Body Fluids And Circulation Multiple Choice Question
Question 1. Adult human RBCs are enucleated. Which of the following statement(s) is/are the most appropriate explanation for this feature?
- They do not need to reproduce
- They are somatic cells
- They do not metabolize
- All their internal space is available for oxygen transport
Choose the correct answer
- Only 1
- 1, 3 and 4
- 2 and 3
- Only 4
Answer: 4. only 4
Read and Learn More WBCHSE Multiple Choice Question and Answers for Class 11 Biology
Question 2. The hepatic portal vein drains blood to the liver from—
- Stomach
- Kidneys
- Intestine
- Heart
Answer: 3. Intestine
Body fluids and circulation multiple choice questions with answers PDF
Question 3. MALT constitutes about per cent of the lymphoid tissue in the human body.
- 20%
- 70%
- 10%
- 50%
Answer: 4. 50%
| Class 11 Biology | Class 11 Chemistry |
| Class 11 Chemistry | Class 11 Physics |
| Class 11 Biology MCQs | Class 11 Physics MCQs |
| Class 11 Biology | Class 11 Physics Notes |
Question 4. Frog’s heart when taken out of the body continues to beat for some time Select the best option from the following statements.
- Frog is a poikilotherm
- The frog does not have any coronary circulation
- The heart is ‘myogenic’ in nature
- Heart is autoexcitable
Choose the correct answer
- Only 4
- 1 and 2
- 3 and 4
- Only 3
Answer: 3. 3 and 4

Question 5. Reduction in pH of blood will—
- Reduce the blood supply to the brain
- Decrease the affinity of haemoglobin with oxygen
- Release of bicarbonate ions by the liver
- Reduce the rate of heartbeat
Answer: 2. Decrease the affinity of haemoglobin with oxygen
Question 6. In mammals, which blood vessel would normally carry the largest amount of urea?
- Dorsal Aorta
- Hepatic Vein
- Hepatic portal vein
- QRS complex
Answer: 2. Hepatic Vein
Question 7. Name the blood cells, whose reduction in number can cause clotting disorder, leading to excessive loss of blood from the body.
- Erythrocytes
- Leucocytes
- Neutrophils
- Thrombocytes
Answer: 4. Thrombocytes
MCQ on body fluids and circulation for NEET with answers
Question 8. Serum differs from blood in—
- Lacking globulins
- Lacking albumins
- Lacking clotting factors
- Lacking antibodies
Answer: 3. Lacking clotting factors
Question 9. Which one of the following animals has two separate circulatory pathways?
- Shark
- Frog
- Lizard
- Whale
Answer: 4. Whale
Question 10. Doctors use stethoscopes to hear the sound; produced during each cardiac cycle. The second sound is heard when—
- The AV node receives a signal from SA node
- AV valves open up
- Ventricular walls vibrate due to the gushing of blood from the atria
- Semilunar valves close down after the blood flows into vessels from the ventricles
Answer: 4. Semilunar valves close down after the blood flows into vessels from the ventricles
Question 11. The person with blood group AB is considered a universal recipient because he has—
- Both A and B antigens on RBC, but no antibodies
- Both A and B antibodies in the plasma
- No antigen on RBC and no antibodies in the plasma
- Both A and B antigens in the plasma, but no antibodies
Answer: 1. Both A and B antigens on RBC, but no antibodies
Question 12. Thrombokinase is associated with—
- Elimination of urea and other excretory products from the body
- Production of erythrocytes from the bone marrow
- Pulmonary and systemic circulation
- Cardiac cycle and its regulation
- Enzymatic reactions in coagulations of blood
Answer: 5. Enzymatic reactions in coagulations of blood
Question 13. Identify the correct statement regarding cardiac activity—
- Normal activities of the human heart are regulated intrinsically, hence it is neurogenic
- A special neural centre in the medulla oblongata can moderate cardiac function through the CNS
- Parasympathetic neural signals increase the rate of heartbeat
- Adrenal medullary hormones can increase cardiac output
- The end of a T-wave marks the end of diastole
Answer: 4. The end of a T-wave marks the end of diastole
Class 11 biology body fluids and circulation MCQ with solutions
Question 14. Choose the wrong statement regarding the circulatory system of frogs—
- Sinus venosus receives blood through major veins called vena cava
- The ventricle opens into a sac-like conus arteriosus
- The erythrocytes are nucleated
- A special venous connection between the liver and intestine called the renal portal system is present
- The lymphatic system consists of lymph, lymph channels and lymph nodes
Answer: 4. Lymphatic system consists of lymph, lymph channels and lymph nodes
Question 15. The depolarization of the atria is represented by—
- P wave
- Q. wave
- T wave
- QRS complex
Answer: 1. P wave
Question 16. Which diagram is correct for the circulation of blood through the human heart?
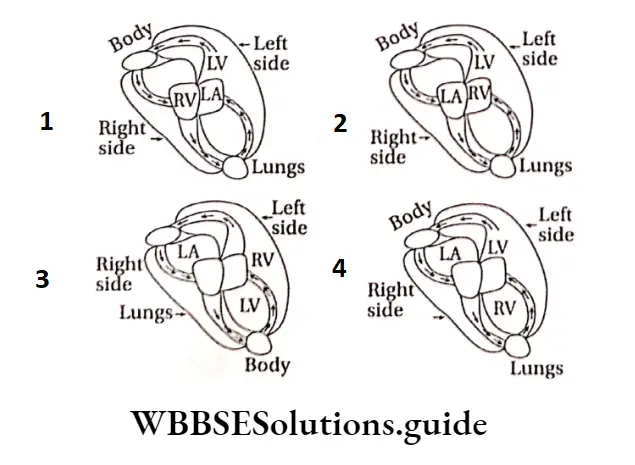
Answer: 1. 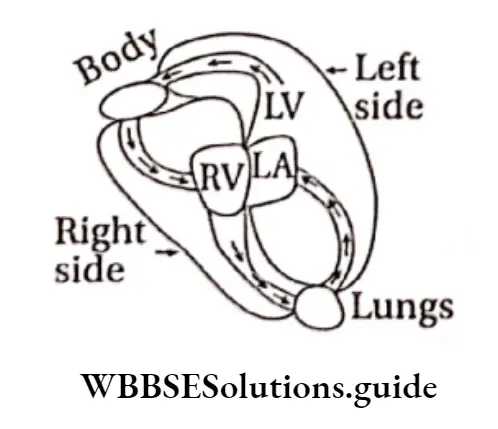
Question 17. Which option is correct for the formation of intrinsic factor-X activator complex for blood coagulation?
- Activated Christmas factor + AHG + Phospholipid + ca2+
- Inactivated Christmas factor + AHG + phospholipid + Ca2+
- Converting + AHG + ca2+ FSF
- Phospholipid- protein complex + Proconvertin
Answer: 1. Activated Christmas factor + AHG + Phospholipid + ca2+
Question 18. An increase in blood pressure leads to
- Hypotension
- Bradycardia
- Both 1 and 2
- Hypertension
Answer: 4. Hypertension
Question 19. Red cell count is carried out by—
- Haemocytometer
- Haemoglobinometer
- Sphygmomanometer
- Electrocardiogram
Answer: 1. Haemocytometer
Question 20. Which of the following matches correctly?
- Inferior vena cava—Receives deoxygenated blood from the head and body
- Superior vena cava—Receives deoxygenated blood from the lower body and organs
- Pulmonary artery—Carries deoxygenated blood to the lungs
- Hepatic artery—Carries deoxygenated blood to the gut
Answer: 3. Pulmonary artery—Carries deoxygenated blood to the lungs
Question 21. The main function of the white blood cell in the human immunological system is to—
- Combat and destroy antigenic particles
- Produce antigens to combat antibodies
- Carry oxygen around the body
- Transport antigens to B memory cells in the lymph nodes
Answer: 1. Combat and destroy antigenic particles
Question 22. In which, blood circulation starts and ends in capillaries?
- Portal system
- Arterial system
- Capillary system
- Lymphatic system
Answer: 1. Portal system
Important MCQs on body fluids and circulation for competitive exams
Question 23. The diagram given here is the standard ECG of a person. The P-wave represents the—

- Contraction of both the atria
- Initiation of the ventricular contraction
- Beginning of the system
- End of systole
Answer: 1. Contraction of both the atria
Question 24. Arteries that carry deoxygenated blood are—
- Pulmonary arteries
- Carotid Arties
- Coronary Arteries
- Phrenic Arteries
Answer: 1. Pulmonary arteries
Question 25. Blood leaving the liver and going towards the heart is rich in—
- Pulmonary Arteries
- Urea
- Ammonia
- Oxygen
Answer: 2. Urea
Question 26. The following Are The Branches Of Dorsal Aorta
- Intercostal
- Phrenic
- Colieac
- Anterior Mesenteric
Choose the correct Answers
- 1 and 2
- 2 and 4
- 4 and 5
- 2 and 3
Answer: 2. Phrenic
Question 27. The Leucocytes Contain Which Of The Following Large Quantity?
- Basophils
- Neutrophils
- Eosinophils
- Monocytes
Answer: 2. Neutrophils
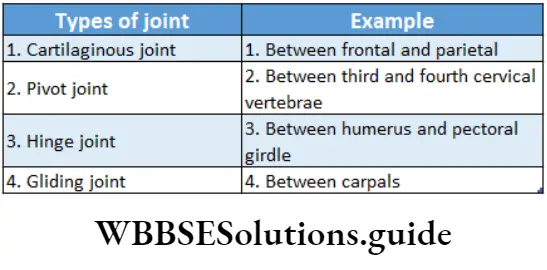
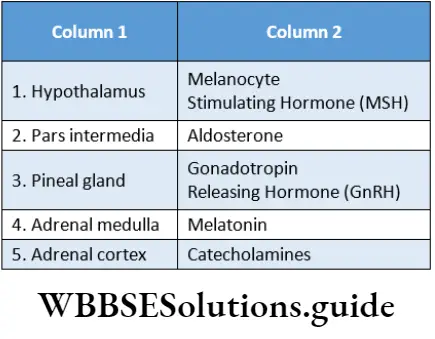
Question 28. Match The Column
- 1-3,2-1,3-2,4-4
- 1-2,2-3,3-1,4-4
- 1-1,2-2,3-3,4-4
- 1-4,2-3,3-2,4-1
Answer: 1. 1-3,2-1,3-2,4-4
Body fluids and circulation chapter MCQ with explanation
Question 29. WBC is true Cell Due To
- Absence of nucleus
- Devoid Of Haemoglobin
- Absence Of Cell Wall
- Presence Of Nucleus
Answer: 4. Presence Of Nucleus
Question 30. If The Systolic Pressure is 120mm Hg Diastolic Pressure Is 80mm hf The Pulse pressure is-
- 120-80=40 mm Hg
- 120/80=1.5 mm Hg
- 120×80 =9600 mm Hg
- 120+80 = 200 mm Hg
Answer: 1. 120-80=40 mm Hg