rates and in different phases.
After primary growth, some plants increase in breadth by the division of lateral meristem and this type of growth is known as secondary growth. We have learned about these in Chapter 4.
Phases Of Plant Growth
Continuous division of meristematic tissue gives rise to new cells. These cells contain a thin cell wall and a large amount of cytoplasm. Initially, these cells do not contain any vacuole. During maturation, their size increases, and vacuoles appear. Permanent tissues also take part in growth by the process of differentiation.
The period of growth is generally divided into three phases, namely—
- Meristematic phase or phase of cell division,
- The phase of elongation and
- Phase of cell maturation or differentiation.

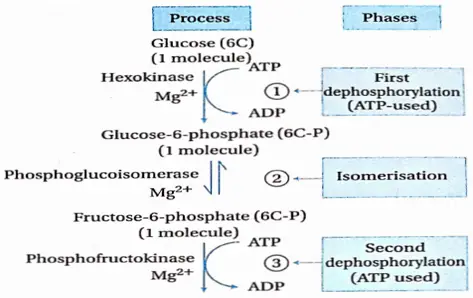
The phase of cell division
- In this phase, the cells of the meristematic tissue, present both at the root apex and the shoot apex, start dividing by mitosis. As a result, the number of cells increases rapidly.
- The rate of anabolism is very high in these cells. This causes a rapid increase in the dry weight.
- Amino acid synthesis also takes place in this phase.
- This phase is regulated by different phytohormones like auxin, gibberellic acid, and cytokinin.
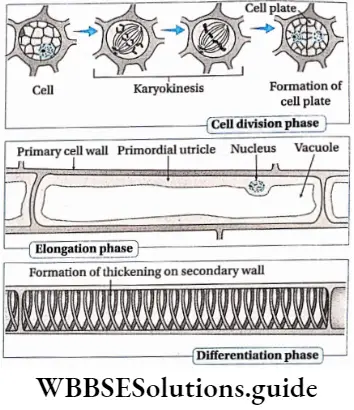
Phase of elongation
- In this phase, the cells increase in size by endosmosis of water.
- More vacuoles are produced and the turgor pressure inside the cell increases. This results in cell enlargement or elongation. Auxin plays an important role in cell elongation.
Phase of differentiation
- In this phase, the mature and normal-sized cells start differentiation and give rise to different cells—tracheids, trachea, collenchyma, etc.
- After differentiation, the cells stop growing.
- In this phase, the cells attain their maximal size in terms of wall thickening and protoplasmic modifications.
- Differentiation occurs in the cell for the completion of several physiological activities.
- After differentiation, several modification occurs in the cell such as the formation of a secondary cell wall. The secondary cell wall is composed of hydrophobic substances like lignin, wax, etc. As a result, the cells become impermeable and die.
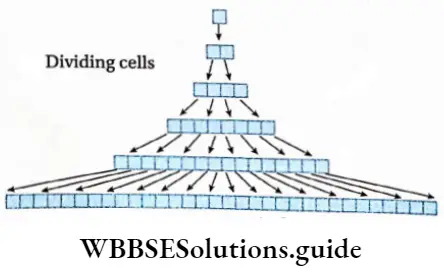
Demonstration of the phases of growth
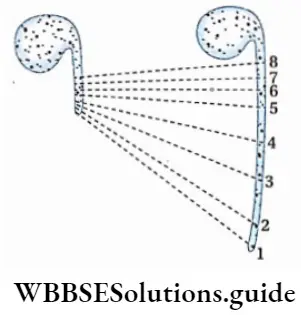
A wet filter paper is kept on a Petri dish. Now, some peas are kept on the wet filter paper and the seeds are covered with that filter paper. The peas are kept in this condition for two days. After two days, it is found that most of the seeds have produced radicles.
Now the seeds with straight radicles are separated. Water vapor on the radicles is removed by blotting paper. Gradations (1, 2, 3, 4, 5, 6, 7, 8) are marked at a gap of 2 mm on the radicles by using a marker pen. Now, these seeds are kept on wet filter paper for two days again.
After two days, the seedlings are placed on a graph paper. It is found that the distance between the two points (such as 2 and 3, 3 and 4) near the anterior portion has increased than the other portions. This region is known as the elongation region.
The posterior part of the radicle (5, 6, 7, 8) is known as the mature region. The growth rate of this region is comparatively low. This experiment proves that maximum growth of the root occurs at the region just above the tip.
plant growth regulators
Types Of Growth In Plants
Depending on different factors growth can be divided into various types.
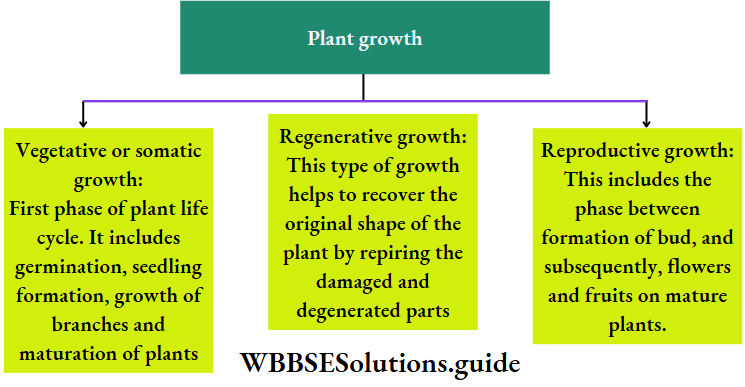
On the basis of nature
On the basis of nature, growth is of three types. They are briefly described in the chart given below.
On The Basis Of The Site Of Growth
On the basis of the site of growth, growth is of two types. They are discussed briefly in the chart given below.

On The Basis Of Metabolic Rate
On the basis of metabolic rate, growth can be divided into two types. They are discussed briefly in the chart given below.

Some Facts Related To Plant Growth
Some important and interesting facts related to plant growth are discussed below.
Region of growth: In plants, growth is localized in the meristematic regions. These can be apical, lateral, or intercalary according to their position in the plant body. They are responsible for the elongation of root and shoot, increase in breadth, and growth in internodal regions respectively.
Time of growth: Plants retain the capacity for unlimited growth throughout their life. The cells of meristems have the capacity to divide and change on their own.
Process of growth: The differentiated cells increase in volume and size by cell elongation and increased vacuolation. They form one or many types of tissues. Cellulose, pectin, lignin, etc., are deposited on the cell walls of newly formed cells, making the cell voluminous. As a result, cell size increases.
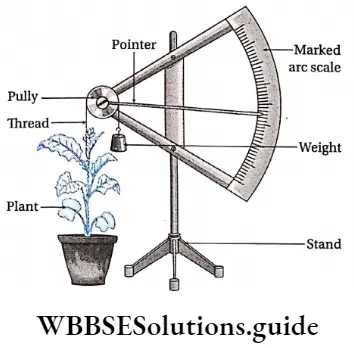
Growth measuring instrument: Auxanometer is used for measuring plant growth. This is of two types an auxanometer and an automatic auxanometer.
Annual ring: The concentric rings found in cross sections of gymnosperms as well as angiosperms (dicotyledonous) plants with woody stems are known as annual rings.

During secondary growth of cambium in woody plants, a secondary xylem forms these rings each year in spring and autumn. The age of a tree can be determined by counting its annual rings. The process of age determination in plants by counting the number of concentric rings is known as dendrochronology.
Growth Rate
Definition: Growth rate is defined as an increase in growth per unit of time.
Plant growth can be determined by various methods such as measuring changes in area, length, volume, height, and/or dry weight. Also, growth can be characterized by the development of leaves, flowers, and fruits. The actual growth of a plant can be measured by observing the growth rate.
“plant hormones and growth regulators notes”
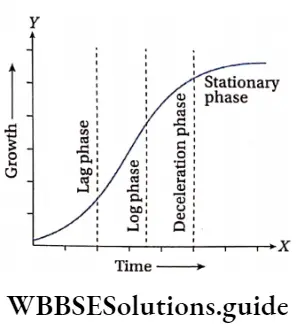
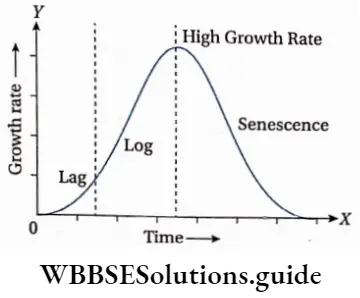
Phases of plant growth on the basis of differential growth rate
In the life cycle of a plant, the growth rate is different in different phases of its life. Initially, the growth rate remains very slow (lag phase). After that, the growth rate increases rapidly (log or exponential phase). The growth rate again slows down (deceleration phase) thereafter and at last stops completely (stationary phase).

Mathematical explanation of plant growth rate
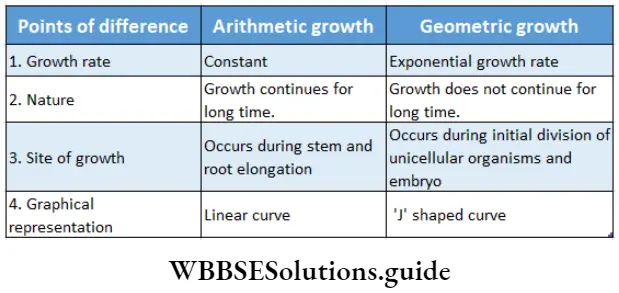
The growth rate can be expressed mathematically. An organism or a part of that organism can produce cells in a variety of ways. The growth rate can be determined arithmetically or geometrically.
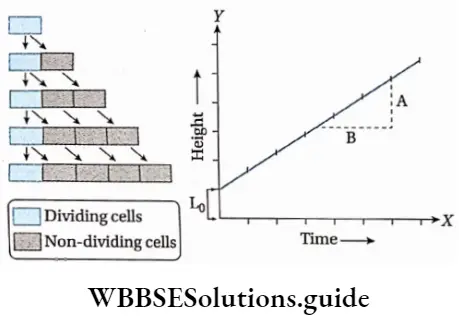
Arithmetic growth: The growth in which the growth rate remains constant from the beginning and the growth occurs arithmetically is known as arithmetic growth.
Explanation: Here, after the mitotic division, only one daughter cell continues to divide. Another daughter cell undergoes differentiation and becomes mature. Such as the root grows at a constant rate. This kind of growth gives a linear curve.
Mathematically, it is expressed as—
Lt = L0 + rt
[where Lt = length at time ‘t’
L0 = length at time ‘zero’
r = growth rate per unit of time
t = time of growth]
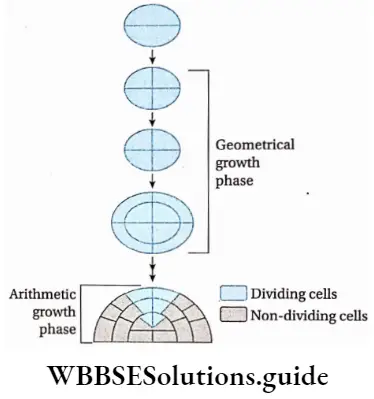
Geometric growth: The growth in which, all the daughter cells continue to divide and the number of cells increases geometrically is known as geometric growth.
Explanation: This type of growth can be observed in unicellular organisms and in the embryo, during their growth phase. Here, initially, the growth rate remains very slow, but after that, it increases rapidly and gives a T-shaped curve. However, the geometric growth rate is not constant.
So, in the case of plants, the curve becomes ‘S1-shaped (Sigmoid curve) in later stages. In most of the plants, primary growth occurs geometrically and the growth rate is different in different parts of the plant.
“what is plant growth “
The exponential or geometrical growth can be expressed mathematically as—
W1 = W0ert
[where W1 = final size (weight, height, number, etc.)
W0 = initial size at the beginning of the period
r = growth rate t = time of growth
e = a constant, its value is about 2.71]
Types of plant growth rate
Plant growth rate can be expressed in two ways. Both are briefly described below—
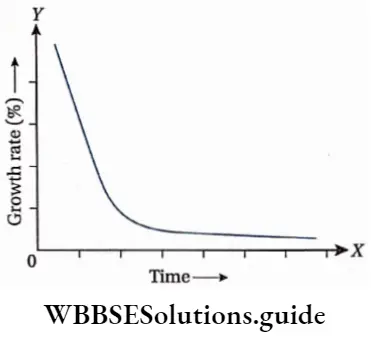
Absolute growth rate: The comparative measurement of total growth per unit time is called the absolute growth rate.
Mathematical expression
Absolute growth rate = \(=\frac{A_2-A_1}{T_2-T_1}\)
[Ai = Initial volume of the plant, A2 = Absolute volume of the plant, Tj = Initial time, T2 = Absolute time]
When a graph is plotted for absolute growth rate with respect to different major periods of growth, then the graph appears bell-shaped.
Relative growth rate: The increase in size per unit time of the initial size is known as relative growth rate.
Mathematical expression
\(\text { Relative growth rate }=\frac{1}{A_1} \times \frac{A_2-A_1}{T_2-T_1}\)
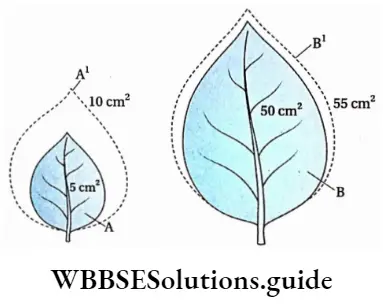
Example
Suppose,
- A leaf of 5cm2 becomes 55cm2 in 5 days.
- Another leaf of 10cm2 becomes 60cm2 in 5 days. Absolute growth rate: In case of first leaf = 55-5/5 = 10%
In case of second leaf = 60-10/5 = 10%
Therefore, in both cases, the absolute growth rate is the same.
Relative growth rate: In case of first leaf = 1/5 * 55-5/5 = 2%
In case of second leaf = 1/10 x 60-10/5 = 1%
Therefore, the relative growth rate of the first leaf is twice that of the second leaf.
Condition For Growth
Several external and internal conditions or factors are responsible for plant growth. Various factors are
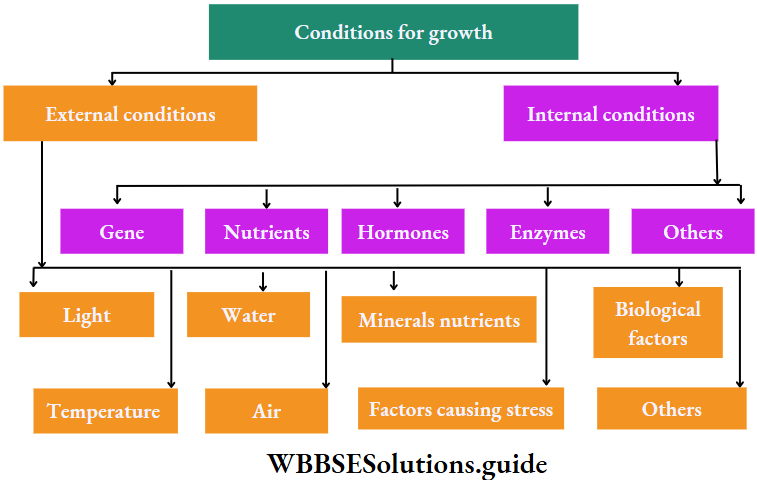
“chapter 15 biology class 11 notes “
External Factors
Plants also require certain environmental or external factors to synthesize food for their survival.
The environmental factors affecting plant growth include—
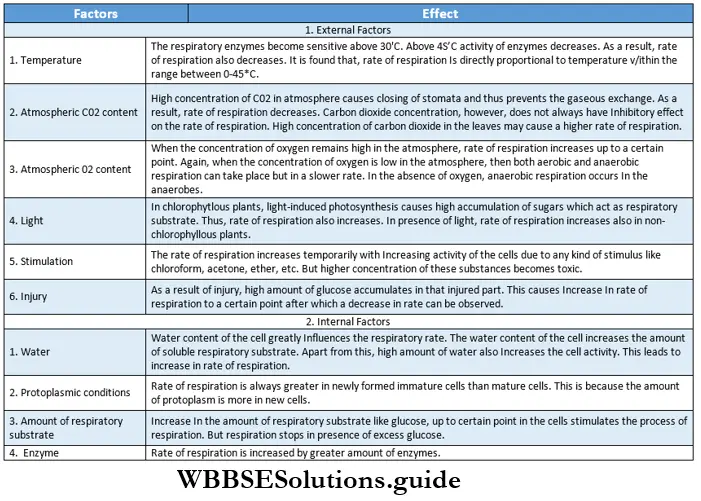
Light: Adequate light is perhaps one of the most important external factors influencing plant growth. The intensity, wavelength (color), and duration of light exposure are some influencing attributes of light. Various sources can be used to provide light to the plants.
The sources of light can be classified as natural and artificial sources. The natural source of light is the sun whereas the artificial sources include various types of lighting equipment.
The intensity of light: Photosynthesis and growth increase with the proper intensity of light. At the extremely high intensity of light, chlorophyll undergoes photo¬ oxidation.
As a result, leaves turn colorless, the rate of photosynthesis reduces and the growth rate also becomes low. Again at a high intensity of light, the rate of transpiration increases, and plant growth may be retarded due to dehydration.
Color of light: The rate of photosynthesis increases under blue, red, and purple light. Hence, the growth rate also increases under the illumination of these colors. Maximum chlorophyll formation occurs in the presence of red light. By absorbing red light phytochromes promote seed germination.
Blue light is essential for the growth of the leaves, whereas a combination of red and blue light promotes the flowering of plants.
The artificial light sources can be manipulated to adjust the intensity of the light as well. Also, there are certain plants, which require less light for growth. In such cases, the light can be filtered using protective shelters. This will minimize the exposure of the plants to sunlight.
Duration of light: Growth depends on the duration of light, i.e., photoperiod. Long-day plants (mostly of tropical climate) produce flowers when they receive long photoperiods or light hours above critical periods.
Likewise, short-day plants (mostly of temperate climate) are exposed to short light hours below the critical period. Day length may also affect the time when the first flower blooms, the number of flowers produced, and the number of fruit set. This topic is discussed in detail later in this chapter.
Temperature: Temperature is a crucial factor that influences the growth of plants. The temperature of the surrounding atmosphere as well as the temperature of the soil affects plant growth. Optimum temperature is essential for various plant processes, like photosynthesis, respiration, germination, and flowering.
The temperature that supports plant growth generally ranges from 25°C-30°C. Optimum temperatures for growth vary with species and the stages of development and usually fluctuate from night to day too.
Water: Most growing plants contain about 90% water. Water is the medium for transport within the plant and is the solvent for several substances which are important for the growth of a plant.
It is one of the raw materials for photosynthesis in higher plants. Water serves as the electron donor in the reducing reactions of photosynthesis.
A growing plant absorbs water from the soil and gives it off through transpiration. CO2 enters the plant through a film of water that surrounds the leaf. As the film evaporates, it is replenished by the plant.
Therefore, transpirational loss of water is important for growth. Water helps in the transportation of nutrients and activates the enzymes responsible for hydrolysis in the protoplasm.
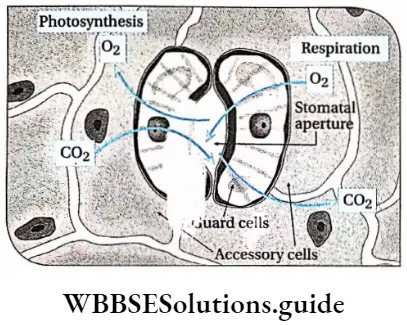
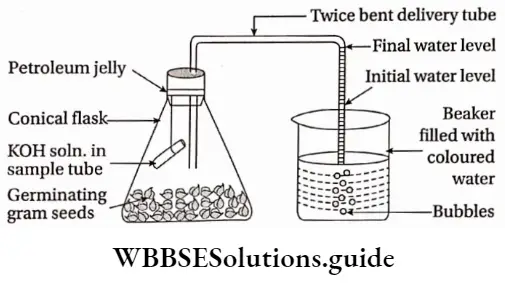
Oxygen: Aerobic respiration and other metabolic processes increase in the presence of oxygen. Aerobic respiration provides more energy required for biosynthesis, anaerobic activities, cell division, and growth. Seeds require oxygen to germinate. However, a very high concentration of oxygen sometimes causes growth retardation.
Carbon dioxide: Carbon dioxide is a raw material required for photosynthesis. Carbon assimilation of photosynthesis depends on the concentration of carbon dioxide. A high rate of photosynthesis produces a high amount of glucose which helps in the growth of the plant. Excess carbon dioxide causes retardation of growth.
Nitrogen: Nitrogen present in the air is trapped in soil by the process of nitrogen fixation. This increases the fertility of the soil. Plants take up nitrogen from the soil in the form of nitrogenous compounds for protein synthesis.
“plant growth and development handwritten notes”
Mineral nutrients: Plants get nutrients from the soil through water. Sixteen elements are considered to be essential for growth and development in plants. These elements help in the formation of various components of the cell wall, the formation of chlorophyll molecules, and also act as co-enzymes and help the growth of the plant.
The essential elements are divided into two groups macronutrients and micronutrients.
Macronutrients: These are elements or minerals that are required in relatively large amounts. These include carbon, hydrogen, oxygen, nitrogen, phosphorus, potassium, calcium, magnesium, and sulfur.
Micronutrients: These are elements or minerals that are required in small quantities but are essential for plant growth. These include iron, chlorine, manganese, boron, zinc, copper and molybdenum.
Soil: Soil, with proper humidity and the correct balance of all the minerals and nutrients, is one of the essential factors in plant growth. The right pH balance measures the alkalinity or acidity of the soil. The presence of certain chemicals is also necessary for the growth of plants.
Factors causing stress: At the region of the wound, growth is always high. This is because of the high rate of respiration and secretion of hormones at that place. Again lack of any factor in sufficient amount causes a low growth rate.
Biological factors: The external biological factors controlling plant growth, are
Growth promoting factors: Nitrogen-fixing microorganisms such as Clostridium, Anabaena, Nostoc, Azotobacter, and mycorrhizal fungi help in the growth of the plant.
Inhibitory factors: Parasitic organisms, disease-causing organisms as well and grazing of herbivorous animals cause a reduction of plant growth.
Internal Conditions
Besides external factors, the internal substances which help in plant growth, are known as internal conditions or internal factors.
Gene: Metabolic activities of a plant, such as cellular metabolism, synthesis of enzymes growth regulating chemicals, etc., depend on the genes. As a result, genetic factors influence the growth and development of the plants.
Nutrients: Different kinds of nutrients are required for the growth and development of a seedling. Nutrients increase metabolic activities, as a result of which the synthesis of enzymes, hormones, and protoplasmic substances increases.
Lack of nutrients affects the synthesis of hormones, enzymes, and protoplasmic substances, which in turn hinder plant growth. Nutrients play an important role in plant growth.
Variations in plant growth occur due to variations in nutrients
If a plant gets more nitrogenous nutrients than carbohydrates then its cells will produce more cytoplasm. Thus, plants will lack mechanical tissues. In this case shoot will be longer than the root and blackish-green colored leaves will grow.
If a plant gets more carbohydrates than nitrogenous nutrients then its cells will produce less cytoplasm. Cell walls will be thicker as carbohydrate is the main component of the cell wall. The plant will have more mechanical tissues.
Hormones:
Depending on their effect on growth, hormones are of two types—
Growth promoters: Hormones such as cytokinin, auxin, GA3, etc., promote plant growth.
Growth inhibitors: Hormones such as abscisic acid and ethylene are known to decrease growth rate and inhibit plant growth.
Enzymes: Concentration of enzymes responsible for various metabolic activity, rise by the activity of hormones in the cell. This in turn increases metabolic activities which help in the growth of the plants.
Besides all the factors discussed above, plant growth rate depends on the activity of protoplasm, number of stomata and their location, presence of chlorophyll, rate of transpiration, photosynthesis, etc.
Differentiation De-Differentiation And Re-Differentiation
The growth of a plant from a single-cell zygote includes various phases. Completion of these phases is not possible only by cell division or enlargement but also through differentiation. The life cycle of the plant is given in the flow chart below—

Some cells after cell division become permanent cells through certain structural and functional events. The rest of the newly produced daughter cells retain their capacity for division.
So in some parts of plants, indeterminate growth can be noticed. When the daughter cells of primary meristem undergo repeated divisions to form tissues, then it is called differentiation. After differentiation, through de-differentiation and re-differentiation, the whole plant body develops.
Differentiation
Definition: The process by which cells derived from the root and shoot apical meristems and cambium change into permanent tissues during the development of a plant body to serve a specific function is known as differentiation. In plants, permanent tissues are formed from meristematic tissues. It is an example of differentiation.
Characteristics:
- The meristematic tissues change into parenchyma cells by the process of differentiation.
- Some special cells can be formed by differentiation.
- Cells originating from the same meristem are different in function and structure due to differentiation.
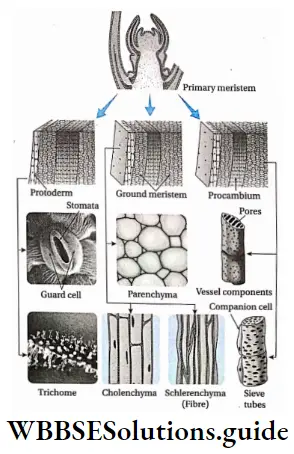
De-differentiation
Definition: The differentiated living cells, that have lost the capacity to divide but can regain the capacity of division under certain conditions is called de-differentiation.
In plants, the development of interfascicular cambium and cork cambium (phellogen) from fully differentiated permanent cells, is an example of de-differentiation.
Characteristics:
- De-differentiated cells mostly produce secondary meristematic tissue or cambium.
- Two main types of cambium thus produced are cork cambium and interfascicular cambium.
- Cork cambium is formed from the de-differentiation of collenchyma (living) or parenchyma cells lying immediately beneath the epidermis.
- Cells of medullary rays become meristematic and form interfascicular cambium.
Re-Differentiation
Definition: The process in which cells produced by de-differentiation of permanent tissues again divide and form new permanent cells to perform specific functions is called re-differentiation.
Phellogen or cork cambium gives rise to the phellem and phelloderm layer in plants. This is an example of re-differentiation.

Characteristics: The differentiated tissues again divide and form new cells to perform special functions.
Importance Of Differentiation De-Differentiation And Re-Differentiation
Formation of tissues and different parts: Different parts and tissues, are formed by the process of differentiation, for performing specific functions.
Development: De-differentiation along with growth causes development.
Secondary growth: Differentiation and re-differentiation are important for the secondary growth of a plant. Cambium plays a key role in this process.
Repairing: A damaged region can be repaired by de-differentiation and re-differentiation of permanent tissue present at that region.
Development And Sequence Of Development Process In A Plantcell
A zygote is formed by the union of a female gamete. It is the first cell in organisms. The sequential occurrence of the different functions like division, growth, maturation, senescence, and death completes the process of development of plants.
Definition: The sequence of changes that occur in the life cycle of a cell or an organism until its death, is known as development.
Explanation: The process of development includes germination of the embryo into a seedling, maturation of the seedling into a mature plant body, blooming of flower, reproduction, seed formation, and senescence of the plant parts with the culmination at death.
Sequence Of Developmental Process In Plant Cell
During the process of development, a plant cell goes through some sequential changes. They are—
Cell division: After protoplasmic growth, the zygote starts dividing by the process of mitosis or meiosis. Generally, cell divides in plants once each 10-20 hours. This time is known as generation time.
Cell growth: Protoplasmic growth occurs in the newly formed cells. Protoplasmic growth is brought about by the synthesis of different components of protoplasm like DNA, RNA, protein, etc. Due to this growth, the volume of the cell increases.
Elongation: Newly formed plant cell increases mainly in length. Little expansion in the breadth of the plant cells can be seen. Thus a plant cell elongates along the length of the plant axis.

Maturation: During this period, the vacuoles start increasing in size by absorbing and accumulating water in them. Protoplasmic growth is thus completed and the cells become fully developed and attain maturity.
Differentiation: Along with these elongation and maturation processes, cells get differentiated and matured. These mature cells now form tissue to perform specific functions and ultimately a tissue system.
Senescence: After a certain period of maturation, gradually the cells lose their activity and at last die.
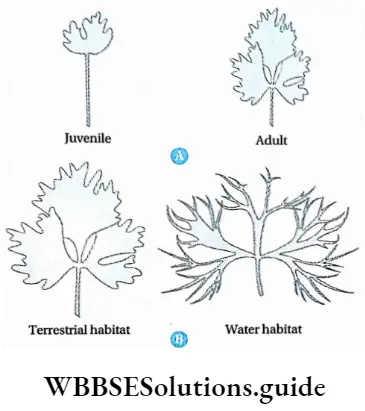
Plasticity
In response to changes occurring in the environment or | during different phases of life, plants form different j kinds of structures following different pathways. This ability is called plasticity.
An example of plasticity is heterophylly as observed in cotton, coriander, and larkspur. In these plants, the leaves of the juvenile plants differ from those of mature plants. In the water buttercup plant (Rannanculus aquatilis), aerial leaves differ from those produced in water.
Plant Growth Regulators
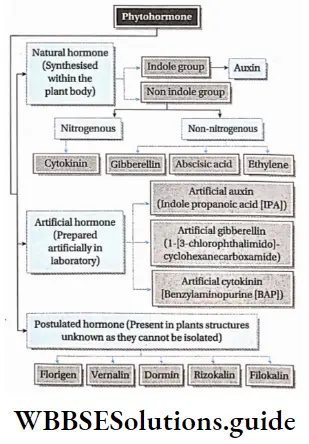
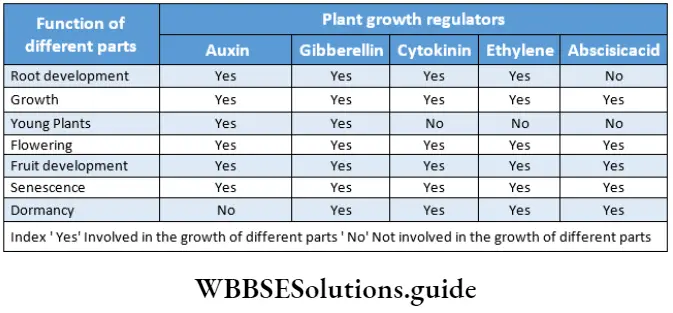
Phytohormones or plant growth substances regulate plant growth and development. Hence, they are also called plant growth regulators (PGRs). They are broadly divided into two groups based on their functions in a plant body.
One group of PGRs takes part in the promotion of plant growth such as cell division, cell enlargement, flowering, fruiting, seed germination, etc. They are called plant growth promoters. The other group is involved in various growth-inhibiting activities such as dormancy and abscission. They are called growth inhibitors.

These are signaling molecules produced in extremely low concentrations within certain tissues of the plant and exert their functions in different places (target site) after being transported.
According to Went and Thimann (1948), the definition of phytohormones is given below.
Definition: A phytohormone is an organic compound of plant origin, produced naturally in minute amounts controlling growth or other physiological functions at a site remote from its place of production and active in minute amounts.
Characteristics of plant hormones:
- Phytohormones are secreted from certain special regions such as apical meristematic tissue, in the plants.
- The concentration of hormones required for plant responses is very low (10-6 to 10-5 mol/I). The hormones are destroyed by specific enzymes after completing their assigned functions.
- Phytohormones can act at sites remote or near their site of production.
- They regulate all physiological activities related to the development and growth of plants.
- They also establish the chemical coordination between the cells. They can be produced at more than one site in the same plant body and are transported through the conducting system.
- The plant hormones differ in their chemical nature. They could be—indole compounds (auxins), adenine derivatives (cytokinins), derivatives of carotenoids (abscisic acid), terpenes (gibberellic acid), or gases (ethylene).
Classification of plant hormones:
Phytohormones can be classified into different types. They are given in the chart below—
Artificial phytohormones
- PCIB: para-chlorophenoxy isobutyric acid
- TIBA: 2,3,5-triiodobenzoic acid
- PICLORAM: 4-amino trichloropicolinic acid
- NAA: α-naphthalene acetic add
- ANOA: α-napthoxyacetic add
- BNOA: β-napthoxyacetic add
- 2,4-D: 2,4-dichloro phenoxy acetic add
- 2,4,5-T: 2,4,5-trichloro phenoxy acetic add
- MCPA: Methyl chlorophenoxyacetic add
Discovery Of Plant Growth Regulators
All of the five major groups of plant growth regulators were discovered accidentally. The first one to be discovered was auxin.
Discovery of Auxin
The presence of auxin in plants was confirmed by many scientists through different experiments.
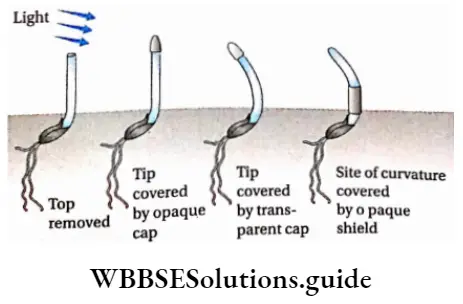
Darwin-Darwin’s experiment: Charles Darwin in his book The Power of Movement in Plants described 1 the phenomenon of bending of light in Phalaris canariensis (canary grass). Darwin along with his son Francis Darwin performed an experiment with Phalaris and observed that
- When the coleoptile was exposed to unidirectional light, it bent toward the direction of the light.
- When the coleoptile tip was covered with an opaque cap-like material, no bending occurred towards the light source.
- However, when the coleoptile tip was left uncovered but the portion just below the tip (growth zone) was covered, exposure to unidirectional light resulted in bending towards the light.
Darwin-Darwin’s experiment suggested that the extreme tip of the coleoptile is responsible for the perception of the light. It produces some biochemical substance which is transported to the lower part of the coleoptile where the physiological response of bending occurs.
He then decapitated the coleoptile and exposed the rest of it to unidirectional light to observe if any bending occurred. Bending was not observed, confirming the results of his first experiment.
Boysen-Jensen’s experiment: In 1913, P. Boysen-Jensen inserted pieces of mica in separate oats (Avena sp.) coleoptiles and on different sides to block the transport of the signal.
They showed that the transport of growth substance toward the base occurred on the darker side of the coleoptile unlike the side that was exposed
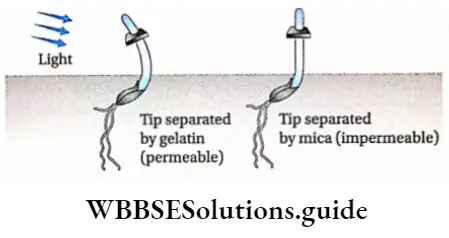
Paal’s experiment: In 1919, Paal confirmed Boysen-Jensen’s results by cutting off coleoptile tips asymmetrically and exposing only the tips to the light, replacing the coleoptile tips asymmetrically on the cut end. He found that whichever side of the coleoptile was exposed to light, bending occurred towards the other side.
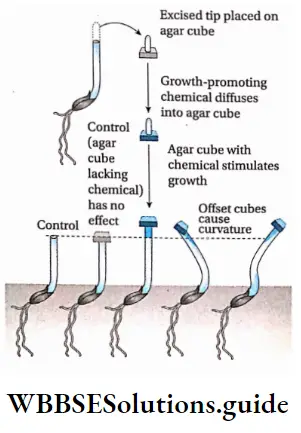
F. W. Went’s Experiment: In 1928, F. W. Went isolated a plant growth substance by placing agar blocks under freshly cut coleoptile tips for a period of time.
He did this, as he apprehended that agar would absorb the signaling chemical. He then removed agar blocks and placed them on new, freshly decapitated Avena stems. After the placement of agar blocks, stems resumed growth.
Discovery of gibberellin
Japanese farmers first observed the phenomenon of abnormal elongation in certain rice plants early in the season leading to unhealthy and sterile conditions. They gave many names to this disease but most commonly called it bakanae (foolish seedling).
In 1898, the causal agent of the disease Bakanae was deduced as the fungus Fusarium moniliformae.
“short notes on plant growth and development”
In 1926, Kurosawa discovered that the disease was caused by a substance secreted by the fungal species Gibberella resulting in a controversy about the true pathogen.
In 1935, Yabuta isolated the compound from Gibberella and called it gibberellin A. It was also revealed that Fusarium moniliformae is the asexual or imperfect fungi of Gibberella. This compound was found to stimulate growth when applied to dwarf rice roots.
Discovery of cytokinin
In 1913, Gottlieb Haberlandt discovered that a compound found in phloem had the ability to stimulate cell division. In 1941, Johannes van Overbeek discovered that the milky endosperm from coconut also had this ability. He also showed that various other plant species had compounds that stimulate cell division.
In 1954, Jablonski and Skoog extended the work of Haberlandt showing that vascular tissues contain compounds that promote cell division. In 1955, Hall and de Ropp reported that kinetin could be formed from DNA degradation products.
The first naturally occurring cytokinin was isolated from corn in 1961 by Miller. It was later called zeatin.
Almost simultaneously with Miller, Letham published a report on zeatin as a factor inducing cell division and later described its chemical properties. It is Miller and Letham who are credited with the simultaneous discovery of zeatin. Letham isolated 6- (4-hydroxy -3-methylbut-trans-2-enylamino) purine from immature kernels of Zea mays and named it ‘zeatin’.
Discovery of ethylene
The effect of ethylene on plant growth was observed by Fahnstock in 1858. In 1901, Neljubow observed that the etiolated pea seedlings in the presence of ethylene undergo a ‘triple response’, consisting of—
- Thickening of the subapical portion of the stem,
- Depression in the rate of elongation, and
- Horizontal bending of the stem.
Cousins (1910) reported that a volatile substance was released from ripened oranges that hastened the ripening of stored bananas. Sievers and True (1912) also confirmed the ripening of fruit by ethylene.
Almost all efforts were diverted to this economically important aspect of ethylene action. By the mid-1930s it was established that ethylene is produced autocatalytically just in advance of fruit ripening (Gane, 1934).
Discovery of abscisic acid
In 1963, abscisic acid was first identified and characterized by Frederick Addicott and his associates. They were studying compounds responsible for the abscission of fruits (cotton). Two compounds were isolated and called abscisin-l and abscisin-ll.
Abscisin II is presently called abscisic acid. Two other groups, at about the same time, discovered the same compound. One group headed by Philip Wareing was studying bud dormancy in woody plants.
They were able to isolate a substance from maple leaves responsible for stimulating cold season dormancy and gave the name ‘Dormin’. The other group led by Van Steveninck was studying the abscission of flowers and fruits from lupine plants. Plant physiologists agreed to call the compound abscisic acid.
Auxins
Definition: The indole group containing phytohormones produced at the plants’ apices naturally and responsible for accelerating the growth and development of the plant, are known as auxins.
The term auxin was coined by Charles Darwin in 1880 AD. The term ‘auxin’ has come from the Greek word auxin meaning ‘to enlarge or to grow’. It was first isolated from human urine. Indole Acetic Acid (IAA), and Indole Butyric Acid (IBA) are some examples of naturally found auxin.
Chemical nature of auxin
Auxin is composed of a carboxylic group (-COOH) and an unsaturated organic ring. It may contain nitrogen.
Characteristics of auxin
- Auxin is water soluble and moves from the morphological apex to the morphological base of a plant
- Auxin influences the phototropism and shows its action in the shaded part rather than the lightened part in the stem.
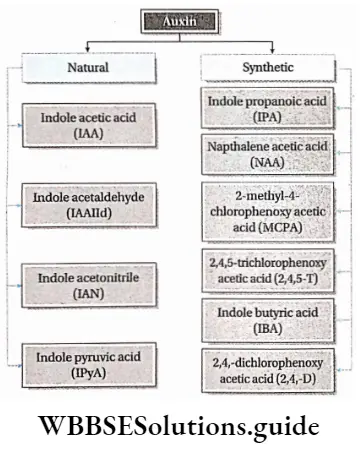
Types of auxin
On the basis of origin auxins are of the following types—
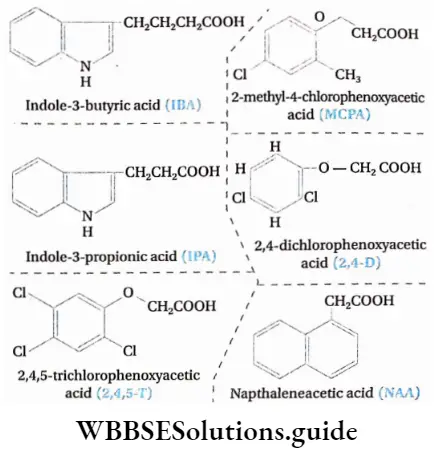
Functions of auxin
Auxin plays an important role in the growth of different parts of the plant. Its roles are briefly discussed here.
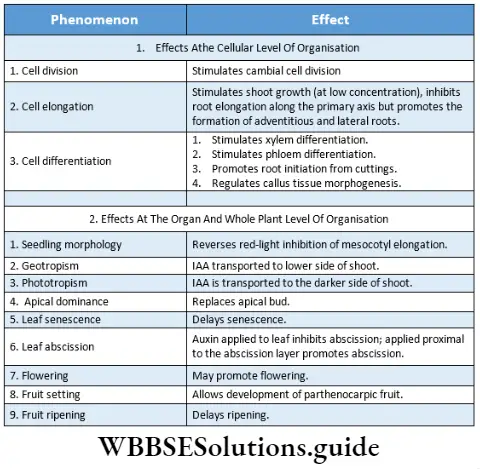
Cell Elongation: Cell expansion or elongation is one of the important functions of auxin. Cell elongation is the increase in cell size accompanying the process of plant growth. Auxin causes loosening of the cell wall by breaking the cellulose microfibrils. Thus it helps in elongation and expansion of cells.
Cell division: Auxin induces growth in secondary meristematic tissue by increasing the DNA content. As a result, mainly nuclear division occurs. Auxin is also responsible for the cell division that occurs during callus formation and root regeneration.
Regulation of apical dominance: Apical dominance may be defined as the control exerted by the shoot apex or apical buds over the growth of the lateral buds due to the presence of auxin. This is an example of developmental correlation where one organ of a plant affects another organ.
“plant growth and development notes with diagrams”
The dominance of the main apex is seen to suppress further development of the lateral apices. Thus they remain as axillary, buds often for long periods and sometimes permanently unless the main apex is removed.
If the shoot apex is subsequently decapitated (also referred to as apex removal), apical dominance is removed. Then only one or more of these lower axillary buds begin to grow out.
Growth of roofs and formation of adventitious roots: A minute concentration of auxin induces the growth of roots but excess concentration prevents root growth. Excess concentration of auxin also induces the development of adventitious roots from the upper nodes and lowermost nodes.
Activation of cambium: A low concentration of auxin increases cambium activity. In the process of grafting, auxin is used to increase the activity of the cambium that joins the vascular cylinders of stock and scion.
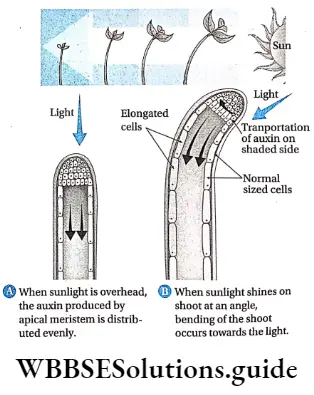
Tropic movements: Tropic movements (movement of the plant depends on external stimuli), such as phototropism and geotropism, depend on the concentration of auxin.
The concentration of auxin is always high on the opposite side of the light source, i.e., on the shaded side. As a result, the coleoptile end always bends towards the light and grows opposite to gravity.
The rate of cell division increases at the shaded region and the tip moves towards the light. The movement of coleoptile opposite to gravity is known as acropetal movement.
Again, due to less amount of auxin, the root shows movement towards gravity. This type of movement in roots is known as the basipetal movement.
As a result of the non-uniform distribution of auxin, plant growth is also non-uniform. The shoot shows phototropics and the root shows geotropic movements. to combine the best quality of different species.
Before grafting, the cut ends are treated with IAA. IAA stimulates the cambial activity to generate secondary vascular tissues. Secondary vascular tissues make connections between the vascular cylinders of stock and soon. Thus, a continuous vascular cylinder is produced.
Regulates abscission: The process through which a plant sheds leaves, flowers, seeds, and fruits is called abscission. An abscission zone forms on the base of these parts due to the low concentration of auxin gradually decreasing with increases in the age of pant.
Abscission and Auxin
The early investigations established that auxin commonly functions to retard abscission. Further investigations disclosed other influences of auxin. The onset of abscission was found to be correlated with the gradient, or balance of auxin across the abscission zone.
Auxin distal to the abscission zone tends to retard abscission, and auxin proximal to the zone tends to accelerate abscission.
Production of seedless fruit: Sometimes production of fruits occurs without pollination, this is known as parthenocarpy. If any plant is unable to produce fruit naturally, then the plant is treated with artificial auxin in order to produce seedless fruits. During parthenocarpy, usually, the concentration of auxin is higher in the pistils.
Sex expression: In 1950, Laibach and Kribben treated many plants of the family Cucurbitaceae with auxin. They found that the treated plants produced more female flowers. Hence, it is proved that a high concentration of auxin induces the production of female flowers (feminizing effect) in plants such as pumpkin, cucumber, etc.
Effect on water absorption: Auxin helps to increase the turgor pressure inside the cell. It affects the osmosis between the cells and helps in water absorption.
Callus formation: Application of auxin causes the formation of callus or tumor in the pith, cortex, etc. Differentiation of tissues in the callus is also induced by auxin in association with cytokinin.
Respiration: Auxin increases the availability of respiratory substrates to the respiratory enzymes thereby increasing the rate of respiration.
Metabolism: The application of auxin increases the rate of metabolism by increasing the functions of plant resources.
Nodule formation: An increase in the amount of auxin causes an increase in the number of root nodules in leguminous plants.
Commercial uses of auxin
Artificial hormones are used in agriculture and horticulture to get good yields. Auxin has several commercial uses. Some of those are—
Rooting in grafting: the stem cutting of different plant is kept in auxin stimulates the formation of adventitious roots on the stem cuttings.
Parthenocarpic fruit production: Auxin helps in the formation of parthenocarpic fruits such as grapes, papaya, bananas, etc. The hormones commonly used in this process are NAA, IBA, etc.
Prevent early abscission: Auxins help to prevent premature abscission of leaves and fruit. Though it promotes the abscission of older leaves and fruits.
Controlling weeds: 2,4-D, 2,4,5-T are widely used to kill dicotyledonous weeds from crop fields. These chemicals destroy the root system of the weeds but do not affect mature monocotyledonous plants.
Flowering: Application of NAA causes prompt flowering in some plants such as litchi, and pineapple.
Increase in yield: Yield can be increased by using IAA, IBA, and NAA in certain plants such as apples, pears, etc.
Preventing abscission: To prevent premature abscission in flowers, leaves, and fruits, artificial auxin such as 2,4-D or NAA is used.
Resistance towards frost: 2,4,5-T and sodium salt of NAA are used to protect vegetables and fruits from frost injury in hilly regions.
Wound repairing: A small amount of dilute artificial auxin is used to repair the wounds after pruning in the garden plants.
Increase in flower and cereal production: The number of flowers and quantity of fibers in cereals can be increased by applying auxin.
Sweetening of fruits: The sweetness of many fruits can be increased by applying IBA.
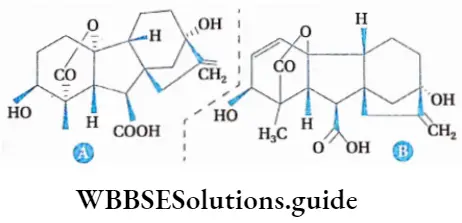
Gibberellins
Definition: Gibberellin is a tetracyclic diterpenoid substance, secreted from embryos and cotyledons, that activates genes of the target cell and breaks the genetic dwarfness and seed dormancy.
Gibberellin is a growth-controlling phytohormone. According to the scientists Hopkins and Huner, about 125 types of gibberellin are known. Among all these types, GA3 is the most important for plant growth, reproduction, and development.
Gibberellins are most often associated with the promotion of stem growth, and the application of gibberellin to intact plants can induce a large increase in plant height by elongating internodal parts. Gibberellins play important roles in a variety of physiological phenomena.
Chemical nature of gibberellin
Gibberellin is a non-nitrogenous tetracyclic diterpenoid compound.
Functions of gibberellin
Gibberellin influences plant growth and development. Its effects on plants are briefly discussed here.
Stem elongation: On application to the whole plant, gibberellin can induce remarkable elongation of the stem particularly in rosette and dwarf plant species. It affects mainly the intercalary meristems.
Sugarcane stores carbohydrates as sucrose in their stems. Application of gibberellins increases the length of grape stalks which ultimately raises yield. Gibberellin also increases the size of flowers, fruits, leaves, etc.

Seed Germination: Gibberellins are essential for accelerating seed germination and breaking dormancy. Gibberellic acids (GAs) can induce germination in seeds especially those which normally require low temperature or light to break dormancy.
Growth in aerial parts of the plants: If gibberellin is applied along with auxin, then aerial parts of plants will grow rapidly.
Bolting and flowering: The process in which internodal elongation of the rosette plants attain the normal height is called bolting. Gibberellin treatment on short day plants such as beet, and cabbage with rosette habit leads to stem elongation. It also induces flowering in many rosette long-day plants.
When the long-day plants growing in low temperature (2-4°C), are treated with gibberellin, then they show bolting and flowering in the absence of suitable conditions.
Breaking of bud and seed dormancy: Each and every seed and bud show dormancy for a certain period of time. During this period their growth has ceased. If any bud or seed is treated with gibberellin, then the gibberellin helps to induce growth in that bud or seed. Gibberellin affects the synthesis of mRNA and activates the gene responsible for growth.
Sex expression: Gibberellin helps in the sex expression of summer squash, cucumber, etc. A lower concentration of gibberellin stimulates the male flowers and a higher concentration stimulates the female flowers. In maize, the application of gibberelins induces the growth of gynoecium but prevents the growth of androecium.
Bud formation: Gibberellin helps in apical and axillary bud formation.
Parthenocarpy: The application of gibberellin causes the development of ovules and the formation of seedless fruits in many plants.
Commercial uses of gibberellin
Gibberellin has several commercial uses. Some of those are—
Production of seedless fruits: Gibberellin plays an important role in the formation and size of many fruits such as apples, grapes, etc. In the case of tomato, gibberellin is 500 times more effective than auxin.
Delayed fruit ripening: The application of gibberellin delays the ripening of certain fruits such as lemon, etc. It helps in the prolonged storage of those fruits.
Early maturation: For early growth and maturation of gymnosperms, GA3 and GA7 are used. These also induce early maturation of the fruit.
Role of gibberellin on the growth of- different plants:
- If GA3 is sprayed over barley grains, then along with ar-amylase, the production of other enzymes also increases. This, in turn, increases the yield of malt from barley.
- Application of gibberellin on sugarcane enhances the yield by increasing the internodal length.
- Gibberellin is also used for the production of a large number of seeds of the same size in lettuce.
- The application of gibberellin also helps to produce good-quality fruits. Gibberellin improves the size of fruit in apples.
- The size and stability of flowers in geranium plants can be maintained by the application of gibberellin.
- Gibberellin helps to break dormancy in potato tubers.
- Gibberellin speeds up the malting process of cereals in brewing industries.
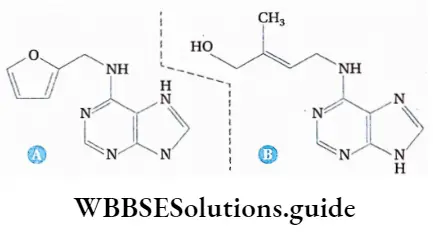
Cytokinin
Definition: The basic biochemical substances of the adenine group, produced in the growing regions of roots and shoots, immature endosperm, and leaves that induce cell division and budding, are known as cytokinins.
Naturally occurring cytokinins are purine compounds. Their structure resembles adenine and promotes cell division. Strong, Miller, and Skoog classify all the hormones related to cell division as cytokinin. These substances also help in the retardation of senescence.
Chemical nature of gibberellin
Cytokinin is a basic adenine-like nitrogen-containing purine molecule. Chemically it is known as 6-furfurylaminopurine. It is composed of nitrogen, oxygen, and hydrogen. Cytokinin is found as isopentenyladenosine (IPA) in most plants. The chemical formula of cytokinin is C10H9N5O.
Functions of cytokinins
The functions of cytokinins in plants are discussed here.
Cell division: In the presence of sufficient amount of auxin, cytokinin promotes cell division in plants by controlling the activities of cyclin-dependent kinases.
Cell enlargement: Cytokinins help overcome the stem tissue. In fact, cytokinin appears to promote the overall enlargement of cells when applied to a culture medium Growth can also be seen in cotyledons of some plants such as mustard, cucumber, sunflower, etc., by application of cytokinin.
Growth regulation in root and stem: Cytokinin prevents the uncontrolled growth of root and stem by inhibiting cell elongation. Thus, endogenous cytokinin seems to regulate the growth of the root and stem.
Tissue differentiation: Organs in tissue culture show a spectacular response to cytokinin. With a low cytokinin supply, the tissue remains as an amorphous undifferentiated callus. Bud formation and shoot initiation depend on a higher concentration of cytokinin.
An interesting observation on morphogenesis in tobacco callus culture is that a high cytokinin-auxin ratio results in the production of shoots but not roots. But a low ratio leads to the opposite effect producing roots only.
In addition to their role in leaf expansion, cytokinins also regulate chloroplast formation. When cytokinin is absent, plastids are formed but remain undifferentiated. Both light and cytokinin are necessary for grana development and conversion of proplastids into chloroplasts.
Retardation of senescence: The retardation of senescence by cytokinin is a well-known phenomenon. Richmond and Lang (1957) first discovered that when leaf discs are kept in water, senescence appears within a few days as evident by the drainage of chlorophyll, protein, and other nutrients.
But when cytokinin is added to the leaf discs, senescence is delayed through the drainage of nutrients and checking the degradation of chlorophyll and protein. This senescence retarding property of cytokinin as mediated through the retention of chlorophyll is known as the Richmond-Lang effect.
Senescence
The slow deterioration of structural and functional characteristics, etc., in a mature plant due to aging, preceding the death of an organ or the whole plant is known as senescence.
1. Types: Scientist Leopold (1961), discussed about four types of senescence in plants,
- Complete senescence (the whole plant is affected at once and dies slowly, for example, paddy, wheat, bamboo, etc.);
- Stem senescence (parts above the ground (stem) are affected and die, for example, rhizomes of banana, ginger, yam, etc.);
- Sequential senescence (older parts die first followed by younger parts, for example, pine, eucalyptus, etc.);
- Simultaneous senescence (all the leaves fall off leaving the stem and root alive, for example, apple, oak, etc.).
2. Changes occurring during senescence:
- The rate of photosynthesis decreases which leads to a decrease in the storage of starch,
- Chlorophyll disintegrates and the storage of anthocyanin increases in cells.
- The amount of protein decreases, hydrolyzing enzymes such as protease and nuclease are produced,
- Before detachment of leaves, the nutrient components are transported to shoots.
- The cell membrane and cell organelles disintegrate,
- Anabolic metabolism reduces.
Importance:
- Senescence determines the life span of an individual cell, organ, or whole plant.
- The inactive and/or older plant parts are replaced by new, young, and active plant parts.
- During senescence cellular components are translocated to the newly formed organs from the senescent parts and are used for their growth and development,
- The rate of transpiration is reduced during winter due to the senescence of leaves. It is a kind of adaptation to reduce the loss of water.
Axillary bud formation: Cytokinin is important for bud formation in plants. It mainly helps in the formation of axillary buds.
Mobilization of nutrients: Mothes (1961) observed that when a particular area of the leaf is treated with cytokinin, the area remains green showing a delay of senescence. Whereas, the untreated area loses its green color and becomes yellow, showing symptoms of senescence.
Here the nutrients like amino acids, auxins, and phosphorous are drawn or mobilized preferentially from the other parts of the leaf so that the treated area remains green at the expense of the untreated area.
Breaking of seed or bud dormancy: The application of cytokinins can stimulate germination and break dormancy. When dormancy is imposed either by high temperature (thermo-dormancy) or by accumulation of an inhibitor like abscisic acid, coumarin, etc., then gibberellic acid alone is not capable of overcoming dormancy.
The addition of cytokinin can replace the red light requirement in seed germination opposes the action of inhibitors and induces germination.
“stages of plant growth and development explained”
Flowering: Cytokinin can induce flowering in short-day plants in low light than usual. example Lemna, Wolfia.
Parthenocarpy: Like auxin or gibberellin, cytokinin also helps to produce parthenocarpic fruits.
Protection: The application of cytokinin makes the plants resistant to diseases. It also protects the plants in high and low temperatures.
Chlorophyll production: Colourless idioblast (isolated plant cell that stores pigments and other substances) can be converted into chloroplast as rapid production of chlorophyll occurs in the presence of cytokinin.
If etiolated leaves are treated with cytokinin before being illuminated, they form chloroplasts with more extensive grana and chlorophyll. Also, photosynthetic enzymes are synthesized at a greater rate upon illumination.
Stomatal movement: Cytokinin has a distinct action on the mechanism of stomatal movement. Treatment of whole leaf with cytokinin has been reported to increase the size of stomatal aperture thereby increasing the rate of transpiration also increases.
Apical Dominance: Application of cytokinin on lateral buds counteracts the apical dominance which can be due to the presence of terminal bud or due to applied auxin. This has been interpreted as an increase in IAA transport and mobilization of metabolites from the apical region to the point of application of cytokinin.
Commercial uses of cytokinin
Cytokinins have several commercial uses. Some of those are—
Maintains freshness of flowers: Normally flowers wilt after a few days of plucking. Flowers treated with cytokinin will remain fresh for a longer period. This helps in the storage of flowers.
Protection: Plants become resistant to heat, cold, and diseases on treatment with cytokinin.
Tissue culture: Cytokinin helps in cell division and differentiation in tissue culture.
Ethylene
Definition: The gaseous compound produced in minute amounts that helps in fruit ripening and leaf abscission is known as ethylene.
Ethylene is a gaseous growth inhibitory phytohormone. It inhibits cell division, DNA synthesis, and growth in the meristems of roots, shoots, and axillary buds. Apical dominance often is broken when ethylene is removed, apparently because it inhibits polar auxin transport irreversibly. Often ethylene inhibits cell growth and delays differentiation.
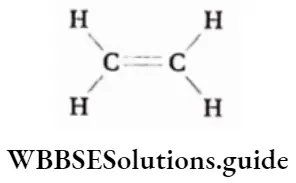
Chemical nature of ethylene
The structure of ethylene is very simple. It is an unsaturated symmetrical hydrocarbon compound. Since it is highly soluble in water as well as in a lipophilic system, it can easily move through plant tissues.
Production of ethylene is controlled by auxin and red light. Auxin promotes ethylene synthesis and red light represses its production. The action of ethylene is competitively inhibited by CO2 and promoted by O2
Functions of ethylene
Ethylene plays an important role in different aspects of plant growth and development. Those functions are—
Fruit ripening: Ethylene plays an important role in fruit ripening. It is produced in mature but unripe fruits and then it a initiates chain of reactions which finally lead to ripening of the fruits.
Ripening usually starts at one region of a fruit, spreading to other regions as ethylene diffuses freely from cell to cell and integrates the ripening process throughout the fruit.
Triple response: Ethylene affects the growth of plants. It inhibits stem elongation and initiates horizontal growth of stems with respect to gravity. It also helps in the thickening of the subapical portion of the stem.
Senescence and abscission: Ethylene has been implicated in the regulation of leaf senescence in certain plants. Exposure of Arabidopsis plants to ethylene induces premature yellowing of the leaves. Ethylene also stimulates the formation of abscission zones in leaves, flowers, and fruits.
Stimulate flowering sit pineapples: The promotion of flowering by ethylene was first observed in pineapples in the 1930s. It has become an important horticultural practice for the production of pineapple and other members of the Bromeliaceae family.
Growth promotion: Ethylene promotes the growth of internode or petiole in deep water rice varieties (grows in flooded fields). It helps the leaves and upper parts of the shoot remain above water. Ethylene also promotes root growth and root hair formation, thus helping the plants to increase the absorption surface of their root system.
Growth prevention: Ethylene inhibits the growth of stem, root, and axillary buds in etiolated plants. The major cause of the overall growth inhibition is due to retardation of the mitotic process in the respective meristems.
Root initiation: Ethylene stimulates rooting from stem cuttings. Also, it helps in root hair proliferation. At low concentrations, it stimulates root growth but at higher concentrations, it inhibits root growth. Release of dormancy: In some species (e.g., peanut, sunflower, potato tuber, etc.), ethylene breaks seed and bud dormancy.
Epinasty: When the upper surface of a leaf grows more than the lower surface, then the leaf bends downwards, this is known as epinasty. This is controlled by ethylene. But high concentration of ethylene causes hyponasty (opening of downward folded flower petals or leaves, etc.).
Negative feedback: Secretion of ethylene prevents the production and secretion of auxin.
Wound and stress response: Ethylene is an important signal in many such abiotic stress situations and also in plant-pathogen interactions. Production of ethylene can be induced by pathogen invasion, by fungal toxins as well as by race-specific and endogenous elicitors.
Ethylene may activate plant defense-related processes such as the production of phytoalexins, pathogenesis-related (PR) proteins, and cell wall alterations.
Responses to physical stimuli: Ethylene has been proposed to function thigmomorphogenesis. Exogenous application of ethylene can result in morphological and physiological changes resembling thigmomorphogenesis. Ethylene production may be one of the responses to mechanical wounds or uneasiness.
Commercial uses of ethylene
Ethylene has several commercial uses. Some of those are—
Sprouting of storage organs: Ethylene is used to break the dormancy in storage organs like tubers, rhizomes, and corm bulbs of potatoes, ginger, onion, etc. The application of ethylene also causes sprouting in certain plants.
Increase in the number of female flowers: The application of ethylene in many plants such as pumpkin, cucumber, etc., shows promising effects. Thus, the number of female flowers increases which in turn results in more fruits.
Flower whorl formation: Ethylene inhibits the growth of apical buds and stimulates the growth of axillary buds. As a result, whorls of flowers are produced by the axillary buds. Thus, a compact flowering stem is produced.
Controls the production of fruits and flowers: The application of ethylene controls the growth of excess flowers and fruits in some plants like cherries. It helps to initiate flowering in mangoes. Aside from flowering it also helps in synchronisation of fruit setting in pineapples.
Abscisic Acid
Definition: The sesquiterpene compound found in plants that act as growth inhibitors, senescence, and dormancy inducer is known as abscisic acid.
Unlike growth-promoting phytohormones such as auxins, gibberellins, and cytokinins, abscisic acid (ABA) plays a mostly inhibitory role in plants. It induces dormancy in seeds and buds, and hence, is known as a dormancy-inducing hormone.
It plays an important role in plants during unfavorable environmental conditions (stress) and helps them to cope with it. So, ABA is also known as the stress hormone.
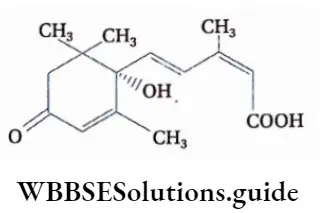
Chemical nature
Abscisic acid is a 15-carbon sesquiterpenoid compound having an asymmetric carbon. It has two optical isomers. the naturally occurring aba is represented as (+) and the synthetic one is racemic, i.e., equivalent mixture of (+) and (-) enantiomers. Its trivial name is 3-methyl-5-(l-hydroxy-4-oxy-2,6,6-trimethyl- 2-cyclohexen-l-yl)-cis,trans-2,4-penta-dienoic acid.
Function of abscisic Acid
As a growth regulator, ABA has several important functions in a plant’s life. They are—
Growth inhibition: ABA acts as an inhibitor of shoot growth in. plants growing in water-deficit conditions. The current understanding of the role of ABA is that it controls root growth.
Endogenous ABA deficiency leads to ethylene production and this interaction is involved in the effects of ABA status on shoot and root growth. ABA can counteract the responses of plants to each of the growth-promoting phytohormones.
Growth promotion: At very low concentrations, ABA has been found to promote some growth processes like rooting of stem cuttings, callus growth in soybean in association with kinetin, and an increase in the frond number of duckweed (Lemna polyrhiza).
Stress response and prevention of transpiration: ABA plays an important role in the regulation of plant responses to environmental stresses, especially drought. The rise in endogenous levels of ABA in leaves under drought situations can inhibit stomatal opening thereby inhibiting transpiration.
Such inhibition plays an important role in water conservation mechanisms. During water stress conditions, guard cells in stomata secret more ABA. This alters the permeability of the plasma membrane of guard cells.
Thus, the plasma membrane renders the exit of K+ ions from guard cells and helps the entry of H+ ions into it. Therefore, stomata remain closed during this time. This helps in the desiccation tolerance of the plant.
Hydrolysis of carbohydrates: ABA prevents translation of or-amylase mRNA. Hence, it prevents the synthesis of or-amylase and carbohydrate hydrolysis Thus it opposes the functioning of gibberellin which helps in the production of the or-amylase enzyme.
Leaf senescence: ABA induces senescence and abscission in leaves and other parts of the plant. ABA is responsible for the activation of the hydrolase enzyme, which dissociates protein and nucleic acid.
Activation of cambium: ABA prevents the activation of the cambium in late autumn, winter, and early spring.
Storage of protein: ABA stimulates protein storage during seed formation.
Seed development, germination, and dormancy: During the development of a variety of seeds, the ABA level rises sharply and then declines. In many seeds, the highest concentration of ABA is found in the embryo at a time when their dry weights increase rapidly.
The germination of most non-dormant seeds can be inhibited by exogenous ABA. Activities of various enzymes, which rise during germination, appear to be specifically inhibited by ABA. At the end of the dormancy, the ABA level decreases and GA3 becomes active.
Seeds cannot germinate in the absence of either of the mentioned conditions. ABA also induces dormancy in adventitious buds. On the other hand, ABA helps the seed or adventitious buds to overcome unfavorable conditions by inducing dormancy in them.
Root geotropism: Experiments have indicated that the root cap is the source of growth-inhibitory substances, formed in response to gravity. These results have led to the hypothesis that when roots are maintained in a horizontal position. i.e. subjected to geotropic stimulus, ABA produced in the root cap moves basipetally to the growing part of the root. It is accumulated in the lower half of the root causing a positive geotropic response.
Fruit growth and flowering: Ripening fruits are the richest sources of ABA, yet the application of ABA to fruits has little effect on the process of ripening. Ripening of grapes is an exception where ABA has the capacity to hasten the ripening and coloring of the fruit. ABA application in a very low concentration has little promoting effect on flower growth. High ABA inhibits or delays flowering in a number of plants.
Commercial uses of abscisic acid
The commercial uses of abscisic acid are—
Dormancy: ABA is applied to prolong dormancy of buds, seeds, and other storage
Root initiation: ABA initiates rooting in stem cuttings of some plants.
Flowering: ABA stimulates flowering in short-day plants growing even in long-day conditions.
Prevents transpiration: ABA causes closure of stomata and prevents transpiration. This does not affect gaseous exchange through the stomata. This does not affect the process of photosynthesis too much.
Abscission
The natural phenomenon of the detachment of different plant parts as a result of aging is known as abscission. In deciduous plants, the abscission of all the leaves occurs at the same time during autumn.
As a result, the plants become leafless. There is no particular time for abscission in evergreen plants. In these plants abscission of leaves occurs throughout the year.
1. Abscission zone or layer: The region of separation between the base of the leaf petiole, flower stalk, fruit stalk, and branches, produced by a thin plate of cells oriented at right angles to the axis of the subsequent organs is transformed into the abscission zone or abscission layer.
2. Changes occurring during abscission:
- The abscission layer occurs at the base of mature leaves due to a decline in auxin content and an increase in ethylene content.
- Metabolic wastes are stored in the leaves and are removed from the plant along with the leaves.
- The cells across the abscission layer contain pectinase and cellulase which hydrolyse the pectin and cellulose respectively and form a separation layer at the abscission zone. Due to the destruction of the cells in the abscission zone, the leaves detach from the plant, [iv] Lignin, suberin, etc., are secreted from the wounded region after abscission. These substances prevent the loss of water by covering the wound.
3. Importance:
- Abscission is a controlled process, resulting in the removal of older and inactive plant parts.
- The process permits the plants to achieve efficient fruit dispersal and to survive an unfavorable period.
- The nutrients remain stored in plants as the nutrients are transported to the stem before abscission.
- The productivity of plants increases as a result of abscission.
Seed Dormancy
Definition: The failure of a viable seed to complete germination under favorable conditions is known as seed dormancy.
Seed is an important stage in the life cycle of higher plants with respect to the survival of the species. It is the dispersal unit of the plant, which is able to survive the period between seed maturation and the beginning of the next generation as a seedling after it has germinated.
For this survival, the seed, mainly in a dry state, is well equipped to sustain extended periods of unfavorable conditions. This period of inactiveness of seed is known as dormancy.
The seeds enter into the dormant state to get the optimum result of germination. Dormancy prevents pre-harvest germination as well. The dormancy period may extend from a few days to a few years.
Non-dormant seeds that are exposed for some time to unfavorable germination conditions may enter a state of dormancy again, which is called secondary dormancy.
Causes Of Dormancy
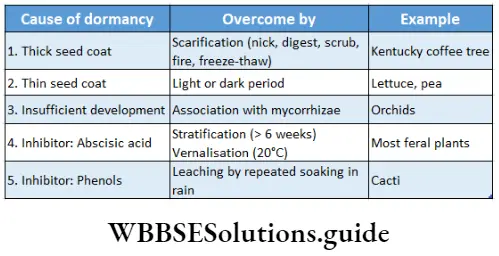
The causes of seed dormancy are discussed below—
Impermeability of seed coat to water: Water is essential for germination. If water does not enter into the seed then enzymes necessary for germination will remain inactive. Seeds of some plants of the family Solanaceae, Fabaceae, etc., bear a hard and thick seed coat through which water cannot enter the seed.
The enzymatic action of microorganisms present in the soil makes the seed coat permeable. This results in the entry of water in the seed and germination is initiated.
Seed coat impermeable to oxygen: Respiration is important for the germination of seeds. But seeds of certain plants have thick and hard seed coats through which oxygen cannot enter.
On the other hand, though certain seeds are permeable to water they are impervious to oxygen. All these conditions inhibit the process of germination.
Mechanical resistance of seed coat: The seed coat of some seeds such as Alisma plantago, Capsell sp., and Brassica sp., are so hard that the mature embryo is unable to rupture the seed coat. As a result, germination is inhibited.
Embryonic dormancy: In many cases, germination does not occur due to embryonic dormancy.
Some of them are as follows—
- Seeds of certain plants like Gingko biloba and orchids, detach from the plants with immature embryos. Germination of such seeds is naturally delayed till the embryo completes its development inside the seeds.
- Seeds of certain plants such as wheat, barley, paddy, etc., do not germinate even after sowing. Hormones play an important role in the initiation of germination in these plants.
Growth inhibitors: Certain chemical substances in plants inhibit germination. These substances are known as growth inhibitors. These substances not only inhibit growth but also inhibit phototropism. Substances such as coumarin, naringenin, etc., inhibit germination.
Effect of light: There are two types of seeds based on the effect of light on their germination
Positively photoblastic seed: The seeds of tobacco, lettuce, etc., do not germinate in the absence of sunlight. Such seeds are known as positively photoblastic seeds.
Negatively photoblastic seed: The seeds of lily, tomato, etc., do not germinate if exposed to sunlight. Such seeds are known as negatively photoblastic seeds. Effect of temperature: Germination in plants such as apples, and peaches, does not take place above or below 5°-10°C temperature.
Methods Of Breaking Seed Dormancy
Several artificial processes to break seed dormancy are discussed below.
Scarification: Any process of breaking, scratching, or mechanically altering the seed coat to make it permeable to water and gases, so that the process of seed germination can be accelerated, is known as scarification. Scarification can be of different types—
Thermal: In this process, seeds are briefly exposed to hot water. This process is also known as hot water treatment. In ‘chaparral’ plant communities (desert biome), some species’ seeds require fire or smoke to achieve germination.
Mechanical: In this process seed coats are filed with a metal file, rubbed with sandpaper, nicked with a knife, or cracked gently with a hammer to weaken the seed coat.
Chemical: Soaking seeds in sulphuric acid makes the seed coat thin and permeable. This helps to break the dormancy. Scarified seeds should not be stored and should be planted immediately. Otherwise, the seeds become non-viable.
Stratification: The second type of imposed dormancy found in seeds is internal dormancy regulated by the inner seed tissues. This dormancy prevents seeds of many species from germinating when environmental conditions are not favorable for the survival of the seedlings.
In certain cases, a brief exposure to a very low temperature is used to break seed dormancy. Stratification is a process in which the seeds are covered with Sphagnum (a moss) and kept at a temperature of 5-10°C. This process is used to break dormancy in seeds of cherry, apple, peach, etc.
Alternating temperature: Moringa (1926) first observed that a variation in the low and alternating high-temperature treatment increased the percentage of germination. The dormancy of many seeds can be broken by alternate use of high (25°C) and low (15°C) temperatures.
Exposure to light: Some plants are sensitive to light. In such plants, dormancy can be broken by using red or white light. Scientists have proved that red light stimulates germination but far-red light inhibits germination. Phytochrome affects these kinds of seed germination.
Effect of excess oxygen: According to Waring and Foda (1957), dormancy in certain plants can be broken by a high concentration of oxygen. In such cases, elevated oxygen concentration decreases the accumulation of germination inhibitors in the seed coat. Thus, it helps in breaking dormancy. This process is used in the case of Xanthium sp.
Hormones and other chemical substances: Some phytohormones such as gibberellin, ethylene, cytokinin, and chemicals such as thiourea, potassium nitrate, etc., help to break the seed dormancy.
Necessity Of Seed Dormancy
Apparently, the phenomenon of dormancy seems to be a negative process as it delays the beginning of a new life.
But, dormancy plays an important role in a plant’s life in various ways. They are—
- The beneficial aspect of dormancy helps us to solve the problem of food scarcity. Without dormancy cereal grains would have germinated thereby, losing their usefulness as a source of food.
- Dormancy allows the seeds to attain internal optimal conditions for germination.
- It helps to maintain the viability of seeds during drought and winter.
- It inhibits viviparous germination.
- It also helps the seed to find their appropriate location. Hence, it helps in the dispersal of seeds.
Photoperiodism
Definition: Photoperiodism is the phenomenon in which certain physiological changes in plants respond to the relative duration of day and night for growth and development, particularly for flowering.
Plants require a certain day length in order to initiate flowering which actually is a process of transformation from a vegetative state to a reproductive state. This phenomenon is called photoperiodism.
Photoperiodism was first described in detail by Garner and Allard (1920). Maryland Mammoth is a mutant variety of tobacco plants that grows very tall and produces very large leaves.
In nature, it blooms during winter when the day length is short. Garner and Allard (1920) performed a series of experiments on this plant by keeping it in a greenhouse and in a dark chamber.
“plant growth and development important points”
By shortening its exposure to light that would be equivalent to a winter day, this plant was compelled to bloom even in summer.
Alternatively, the plant could be kept in a vegetative state during winter months by artificially lengthening the light period. This landmark experiment proved that periods of light and darkness are highly crucial for the blooming of plants.
Types Of Plants According To The Photoperiod
Plants responsive to day-length produce flowers in a specific photoperiod. The minimum day length required by a plant for its flowering is called the critical day length of that plant.
The critical day lengths for tobacco and Xanthium are 12 hours and 15.5 hours respectively. The plant that produces flowers after exposure to light for the time period below its critical day length is called a short-day plant. On the other hand, long-day plants produce flowers when exposure to light exceeds its critical day length.
On the basis of photoperiod, we may classify the plants in the following way—
Day-neutral plants: In these plants, flowering is not influenced by the duration of the light period. There is no known day length requirement for them. Flowering in these plants is controlled by other factors like age, number of nodes, and previous history of cold treatment. E.g., most fruit crops, many vegetable crops (carrot, pea), rice, sunflower, Poa annua (bluegrass).
Long-day plants: In long-day plants flowering occurs in response to days longer than a critical length (or nights shorter than a critical length). Long-day plants may be grouped on the basis of photoperiodic response as follows—
Qualitative (absolute) long-day plants: These plants do not flower when days are shorter than critical length. They never flower at day lengths less than 12 hours, example black henbane, radish, sugar beet, hibiscus, etc.
Quantitative long-day plants: These plants flower sooner and more as day length increases, e.g., petunia, lettuce, wheat, barley, etc.
Short-day plants: Short-day plants flower in response to days shorter than a critical length (or nights longer than a critical length).
They are also categorized as follows—
Qualitative (absolute) short-day plants: These plants never flower when days are longer than their critical length, for example, tobacco, cocklebur, orchid, Chrysanthemum, Kalanchoe, Japanese morning glory, etc.
Quantitative short-day plants: They flower sooner and more as night length increases. E.g., marijuana, sugarcane, onion, blueberry, rhododendron, cotton, cosmos, etc.
Long-short-day plants (LSDP): These short-day plants produce flowers only when they are exposed to a sequence of long days followed by short days. example certain varieties of Kalanchoe (flower in late summer), Bryophyllum, Cestrum nocturnum (night-blooming jasmine), etc.
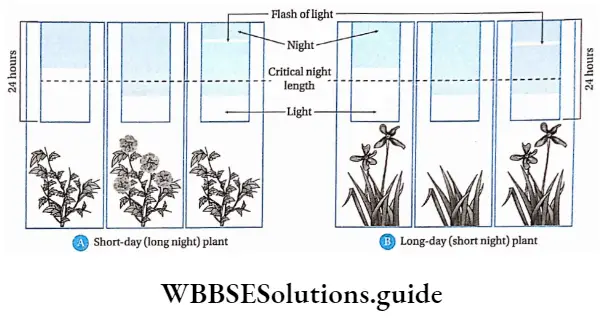
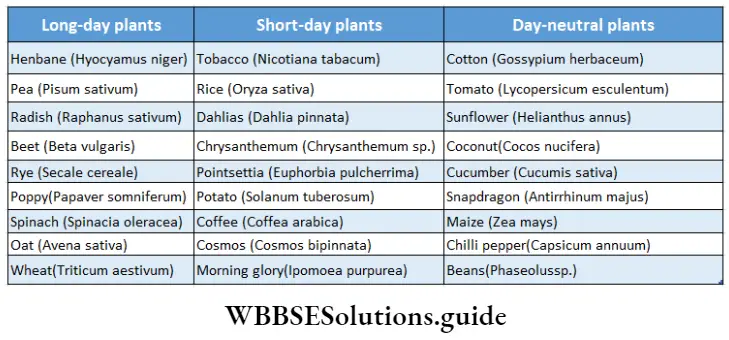
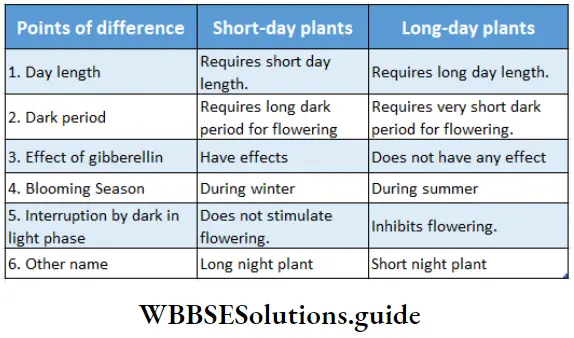
Interesting facts about photoperiodism
Dr.S.M.Sarkar and P.Parija tested the necessity of a short day for high-yielding Aman paddy. According to them, the development of flowers in aman paddy occurs under 32.2°C.
Strawberries, a short-day plant, produce more runners if exposed to long days. If yam, a long-day plant, is exposed to short days, then the plant will produce more tubers.
If long-day plant’ Henko sp., is exposed to short days, then it will show rosette formation by reducing the gap between the nodes. If an onion a short-day plant is exposed to the long day, then bulb formation occurs.
Floral clock
Many plants have an internal biological clock, which regulates the time of day when their flowers open and close. For example, the flowers of Nepeta cataria open between 6 am and 7 am; orange hawkweed opens between 7 am and 8 am; field marigolds open at 9 am and varieties of Helichrysum wake up at 10 am. Other varieties follow, with Convolvulus opening at noon.
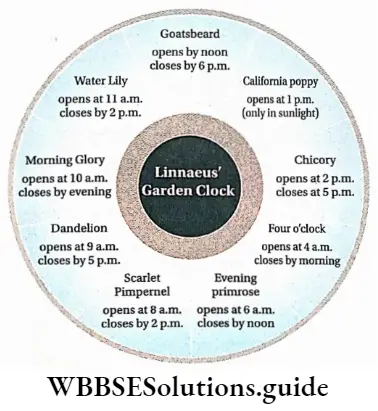
By making observations of the times when flowers open and close during the day, Carolus Linnaeus conceived the idea of arranging certain plants in an order of flowering, so that they constitute a kind of floral clock.
This was described in Linnaeus’s Philosophia Botanica (1751) in which he referred to it as a Horologium Florae (floral clock). Apparently, Linnaeus was able to use his clock to determine the time accurately within half an hour.
Characteristics Of Photoperiodism
The general characteristics of photoperiodism are—
Genetic control: Now it is well known that photoperiodic responses are controlled by genes. Nowadays, it is possible to make a plant flower in any season by using technologies (gene manipulation).
At the National Botanical Research Institute, Lucknow, scientists have bred varieties of Chrysanthemum, which are able to bloom in any season of the year including the summer. More such research is going on for different economically important plants to make them flower in any season.
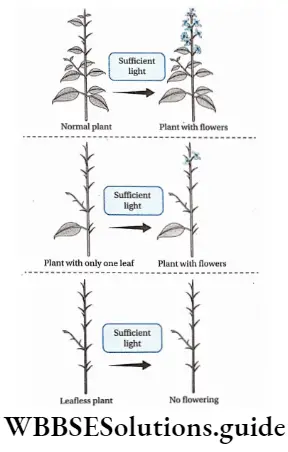
Photoperiodic induction: One complete cycle of light phase and dark phase, required during flowering, is known as an inductive cycle. 1 inductive cycle = 24 hours. This inductive cycle is species-dependent. For example, in Xanthium, only 1 inductive cycle or 24 hours are there.
There are two inductive cycles or 48 hours for Glycine max, while in Plantago lanceolate it is 25 days. Exposure to appropriate photoperiodic conditions induces flowering in both short-day and long-day plants.
The photoperiodic influence continues even if these treated plants are kept in unfavorable conditions. This phenomenon of flowering is known as photoperiodic induction.
Site of perception of photoperiodic induction: Photoperiodic induction is most effectively perceived by the leaves. Experimentally, it has been proved that flowering is possible by keeping a plant with only one leaf under a proper light source.
In a defoliated plant, flowering is not possible even if the plant gets proper day length. This is because the leaf contains the phytochrome pigment which perceives the photoperiodic induction.
Phytochrome: Phytochrome is a covalently bound, chromophore-containing protein pigment system. It is found in two interchangeable forms, which absorb red and far red light [wavelength 660-750nm].
This pigment takes part in photomorphogenesis and photoperiodism. The red and far red light of the action spectrum plays important roles in flowering.
Interchangeable forms: Depending on the variation of absorption of wavelength of light,
Two types of phytochromes are there—
- The first phytochrome Pr or P660 [P=phytochrome; r=red] forms in the absence of light and absorbs red light of wavelength 660nm.
- Pfr [fr= far-red] or P730, absorbs far red light of wavelength 730nm. Pfr is organically active. These Pr and Pfr are interchangeable. Pr changes to Pfr by absorbing light of 660nm. Again, Pfr changes to Pr by absorbing light at 730nm. Also, the Pfr slowly converts to Pr in the dark.
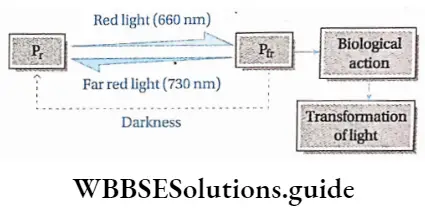
The gradual conversion of Pfr to Pr is due to the effect of far red light. This induces flowering in short-day plants. Hence, in these plants, Pr acts as a flowering stimulant.
When the Pr changes to Pfr due to the effect of red light, flowering occurs in long-day plants. In these plants, a high concentration of Pfr acts as a flowering stimulant.
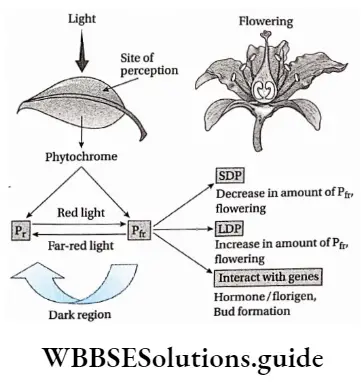
Role of florigen in flowering: According to some scientists, the stimulants for flowering are found in leaves. the stimulants (hormone-like chemical substances) are first produced in leaves and then gradually move to the flower-forming regions (axial and lateral buds).
Chailakhyan (1937) named this stimulant florigen. According to some scientists, this florigen along with gibberellin and anthesin helps in flowering.
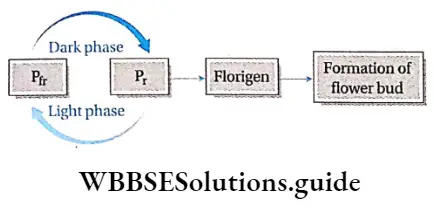

Importance Of Photoperiodism
- Photoperiodism helps in determining the flowering time for economically important plants, especially commercially grown garden plants. Farmers can induce or delay flowering by using this phenomenon according to their needs.
- Some vegetable plants are allowed to continue their phase of vegetative growth by delaying the time of flowering. This induces a higher yield of tubers and rhizomes.
- The phenomenon can be utilized to produce good-quality breeds by the process of hybridization.
- Photoperiodism can also be used to produce more fruits and flowers by altering the flowering time and vegetative growth period.
- Endangered species of plants can be saved by using photoperiodism. Photoperiodism increases the power of adaptability as well as acclimatization of plants. This results in better dispersal of plant groups.
- This phenomenon is also useful for planning crop patterns and gardening in a particular region.
- By controlling day length, flowering can be induced in different varieties of the same species at a time. It helps in cross-pollination between all the varieties at the same time.
Vernalisation
Definition: The physiological process by which flowering is promoted in plants through prolonged exposure to low temperatures is called vernalization.
Plants have evolved a range of strategies so that they can bloom during the most suitable time of the year. In some plants, vernalization is a key requirement of the reproductive strategy. It permits plants to prepare for flowering as winter sets in and enables them to bloom during spring.
The word ‘vernalization’ was coined by Lysenko (1928), although Klippart and Gassner (1918) were the first to demonstrate vernalization. The origin of this word is from the Latin word—vernal, meaning ‘of spring’. The required temperature for vernalization ranges from 1°C to 9°C in plants.
Site of vernalization: In intact plants, the tip of the germinating embryo, root apex, shoot apex and the growing region of leaf lamina are the sites of vernalization. In some cases, other mitotically active tissues can become vernalised. G. Melchers reported that a flowering hormone called vernalin is formed in meristems as a result of vernalization.
There are some specific sites of vernalization in different plant species. Such as—
- In the case of Hyoscyamus niger (henbane), vernalization occurs in the shoot apex.
- In the case of Streptocarpus wendlandii, vernalization occurs in leaves.
Examples of vernalization: Generally, two types of henbane plants are there. One is annual and another is biannual. Both are long-day plants. If they are kept under short-day conditions then they will grow vegetatively and will not bloom.
It has been proved that those plants will bloom if treated with low temperatures. Flowering can be induced in other plants such as beetroot, mustard (black), etc., by this process. Scientists proved that if the winter variety of some seasonal plants is kept at 0°C-5°C for some weeks, then they will bloom in any season.
Importances of vernalisation:
- Vernalisation reduces the period of vegetative growth in the plants.
- Vernalisation promotes early flowering in plants.
- It increases the cold resistance of the plants.
- It reduces the time span between germination and flowering and so, helps to increase crop yield.
- Plants may develop fungal resistance due to vernalization.
- For cereal crops, early harvesting can be done before the drought season because of vernalization.
Essential Conditionsfor Vernalisation
Essential conditions for vernalization are given below—
Low temperature: 0°C-7°C is essential for vernalization. It has been found that vernalization is not possible below— 4°C and above 12°C-14°C.
Age: In cereals, the sprouting seeds act as the site of vernalization. Dry seeds do not show vernalization.
Water: An appropriate amount of water is important for the process of vernalization.
Light: Some seeds require light for vernalization.
Nutrient: In culture medium proper amount of nutrients especially carbohydrates, is needed for the embryo to be vernalised.
Period of low temperature: The duration of treatment with low temperature is different for different plants. It can be 1-3 months for different plants. In some plants like celery, as little as 8 days of cold can cause a substantial promotion of flowering. However, greater than 1 month of cold treatment is required for maximal cases of vernalization.
Oxygen: Vernalisation requires metabolic energy. So, oxygen is important for this process. According to Von Denffer (1950), different inhibitors of flowering are formed under anaerobic conditions even at low temperatures. So, the oxygenated condition is very important for vernalization, otherwise, it fails in the absence of oxygen.
“plant growth and development notes PDF download”
Active cell division: Germinating seeds and active meristem show active cell division. Active cell division requires active metabolic activity. This is one of the most important criteria of vernalization.
Sensitive parts: The leaves are sensitive to vernalization. Leaves get sensitized and secrete hormones that induce flowering.

Process Of Vernalisation
- According to scientists, the application of low temperatures is required for the development of plants. However, vernalization is affected by many hormones.
- At low temperatures, gibberellic acid (GA) and vernalin (postulated hormone) are secreted.
- These hormones in turn activate certain genes called AGAMOUS-LIKE 20 in some plants like Arabidopsis thaliana.
- Some scientists also thought that vernalin helps to activate another hormone called florigen (postulated hormone). According to them, florigen is the actual hormone responsible for flowering.
- Vernalin and gibberellin both act differently, but gibberellin helps in the production and functioning of vernalin. The process of vernalization is depicted in the following chart.

Plant Growth And Development Notes
- Apical dominance: Inhibition of growth of lateral buds while apical buds are present.
- Bolting: Sudden elongation of a condensed part of the stem before flower initiation.
- Diterpenoid: An organic compound made up of four isoprene (5C) units.
- Elicitors: Compounds of endogenous or exogenous origin that activate chemical defense mechanisms in plants.
- Enantiomer: Each of a pair of molecules that are mirror images of each other.
- Endangered species: A species of animal or plant that is at the risk of extinction.
- Etiolation: Yellowing of green parts of the plant in the absence of sunlight.
- Geotropic: Movement towards gravity or soil.
- Indole ring: A cyclic ring structure (of different heterocyclic organic compounds) with formula C8H7N
- Racemic mixture: A mixture of equal amounts of left-handed and right-handed enantiomers of a chiral molecule.
- Synergistic effect: The overall effect created by more than one chemical or biological substance that is greater than the sum of individual effects of any of them.
- Terpene: A class of monocyclic hydrocarbons of the formula C10H16.
- Thigmomorphogenesis: The response by plants to mechanical sensations such as touch, wind, raindrops, etc., by altering their growth patterns.
Points To Remember
- The process by which cells derived from the root and shoot apical meristems and cambium change into permanent tissue during the development of a plant cell to serve a specific function is known as, differentiation.
- A high rate of anabolism compared to catabolism induces growth.
- Cellular growth is characterized by cell division, cell elongation, and cell differentiation.
- The growth curve is the graphical representation of growth with respect to time.
- Plants continue to grow throughout their life, so their growth is known as indefinite or unlimited growth.
- On the basis of nature, there are three types of growth—vegetative or somatic growth, regenerative growth, and reproductive growth.
- Growth is mainly divided into four phases
- Lag phase,
- Log phase,
- Decreasing phase,
- Stationary phase.
- A study related to growth is known as auxanology
- The formation of new organs or body parts in living organisms by the process of differentiation is known as morphogenesis.
- The concentric rings found in the cross sections of plants with woody stems are known as annual rings or growth rings. The age of a tree can be determined by these annual rings.
- Arc indicators, auxanometers, and horizontal microscopes are the instruments used for measuring plant growth.
- The structural and functional deterioration occurring in the living body naturally is known as senescence.
- Blastema is the cells capable of growth and regeneration to produce buds in the place of any cut and wound.
- The minimum day length required to induce flowering in a plant is known as the critical day length of that plant.
- The phenomenon in which plants respond to the relative duration of day and night for growth and development, particularly for flowering is known as photoperiodism.
- In most of the long-day plants, flowering is induced by gibberellin.
- The red and far-red light of the action spectrum plays important roles in the flowering of short-day and long-day plants.
- Phytochromes are chromoprotein-like pigments formed of pyrol chains.
- The formation of new plant parts by the process of tissue culture is known as regeneration.
- Totipotency is the ability of a single cell to divide and produce all of the differentiated cells in an organism. Spores and zygotes are examples of totipotent cells.
- Ethylene helps in abscission, whereas auxin and cytokinin prevent abscission.
- Generally, long-day plants bloom during summer and short-day plants bloom during winter.
- The application of photoperiodism can be used to induce flowering in annual plants in any season of the year.