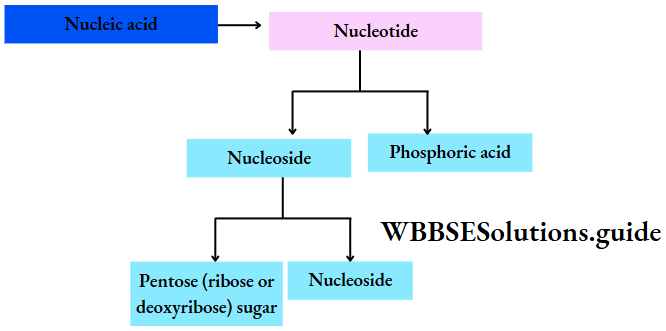
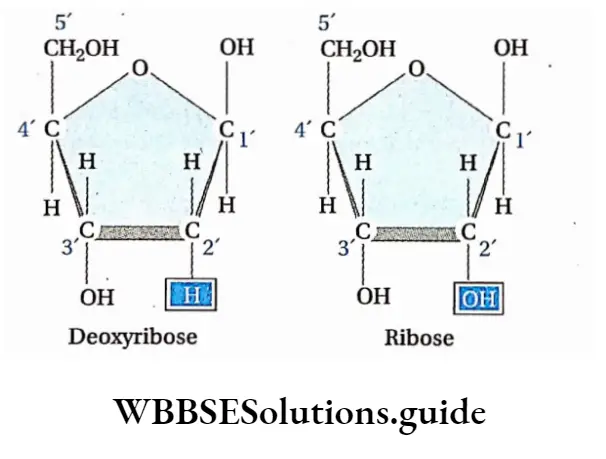
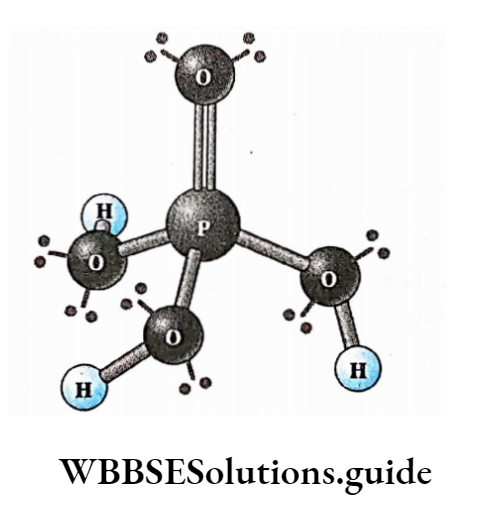
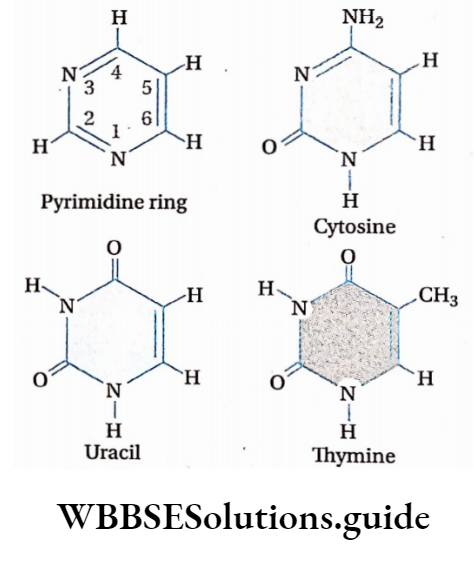
Biomolecules Introduction
Biomolecules
The body of an organism is just like a chemical factory. Numerous chemical reactions are continuously occurring inside our body.
Such reactions constitute the metabolic pathways of the various biochemical processes like respiration, photosynthesis, etc.
These reactions involve different molecules and enzymes. Many of these molecules such as carbohydrates, proteins, and lipids are the major constituents of the living body.
The collection or sum total of all these molecules present within a cell is called the cellular pool. All such molecules that are involved in the maintenance and metabolic processes of the living body are called biomolecules.
Different molecules found in a living body—Biomolecules
Biomolecules occur naturally in living organisms. All forms of life have biomolecules as their constituents.
Biomolecules consist mainly of carbon and hydrogen with nitrogen, oxygen, sulfur, and phosphorus.
The biomolecules are divided into two types—
- Inorganic and Organic.
- Inorganic molecules
These molecules either contain fewer carbon and hydrogen atoms or none at all.
They remain dissolved in the cellular fluid and form a colloidal solution, For example, different minerals (in the form of ions and salts), water, gaseous products, etc.
Biomolecules notes for NEET PDF
Inorganic molecules are of two types—
- Water And
- Inorganic Salts.
Water: About 90% of the cellular protoplasm is made up of water.

Water is required for the following functions—
- Solvent: Water is used as a solvent in different organic and inorganic cellular components.
- Medium for metabolic reactions: Several metabolic reactions take place in aqueous medium.
- Temperature regulation: Water plays an important role in regulating the body temperature mainly in higher animals and also in plants.
- pH regulation: The balance between the acidic and basic components in the body is maintained by water.
Inorganic salts: Inorganic salts can carry out the following functions—
- Structure of body parts: Different body parts such as bones, teeth, etc., have inorganic salts as one of the major components.
- Chemical reactions: Inorganic salts also take part in different chemical reactions within the body.
- Formation of organic molecules: Organic biomolecules such as DNA, RNA, etc., have inorganic salts as one of the major components.
Organic molecules
In these molecules, the number of carbon and hydrogen atoms is more compared to inorganic molecules.
Different sugars, lipids, amino acids, proteins, nucleic acids, etc., fall under this group.
They are divided into two types—
- Macromolecules And
- Macromolecules.
Micromolecules: The molecules, that have a simple structure, low molecular weight, and are mostly water soluble, are called micromolecules.
Class 11 biology biomolecules notes with diagrams
Examples: Monosaccharides, amino acids, nucleotides, etc.
Macromolecules: The larger molecules that are made up of smaller monomeric units (micromolecules), linked by chemical bonds, through polymerization reaction, are called macromolecules.
Examples: Proteins (polymer of amino acids), nucleic acids (polymer of nucleotides) etc.

Different Essential Elements Of —Living Bodies
Among elements, about 22 are essential for the body.
According to their requirement, the elements are divided into two categories—
- Macroelements And
- Microelements.
Macroelements
These elements are required by the body in large amounts. Also, deficiency of any of them may inhibit the growth and physiological functioning of the body.
About 95% of the cellular components are made of these elements. In our body, oxygen, carbon, hydrogen, nitrogen, etc., are examples of macroelements.
Biomolecules Notes Significant Biomolecules
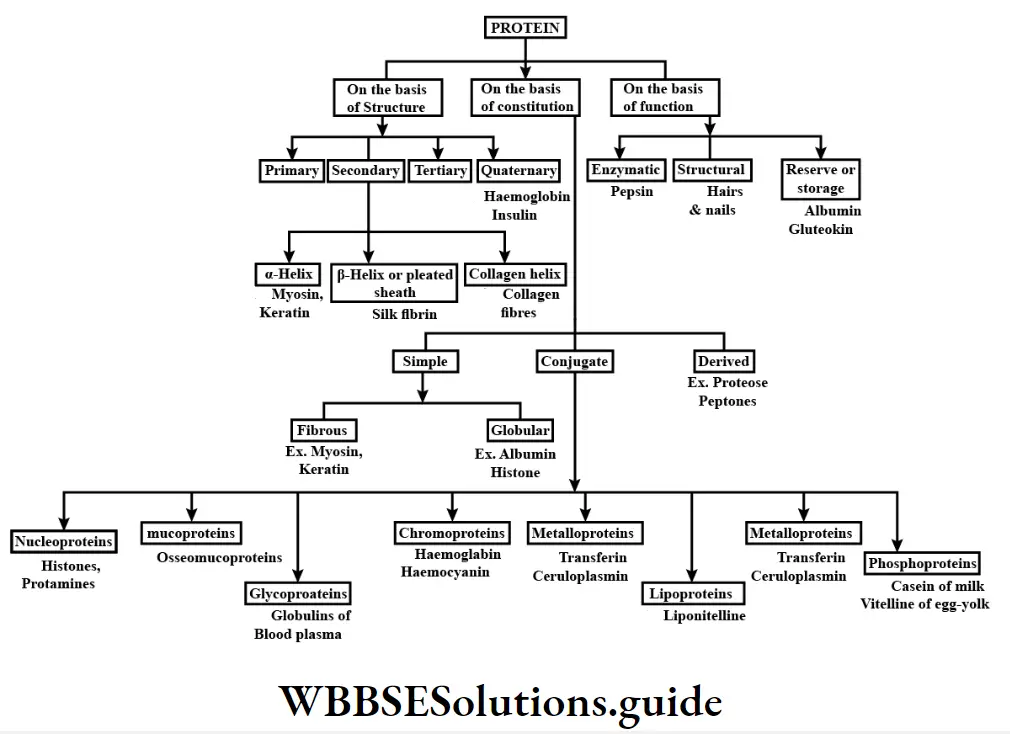
Carbohydrates, lipids, proteins, and nucleic acids are four main macromolecules.
Carbohydrates
Carbohydrates Definition: Carbohydrates are polyhydroxy aldoses, ketoses, and their derivatives that yield- such compounds in hydrolysis where the ratio of carbon to hydrogen to oxygen atom is 1:2:1.
General formula: Cn(H20)n
Nomenclature: French term ‘Hydrate de carbone’, gives rise to the word ’carbohydrates1.
It is a class of compound called ‘saccharide’ derived from the Greek word ‘saccharin’ meaning sugar.
Sources: The natural sources of carbohydrates are plants—grapes, mango, sugarcane, beet, potato, maize, wheat, rice, vegetables, etc.; animals—milk, liver, fish, meat, etc.
Classification of carbohydrates: All carbohydrates are classified into three basic types— monosaccharide, oligosaccharide, and polysaccharide.
Biomolecules Chart
More facts on carbohydrates
Free fructose is not generally available in the body but it is available in free form only in semen. Levulose is present in honey.
In some carbon compounds hydrogen and oxygen may be present in a 2:1 ratio but they are not considered carbohydrates. For example, acetic acid (CH3C00H) etc
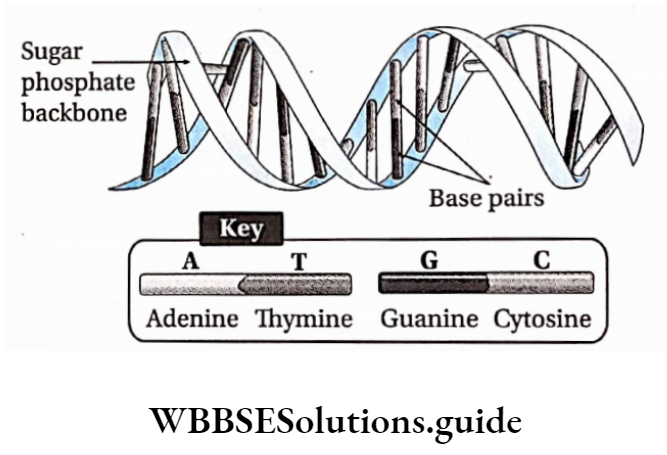
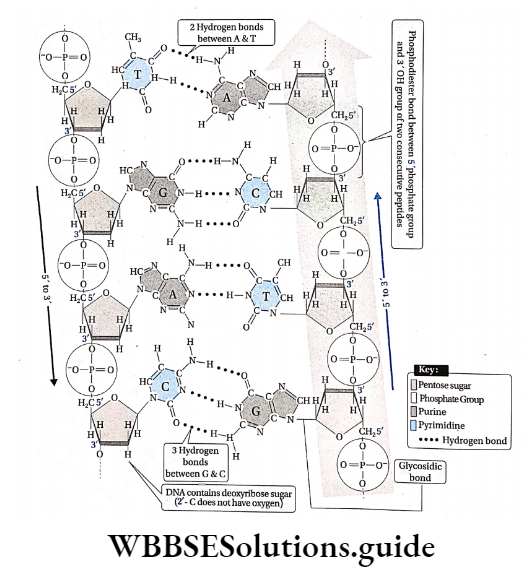
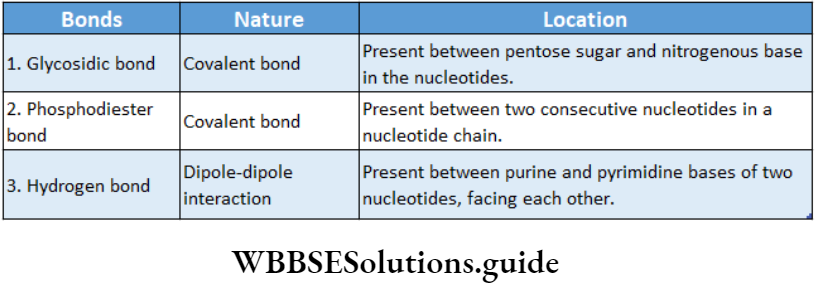
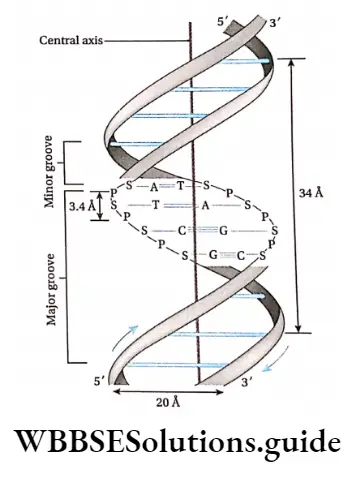
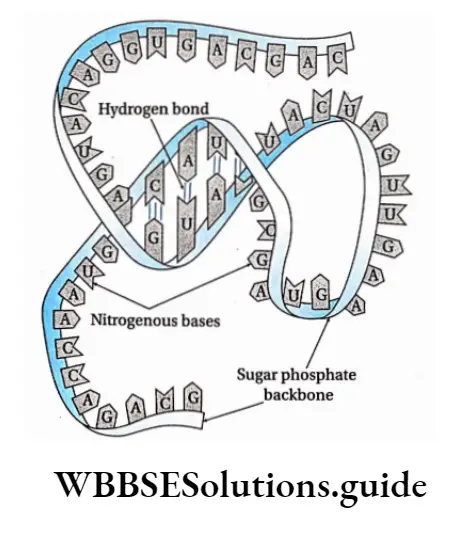
In a DNA molecule, the two intertwined strands are not parallel, i.e., antiparallel. One strand is aligned 5′-3′ while the other is aligned 3′-5′.
The diameter of the DNA helix is about 20A.
In this structure, the helix makes a turn at every 3.4 nm length of DNA, and the distance between two neighboring base pairs is 0.34 nm. Hence, there are about 10 base pairs per turn.
The intertwined strands make two grooves of different widths, referred to as the major groove and the minor groove, which may facilitate binding with specific proteins.
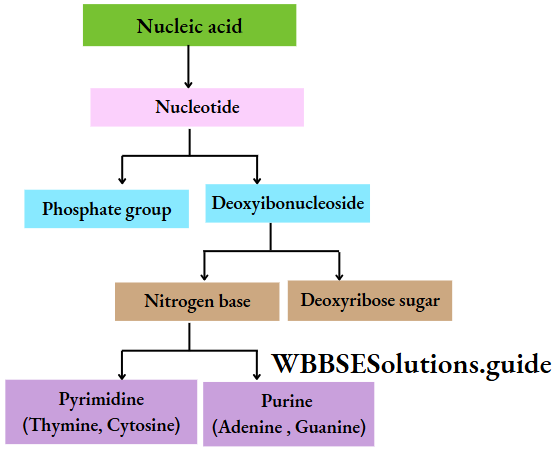
Each DNA molecule has a sugar-phosphate backbone, with nitrogenous bases attached to them.
The nitrogenous bases of one DNA molecule form a hydrogen bond with the adjacent base belonging to the other DNA molecule.
A binds with T with a double bond & G binds with C with a triple bond. This keeps the helical structure of the DNA stable, with its diameter constant.
Biomolecules Notes
In a single strand of DNA, the amount of A is not equal to that of T, while the amount of G is not equal to that of C. On the other hand, in the case of double-stranded, A + G = T + C.
Types of DNA: Most organisms have regular double-helical forms of DNA. Some bacteriophages and animal viruses have single-stranded DNA.
In prokaryotic organisms (bacteria, etc.), the DNA molecule is not linear, rather it is circular.
DNA can be classified according to the following characters—
- The number of nucleotides in each turn,
- The bond angle between the nitrogenous base pairs,
- The diameter of dna double helix,
- The direction often of double helix structure, i.e., Right or left.
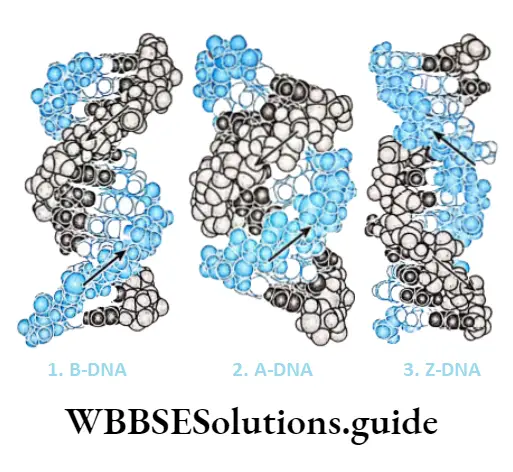
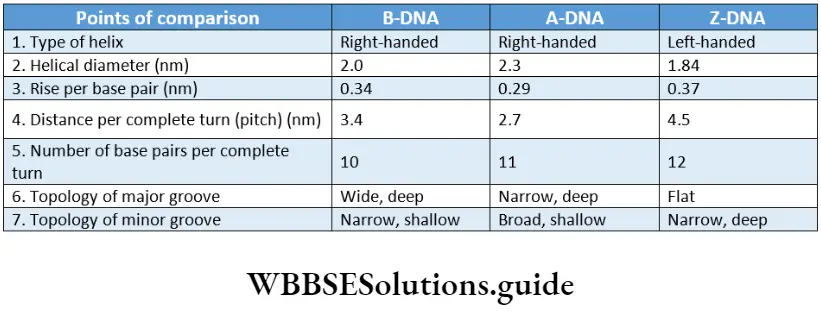
There are three major forms of DNA helices—A-DNA, B-DNA, and Z-DNA. The structure of B-DNA was proposed by Watson and Crick, which has been discussed in detail in this chapter.
The comparisons between the three forms of DNA.
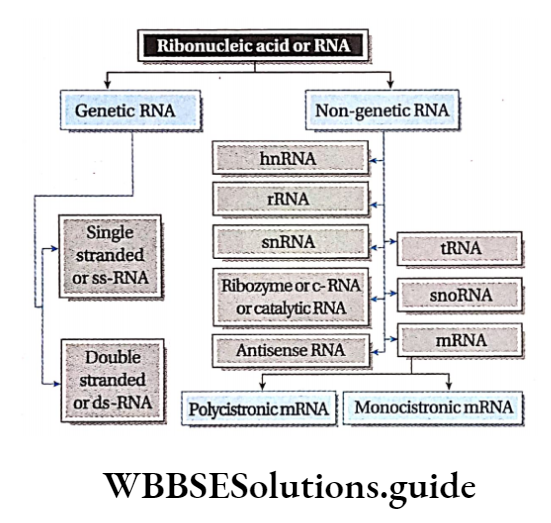
Ribonucleic acid (RNA): RNA is a type of nucleic acid that is comprised of a single-stranded polynucleotide chain and responsible for protein synthesis.
Occurrence: The non-genetic RNA is present in all prokaryotic and eukaryotic cells. It occurs both in the nucleus as well as in the cytoplasm.
In some viruses, like TMV, influenza virus, retrovirus (HIV), etc., RNA acts as a genetic material and is known as ‘Genetic RNA’.
Quantity: The quantity of RNA depends on the metabolic reactions of the cell. During protein synthesis, the amount of RNA increases in cells.
Structural features:
Cellular RNA, both genetic and non-genetic, is single-stranded and may fold upon itself to form hairpin-like structures entirely or at certain.
- Regions Rheovirus has double-stranded genetic RNA.
- The strand is unbranched but may be folded in some cases so that the structure is complex.
- They are made up of several nucleotides linked by 3′-5′ phosphodiester bonds.
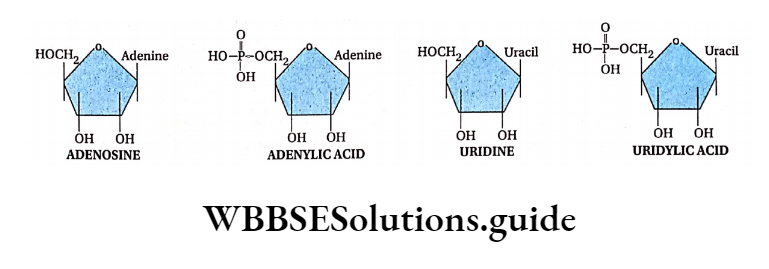
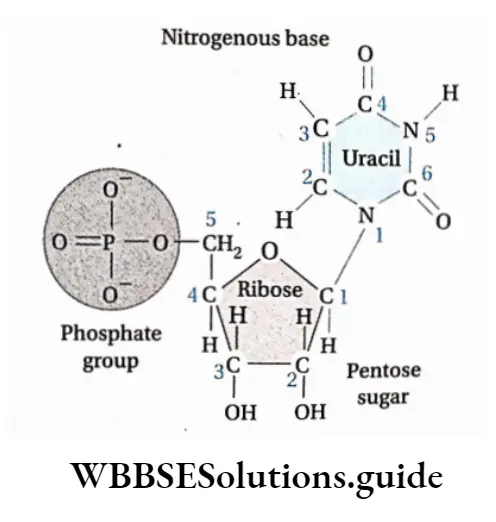
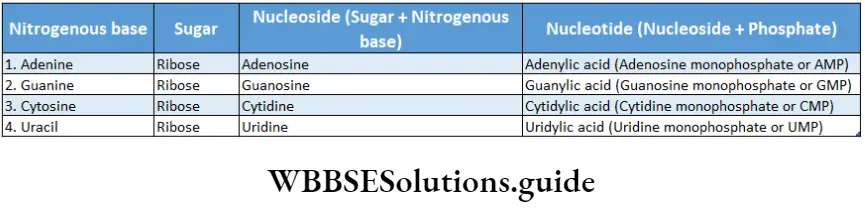
- Every nucleotide is made up of a ribose sugar, a nitrogenous base, and a phosphate. The nucleotides are called ribonucleotides.
- The nitrogenous bases present in RNA are adenine, guanine, cytosine, and uracil.
- The main function of RNA is protein synthesis. It is the main genetic material in many viruses.
- RNA is of different types. They are discussed under separate heads below.
Types of RNA: The different types of RNA have been discussed below.
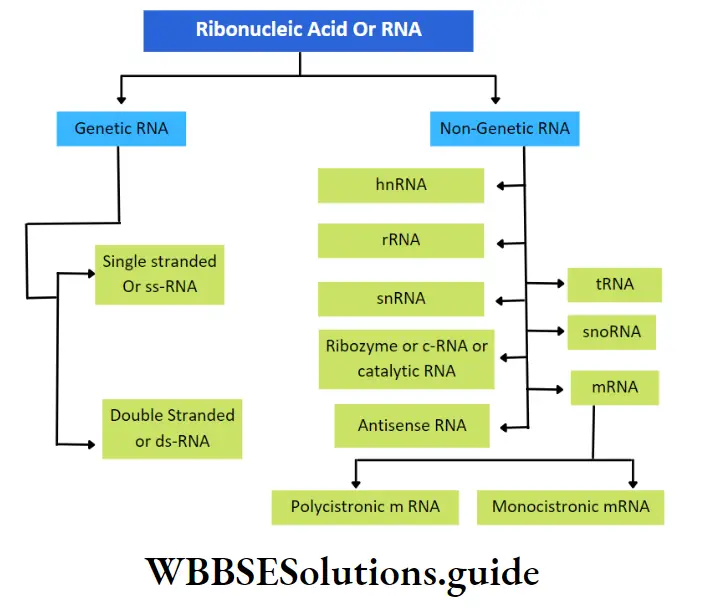
Genetic RNA: The RNA that acts as genetic material in the absence of DNA, is called genetic RNA.
For example, Tobacco Mosaic viruses (TMV), wound tumor viruses, etc. contain genetic RNA.
This type of RNA is again of two types—
Single-stranded RNA: The RNA molecule made up of a single strand is called single-stranded RNA. E.g., TMV (plant virus), influenza (animal virus).
Double-stranded RNA: The RNA molecule made up of two strands is called double-stranded RNA. E.g., Wound tumor virus (plant virus), Rheovirus (animal virus), etc.
Non-genetic RNA: The RNA presenting the prokaryotic and eukaryotic cells, where DNA is the main genetic material, is called non-genetic RNA. These are synthesized from DNA by transcription.
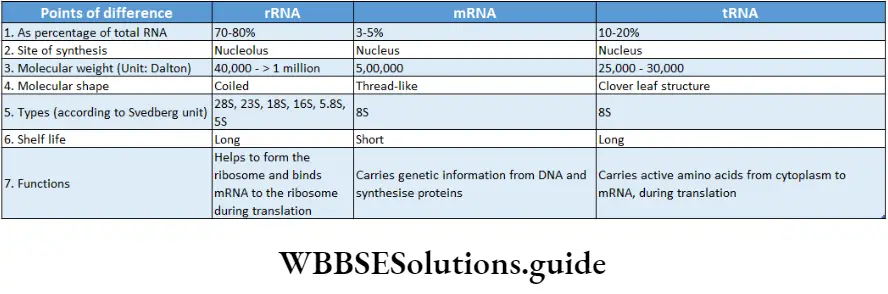
In both prokaryotic and eukaryotic cells, three principal types of RNA are found. They are messenger RNA (mRNA), transfer RNA (tRNA), and ribosomal RNA (rRNA).
Genetic material: DNA is the biological molecule that stores all the genetic information of the cell. In some viruses, RNA may function as the molecule that stores genetic information.
Transmission of hereditary characters: DNA functions as the molecule, that carries the genetic information from parents to offspring.
Synthesis of protein: Coded genetic information is transferred to the mRNA from DNA by the process of transcription. These codes are used to synthesize proteins by translation with the help of rRNA and tRNA.
Thus, information in DNA or genes is expressed in the form of proteins. Messenger RNA (mRNA) is the nucleic acid that carries information from the nucleus to the cytoplasm on which proteins are made.
Biomolecules Notes
The rRNA is a constituent of ribosome. This type of RNA helps in binding mRNA to ribosome and also in decoding the codons.
They also help in catalyzation of amino acid assembly into a polypeptide chain. The tRNA transports amino acids to the growing end of a polypeptide chain at ribosomes.
Role in metabolic activity: DNA indirectly controls the metabolic activity of the cell through the synthesis of necessary protein molecules, like enzymes.
Role In gene mutation and variation: Changes in the sequences of base pairs of DNA alter the arrangement of nucleotides in mRNA.
This causes alterations of codons which lead to changes in proteins. This induces variations in organisms leading to mutations. Mutation when leads to evolution can give rise to new species.
Biomolecules Class 11 Notes
Concept Of Metabolism
Definition: The sum total of all chemical reactions constantly occurring in the living organism, that are necessary for maintaining the living state of cells and that of the organism, is termed as metabolism.
The metabolic reactions take place as a series of linked reactions. These multi-step processes are called metabolic pathways.
Metabolic pathways of a living system can be categorized into two types—
- Anabolic pathways and
- Catabolic pathways.
Anabolic pathways
These pathways are constructive in nature and result in the formation of complex structures from simpler ones. Anabolic pathways consume energy and hence, are endergonic in nature.
For example, amino acids are linked together to synthesize proteins. Protein synthesis requires energy input and is an example of the anabolic pathway.
Catabolic pathways
These pathways are destructive in nature and lead to the breakdown of complex substances into simpler ones.
Catabolic pathways are made up of exergonic reactions because they release energy.
For example, glycolysis is a catabolic pathway where glucose breaks down to produce two molecules of pyruvic acids.
The energy released during catabolic pathways is stored in the form of chemical energy in the high-energy bonds of ATP (Adenosine triphosphate) molecules.
ATP acts as a link between exergonic and endergonic reactions. This is because ATP is hydrolyzed to release the chemical energy needed for various endergonic (energy-consuming) reactions.
Living Stage
A living system exists in a steady state, characterized by particular concentrations of all the biomolecules present within the living system.
The steady state is in a non-equilibrium state because the living system is working continuously. It prevents itself from reaching the state of equilibrium so that it is able to perform work.
The rate of formation, utilization, and transformation of biomolecules is called turnover. The rate of turnover of molecules in a metabolic pathway is called metabolic; flux.
For being in the state of metabolic flux, the metabolites tend to reach equilibrium. The equilibrium state is not reached because the end products of the metabolic pathways are utilized again.
Thus, the living organism tends to attain a steady state by metabolic flux. This takes place in the presence of energy, which is produced as a result of different metabolic reactions.
So, we can conclude that, without metabolism, a living state cannot exist.
According to Lehninger and others, biomolecules are normally present as essential organic components of living systems, and their essential organic compounds are collectively called metabolites.
These are mainly of two types—
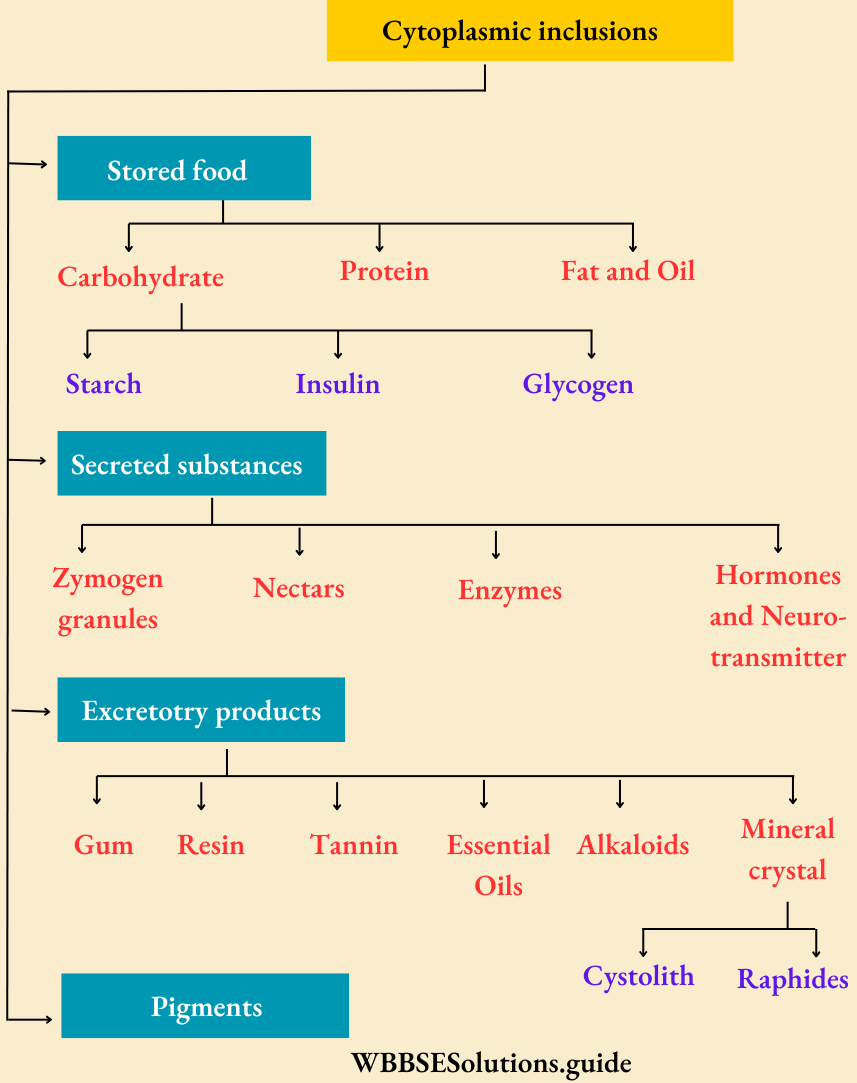
Primary metabolites: They are formed as intermediates and produced via normal metabolic pathways in almost all living organisms, e.g., sugar, amino acids, nucleotides, lipids, proteins, etc.
Secondary metabolites: They are formed especially by the alteration of primary metabolites, mainly in the plant’s body, e.g., alkaloids, pigments, aromatic compounds, terpenoids, etc.
Enzymes
Enzymes Definition: Enzymes are protein molecules acting as biological catalysts that are able to accelerate the rate of chemical reactions in cells. While they remain unchanged at the end of the process.
Enzymes Discovery:
- Jon Jakob Berzilius (1835) was the first to term the activity of enzymes ‘catalytic’.
- Louis Pasteur (1850) concluded from his studies that fermentation of sugar to alcohol is catalyzed by a vital force in living yeast cells, which he termed as ‘ferments’
- The term ‘enzyme’ was coined by W. Kuhne in 1878.
- It is derived from the Greek words, En= in and Zyme = yeast.
- In 1897, Edward Buchner was the first to isolate the enzyme, zymase, which was able to ferment sugar. (f)J.B. Sumner first purified and crystallized the urease enzyme from jack bean.
- After this, John Northrop (1930) isolated many enzymes like ‘pepsin’, and ‘trypsin’ in crystalline form. Sumner and Northrop were awarded the Nobel Prize in Chemistry along with Wendell Stanley in 1946.
- Thomas Cech and Sidney Altman (1983) were awarded the Nobel Prize in chemistry for the discovery of the catalytic property of RNA.
Properties of enzymes: The properties of enzymes are discussed in separate heads below.
Reversibility: Most of the enzymes are able to catalyze reversible reactions.
This property of the enzyme is dependent on various factors. For example, fumarase catalyses the conversion of malic acid to fumaric acid at pH 7.8 and the reverse reaction occurs at pH 6.2.
Biomolecules chapter summary with important points
\(\text { Malic acid } \frac{\text { Fumarase }(\mathrm{pH}: 7.8)}{\text { Fumarase }(\mathrm{pH}: 6.2)} \text { Fumaric acid }\)
Reusability: Enzymes remain unchanged and are released after the completion of the biochemical reactions. The unchanged enzyme is reused again.
Solubility: Enzymes are proteinaceous in nature and are soluble in water, mild glycerol, sodium chloride solution, and alcohol.
Proteinaceous nature: Enzymes are generally globular proteins. All the enzymes have a specific amino acid sequence.
Usually, they are colloidal in nature and have high molecular weights. For example, the molecular weight of bacterial ferredoxin is 6000 Da.
Carry charge: Enzymes are charged molecules.
Due to the presence of amino acids, each enzyme has a charge. The charge depends on the pH of the solution.
Biocatalysis: The enzymes show catalytic properties. They cannot initiate or stop a reaction, but only accelerate the rate of reactions. They also remain unchanged at the end of the reaction and, thus, can be reused as minute quantities are required for every reaction.
Buffering capacity: Enzymes have a buffering capacity (acid-base). They are amphoteric molecules that behave both as acids and bases.
Isoelectric point or pH (I): Each enzyme has a specific isoelectric point. It is a specific pH at which the net charge of protein equals zero so that it does not move in an electric field.
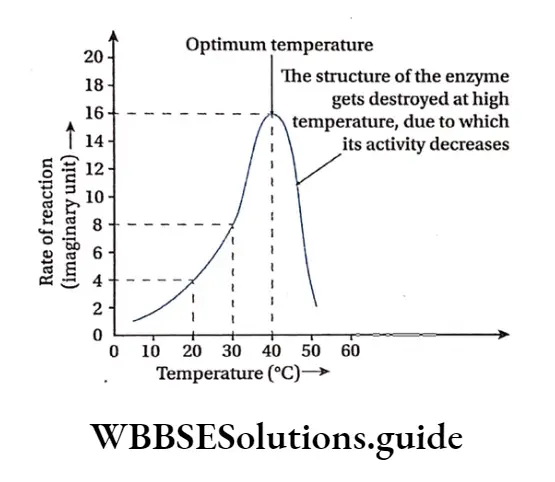
pH temperature sensitivity: Due to the proteinaceous nature, enzymes are extremely sensitive to the pH and temperature. Their structure and activity depend on these two factors.
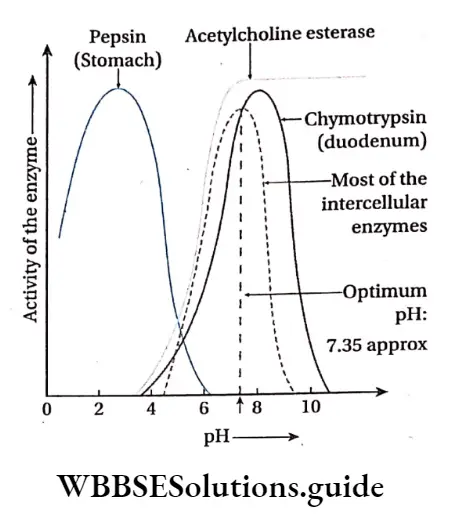
Most enzymes have an optimum pH which ranges between 4 to 9 and the optimum temperature range for the activity of the enzymes is 37°-40°C.
At very low temperatures, the enzymes are not active. At high temperatures of 50°-60, °C usually enzyme activity is usually damaged, however, few enzymes are activated at higher temperatures (e.g., Taq polymerase from Thermus aquaticus).
For most enzymatic reactions, a 10° C rise in temperature doubles the rate of reaction. While the temperature decreases by 10° C the rate becomes half.
This measure of the rate of change of a biological or chemical system as a consequence of the change of temperature by 10° C is called Q10 (temperature coefficient).
\(\begin{array}{r}
\mathrm{Q}_{10}=\frac{\text { Rate of the reaction at temperature }(\mathrm{t}+10)^{\circ} \mathrm{C}}{\text { Rate of the reaction at temperature } \mathrm{t}^{\circ} \mathrm{C}} \\
\text { [Mostly, value of } \mathrm{Q}_{10}=2 \text { to 3] }
\end{array}\)
Denaturation: When enzymes are heated or exposed to extremely high temperatures, the non-covalent bonds between peptide chains break giving rise to the primary structure of the enzyme which is a protein.
This unfolding of protein is due to the loss of secondary, tertiary, and quaternary structures. Due to this effect of denaturation, the loss of activity takes place due to the loss of the active site.
Biomolecules Class 11 Notes
Specificity: Enzymes catalyze specific reactions. Each enzyme is specific for each reaction.
This catalysis occurs in the special site where the reactants (known as substrates in enzymatic reactions) bind and react to form the products.
This site is known as the active site of the enzyme, which is specific for the substrates. For example, sucrase catalyzes the hydrolysis of sucrose only. Similarly, protease breaks down proteins.
Activity: The activity of the enzyme refers to the rate of reaction at which it binds to the substrate. Enzymes lower the activation energy of the reactions.
The measure of enzyme activity, i.e., specific activity is usually expressed as n moles of substrate transformed to products per minute per mg of the enzyme under optimal conditions of measurements.
NEET biomolecules revision notes with MCQs
It can catalyze the conversion of about 104-105 units of the substrate into products, in one minute.
Substrate concentration: The activity of the enzymes depends on the concentration of the substrate. The activity of the enzymes increases up to a certain concentration of the substrate, after which it attains a maximum velocity.
Location:
- It is present in all living cells. They may remain within the cells in inactive form. These are called proenzymes or zymogens. They are converted to active enzymes only in the presence of certain factors.
- Some of the intercellular enzymes remain within certain organelles such as nucleus, mitochondria, plastid, etc. They may also remain within the cytosol or attached to the plasma membrane, etc.
- Extracellular enzymes like pepsin are synthesized within the cells of some organs but are secreted outside the cells, where they show their action.
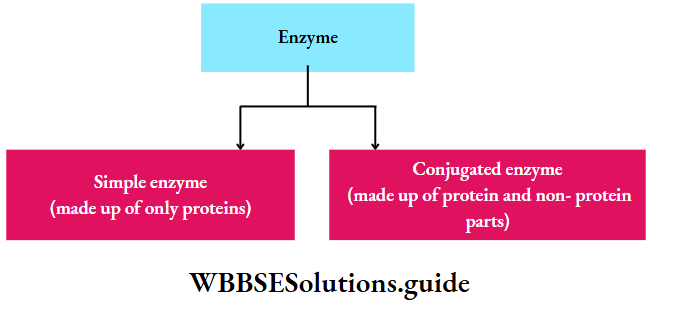
Chemical nature of enzymes: All enzymes are protein in nature but ribozyme is a complex of the ribosomal RNA (rRNA) and protein. This catalyzes the synthesis of protein from the mRNA template. Generally, the enzyme is of two types-
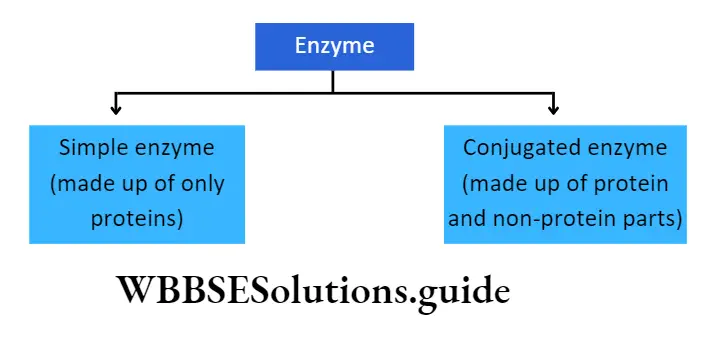
Simple enzyme: It comprises only a protein part called apoenzyme. E.g., pepsin, trypsin, amylase.
Conjugated enzyme: It comprises a protein part or apoenzyme with a non-protein part or cofactor. A cofactor can be inorganic or organic in nature. Conjugated enzymes are also called holoenzymes.
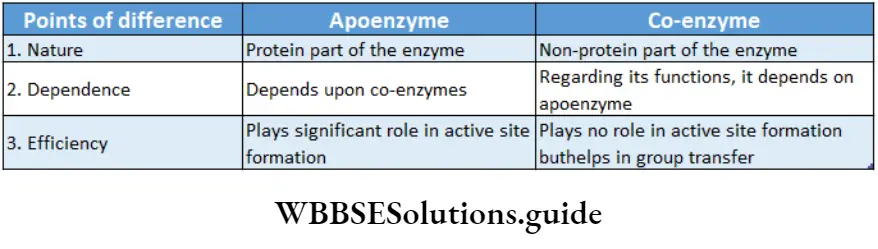
Apoenzyme: The protein part of the enzyme is called apoenzyme. The inactive form of the apoenzyme is known as a proenzyme or zymogen.
The proenzyme may contain several extra amino acids in the protein. These additional amino acids are removed by proteolysis.
This allows the final specific tertiary structure to be formed before it is activated as an apoenzyme.
The apoenzyme is thermolabile. It is destroyed under high temperatures and on application of heat. The apoenzymes have one or more active sites to which co-factors attach and form the holoenzyme.
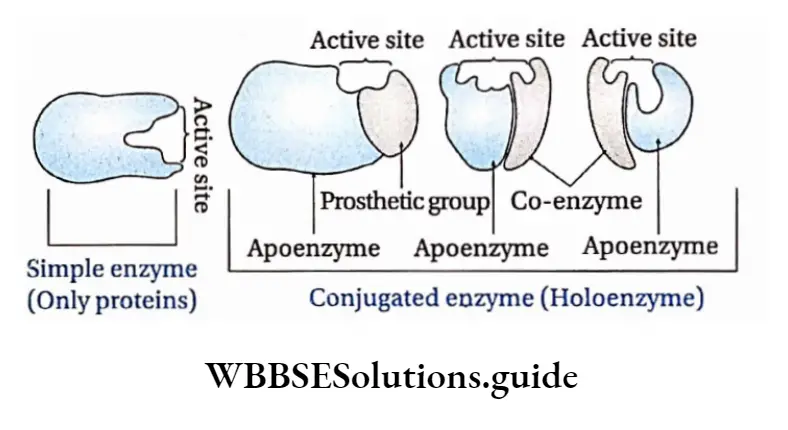

Cofactor: The nonprotein part of the enzyme responsible for the catalytic activity is called cofactor. Cofactor is thermostable. There are three recognized categories of cofactors. These are of the following types
Inorganic ion: Inorganic ions are alternately called metal activators. The different types of inorganic ions like Cu2+, K+, Fe2+, Mg2+, Zn2+, Mn2+, etc., play an important role in activating the catalytic reaction. For example, zinc is a cofactor of the enzyme carboxypeptidase.
Prosthetic group: The organic nonproteneous cofactor or inorganic factor that remains firmly attached to the protein part of the enzyme through a covalent bond and assists in the catalytic activity, is known as the prosthetic group.
For example, in peroxidase and catalase, which catalyze the breakdown of hydrogen peroxide to water and oxygen, haem is the prosthetic group and it is a part of the active site of the enzyme.
Co-enzyme: The organic non-proteinaceous, heat-stable part of an enzyme that remains loosely attached to the apoenzyme, is called a coenzyme.
Usually, they attach only at the time of catalysis. E.g., NAD (Nicotinamide adenine dinucleotide), NADP (Nicotinamide adenine dinucleotide phosphate).
This is derived from nicotinic acid and can exist in both oxidized and reduced forms.
Biomolecules Class 11
Metalloenzyme
The enzymes which contain metal ions, that are directly bound to the protein or to enzyme-bound non-protein components, are called metalloenzymes. E.g., Carbonic anhydrase is a zinc metalloenzyme.
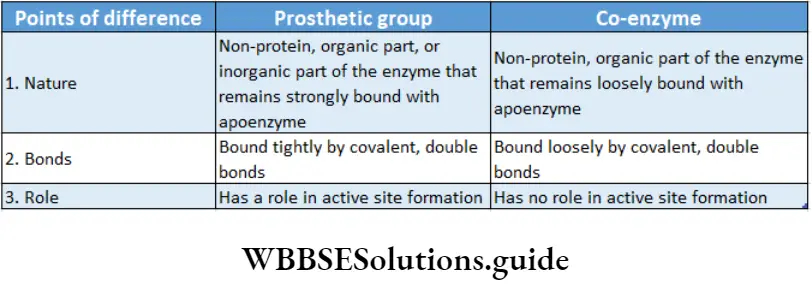
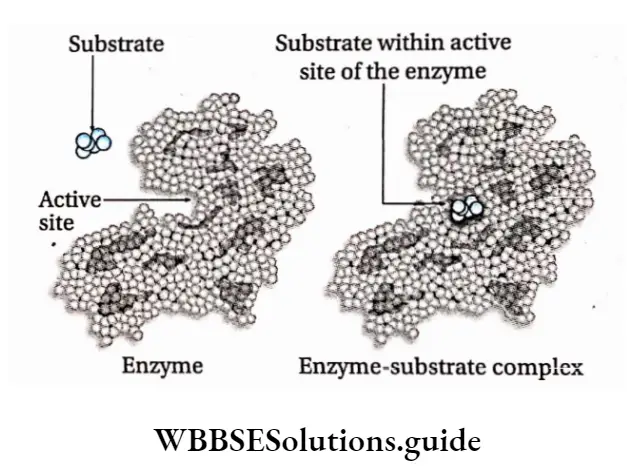
Structure of the enzyme: Enzymes are proteins and are made up of long chains of amino acids. The linear chains may fold to form tertiary structures.
Different enzymes have different sequences of amino acids.
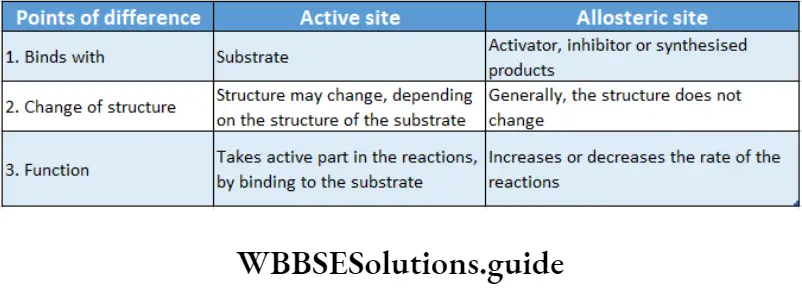
The active site is a location on the enzymes that are formed when the enzymes fold into their functional shape.
The substrate binds to the enzyme at the active site. The complex formed by the union of the enzyme and its substrate is called the enzyme-substrate complex.
Once the substrate has been chemically modified, meaning the reaction ends, it becomes a product.
That product then gets released from the active site of the enzyme.
Nomenclature and classification of enzymes
- Enzymes react upon specific substrates. They are named according to the substrate with the suffix-ose or by adding ‘lytic’ to the name of the substrate.
- Enzymes can be named according to the reactions they catalyze. For example, oxidase (this type of enzyme catalyzes oxidation reactions). Enzymes can also be named according to the type of substrate of the reaction they catalyze.
- In such cases, ‘in’ may be added to the name of the substrate. E.g., pepsin, trypsin, etc.
- Enzymes may also be named according to their source. For example, papain is obtained from papaya.
- According to a modern process of naming, enzymes may have two parts in their names.
- The first part denotes the substrate while the second part denotes the reaction catalyzed by the enzyme. Example: glutamate-pyruvate transaminase.
Nomenclature and classification of enzymes based on International rules
- In 1961 the commission of enzymes of the International Glutamic Acid + Pyruvic Acid Union of Biochemistry and Molecular Biology (IUBMB) framed certain rules for the nomenclature and classification of enzymes.
- The commission recognized six different classes of enzymes, which were further divided into subclasses.
- The enzymes are now named according to their systematic position and are given the commission number. In this pattern of naming the first number indicates the class to which the enzyme belongs and subsequent numbers indicate sub-class in that order.
- For example, nitrate reductase, or nitrate oxidoreductase, is named E.C.1.7.1.1. Here, E.C. stands for enzyme commission.
The number 1, denotes the class of enzyme—oxidoreductase.
Here, the number, 7 means that it belongs to the subclass of enzymes that acts on another nitrogenous compound as electron donors.
The next number denotes that the enzyme has NAD+ or NADP+ as an acceptor of electrons.
The last number 1 indicates the above enzyme as the first enzyme of the series. This enzyme helps in the reduction of nitrate to nitrite.
The main six classes of enzymes are discussed under separate heads below.
E.C.l or Oxidoreductases: The enzymes that catalyze oxidation and reduction reactions of their substrate, i.e., the enzymes that catalyze the transfer of H or O atoms or electrons from one molecule to another are known as an oxidoreductase.
E.g., Oxidases (cytochrome oxidase), reductase (nitrate reductase), dehydrogenase (lactate dehydrogenase), etc.
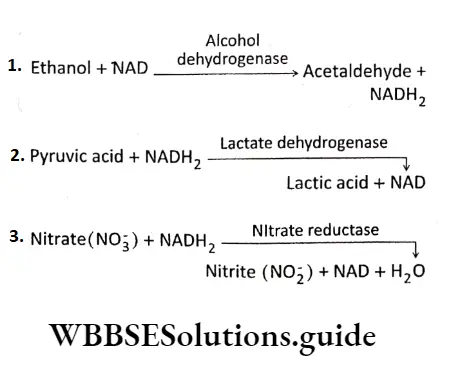
E.C.2 or Transferases: Transferases are the enzymes that catalyze the transfer of a functional group from one molecule to another. E.g., transaminase (leucine Transaminase), Phosphotransferase(2′-Phosphotranferase),etc.

E.C.3 or Hydrolases: The enzymes which catalyze the breaking down of specific bonds of various compounds by the addition of water molecule are known as hydrolases.
In hydrolysis reactions, C-O, C-N, and C-S bonds are cleaved by the addition of H20 in the form of OH- and H+ to the atoms forming the bond. Example Esterase (acetylcholine esterase), glycosidase (protease), peptidase (trypsin), etc.
\(\text { Sucrose }+\mathrm{H}_2 \mathrm{O} \longrightarrow \text { Sucrase } \longrightarrow \text { Glucose }+ \text { Fructose }\)
\(\text { Protein }+\mathrm{H}_2 \mathrm{O} \stackrel{\text { Protease }}{\longrightarrow} \text { Peptides }\)
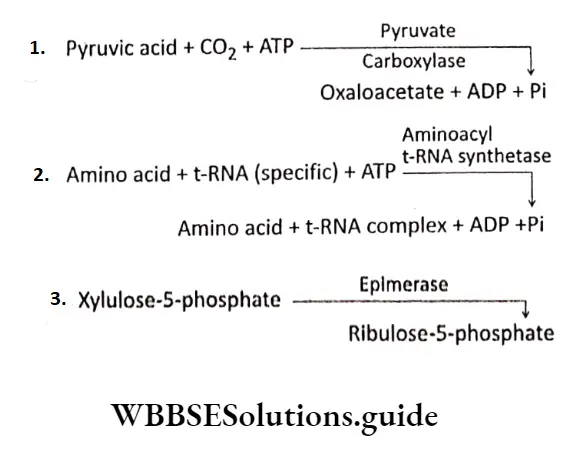
E.C. 4 or Lyases: Lyases are the enzymes that catalyze the cleaving of C-C, C-O, C-N, and C-S bonds without hydrolysis or oxidation. E.g., decarboxylase (pyruvate decarboxylase), aldolase (fructose bisphosphate aldolase), etc.

E.C.5 or Isomerases: Enzymes that catalyze the isomerization reactions that cause intramolecular rearrangement of atoms in the substrates and thus form one isomer from another, are known as isomerases. Examples are isomerase (phosphohexo isomerase), epimerase (4-hydroxyproline epimerase), etc.

E.C.6 or Ltgasc (Synthetase): Ligases are the enzymes that catalyze the synthesis of C-C, C-S, C-O, and C-N bonds in reactions coupled with the cleavage of high-energy phosphate bonds in ATP or some other nucleotide.
Examples: carboxylase (pyruvate carboxylase), synthetase (glutamine synthetase, aminoacyl tRNA Synthetase), ligase (malate CoA Ligase), Etc.
Mechanism Of Enzyme Action
The action of an enzyme depends on different factors. Enzymes are biocatalysts in nature.
All the reactions in a living organism’s body are catalyzed by enzymes.
But to begin the reactions, some amount of energy is required to initiate the reaction. Enzymes enhance the rate of chemical reactions by lowering their activation energy.
Biomolecules Class 11 Activation energy
The activation energy of a reaction is the amount of energy in calories required to bring all the molecules in one mole of a substance at a given temperature to the transition state at the top of the energy barrier.
At this point, there is equal probability for reactants to undergo a reaction to form products or to fall back and remain unreacted.
Michaelis and Menten described the activity of the enzymes with respect to activation energy. They observed that reactions that are not catalyzed by enzymes, occur at a slower rate and require higher activation energy.
For example, hydrolysis of casein requires 20600 kcal/mol in the absence of any enzyme, while only 12600 kcal/mol is required in the presence of the enzyme.
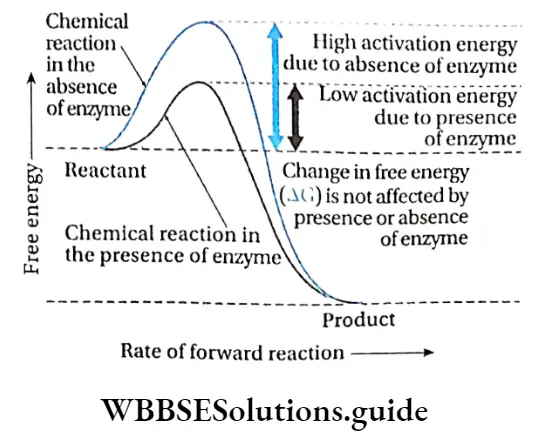
Collision theory
According to this theory, the rate of a chemical reaction is proportional to the number of collisions between reactant molecules.
So, the more often the reactant molecules collide, the more often they react to each other and the faster is the reaction rate.
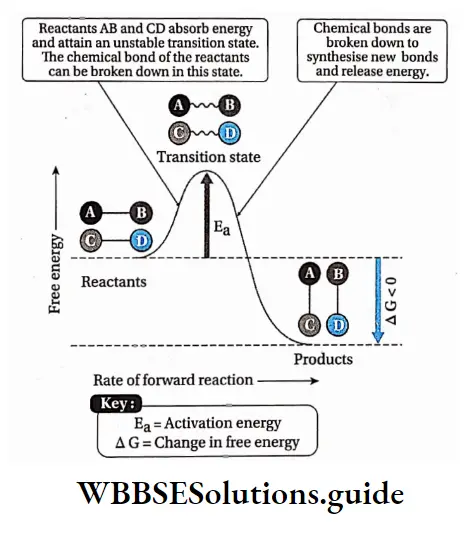
The point in the chemical reaction where there is the maximum value of energy is called the transition state.
The reactant molecules collide with each other, if they hit with enough energy to go through the transition state, they will react and form new molecules.
In the transition state, new bonds are formed while old ones are broken.
Biomolecules Class 11 Models Of Enzyme Action
Two models have been proposed regarding the mode of enzyme action:
Lock and Key hypothesis: According to this hypothesis, the enzyme active site has a configuration complementary to the shape of the substrate so that the enzyme and substrate can recognize and fit each other. This model was proposed by Fischer (1890).
According to this model, Each enzyme [E] has a specific active site or catalytic site into which only the substrate [S] fits.
The enzyme [E] and the substrate [S] both possess complementary conformation, which fits exactly into one another just like a key fits into a lock.
The active site of the enzyme is rigid and fixed, which further helps in the binding.
After binding, the enzyme [E] and the substrate [S] form the ES complex.
This [ES] complex immediately breaks down into the product [P] and enzyme [E] and the product is released from the catalytic site of the enzyme.
\(\mathrm{E}+\mathrm{S} \rightleftharpoons \mathrm{ES} \longrightarrow \mathrm{P}+\mathrm{E}\)
Special groups like -NH2, -COOH, etc., help in binding the enzyme and the substrate. Co-enzymes and activators may also influence the binding.
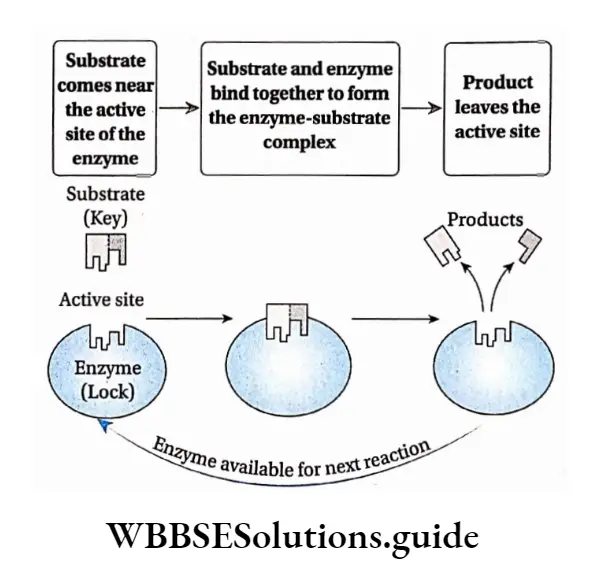
Biomolecules Class 11 Notes
Limitations of the model: Fischer explained enzyme specificity and rigidity of the binding sites of the enzymes.
However, he could not explain the allosteric behavior of enzymes (binding to a molecule at a site other than the active site) which emphasizes that the active site of an enzyme changes its conformational ensemble in order to bind with the substrate.
Induced fit hypothesis: Koshland et al. (1959) suggested another model of enzyme action with some modifications to the lock and key hypothesis.
According to this model, the active site contains two groups—the buttressing region and the catalytic region.
But the tressing region binds the substrate and forms a complex. Catalytic region weakens the bond of the substrate by electrophilic and nucleophilic processes and leads to the formation of the product maximum velocity or Vmax
This hypothesis proposes that the enzymes and their active sites are not rigid, but rather physically more flexible.
When a substrate combines with the buttressing region of an enzyme, it induces conformational changes in the enzyme structure.
This brings the catalytic region opposite to those bonds of the substrate that are to be weakened. The amino acids in the active site are modified into specific structures.
This enables the enzyme to bind with the substrate and perform its catalytic activity effectively.
The substrate changes into a product and gets released from the buttressing site due to structural changes in the site. As a result, the product is released.
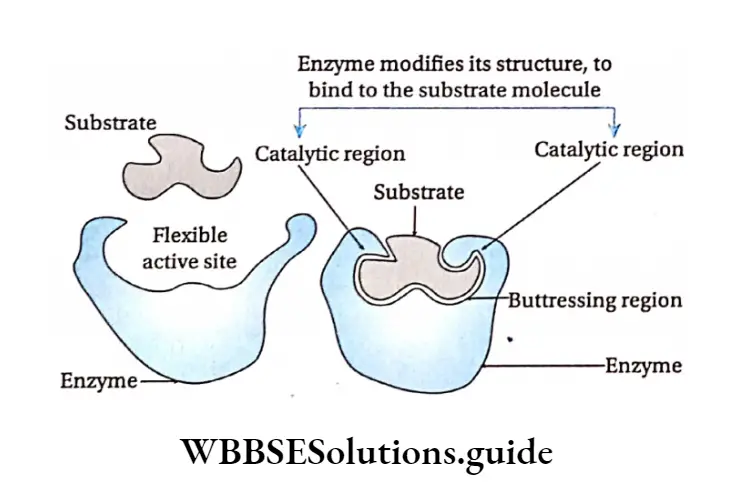
Regulation Of Enzyme Action
The action Of enzymes is regulated by different factors.
Factors affecting enzyme action
Substrate concentration: The catalytic activity of the enzyme is influenced by substrate concentration.
The rate of enzyme action increases with increasing substrate concentration.
If the substrate concentration is increased gradually from a very low concentration, the enzyme activity at first rises proportionately.
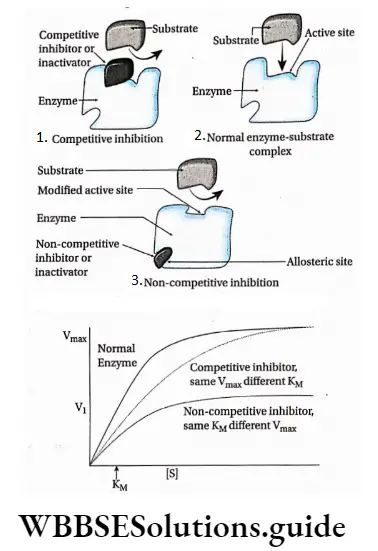
If this is plotted on a graph it will show a steep rise in the initial velocity (V0) With further increase in the substrate concentration, the initial rise in velocity of enzyme action falls and ultimately reaches a point where this velocity neither increases nor decreases. This point is called the maximum velocity or Vmax.
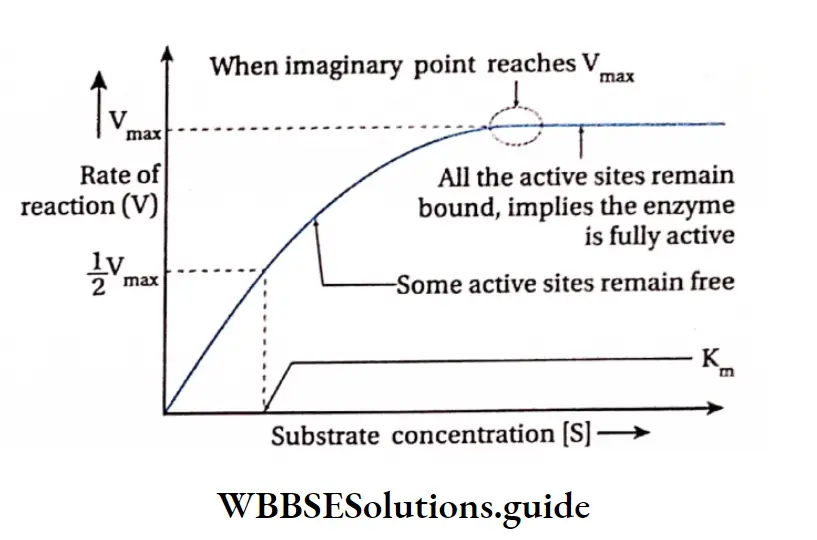
The initial reaction rate, V0 can be described by Michaelis-Menten equation as proposed by Leonor Michaelis and Maud Menten (1930).
\(\mathrm{V}_0=\frac{\mathrm{V}_{\max }[\mathrm{S}]}{\mathrm{K}_{\mathrm{M}}+[\mathrm{S}]}\)
where, Vmax= Maximum velocity
S = Substrate concentration
KM = Michaelis-Menten constant
V0 = Initial velocity
The substrate concentration at which half the maximum velocity, -y is attained by an enzyme catalysed reaction is called KM or Michaelis-Menten constant.
When initial velocity (V0) of an enzyme catalysed reaction is half of the maximum velocity (vmax) i.e., V0 \(v_0=\frac{v_{\max }}{2},\) hen the equation changes as follows—
\(\text { When, } v_0=\frac{v_{\text {max }}}{2}\)
\(\frac{V_{\max }}{2}=\frac{V_{\max }[S]}{K_M+[S]}\)
\(\text { or, } \quad \frac{1}{2}=\frac{[S]}{K_M+[S]} \text { or, } K_M+[S]=2[S]\)
or, Km =[s]
Thus the Michaelis-Menten constant [KM] is the amount of substrate concentration [S] when the initial velocity of an enzyme action is half of the maximum velocity \(\left[\frac{V_{\max }}{2}\right]\).
The KM value is an indicator of the affinity that an enzyme has for its substrate. Therefore, the value of KM is different for different enzymes. The KM value of the protease, which acts on a variety of proteins, varies with the type of protein.
An equation with a high KM indicates that the enzyme does not bind efficiently with the substrate, and Vmax will only be reached if the substrate concentration is high enough to saturate the enzyme.
Temperature: Enzymatic reactions usually take place between 20°-40°C, but at high temperature of 60’-70°C they get denatured as they are thermolabile. Generally the enzymatic action is maximum at an optimum temperature of 37C.
Enzyme concentration: The increase in the concentration of enzyme increases the rate of enzyme action within the cell.
At constant temperature and pH, a high level of substrate concentration increases the rate of enzyme action proportionately.
Product concentration: A high concentration of accumulated product inhibits an enzyme to catalyse a forward reaction.
This type of inhibition is known as feedback inhibition or product inhibition.
pH: Enzymatic action is sensitive to pH. Each enzyme shows its maximum activity at a particular pH known as optimum pH. Some enzymes are active in acidic medium while others in alkaline medium. Each enzyme can act efficiently at its optimum pH. E.g., pepsin acts at an optimum pH of about 2.0 while trypsin acts at an optimum pH of 8.5
Methods of regulation of enzyme action
Enzyme action can be accelerated or inhibited by allosteric modulation and feedback regulation.
Allosteric modulation: The property of an enzyme by which an effector molecule binds to any site other than the active site of the enzyme, resulting in acceleration or inhibition of enzyme action is known as allosteric modulation.
Allosteric site: Some enzymes possess a second site of activity other than the active site, called the allosteric site. Such enzymes are known as allosteric enzymes.
Allosteric enzymes do not obey Michaelis-Menten kinetics. The effector molecule or allosteric modulator binds to the effector or allosteric site while the substrate molecule binds to the active site for the reaction to take place.
Allosterism: Some modulator molecules upon binding to the allosteric sites of the enzymes influence the enzyme activity. This phenomenon is called allosterism.
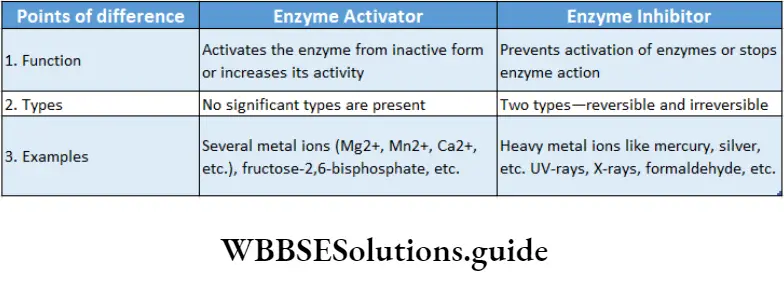
Compounds that can speed up the enzyme action are known as allosteric activators or inducers while those which slow down the reaction rate of an allosteric enzyme are known as allosteric inhibitors.
Types of allosteric: According to the property there are two types of allosteric—
Positive allosteric: When an effector or inducer molecule that binds with the allosteric site of an enzyme, enhances the binding capacity or the functional efficacy of the active site, then it is known as positive allosteric. In this case, the reaction proceeds in the forward direction resulting in formation of the product.
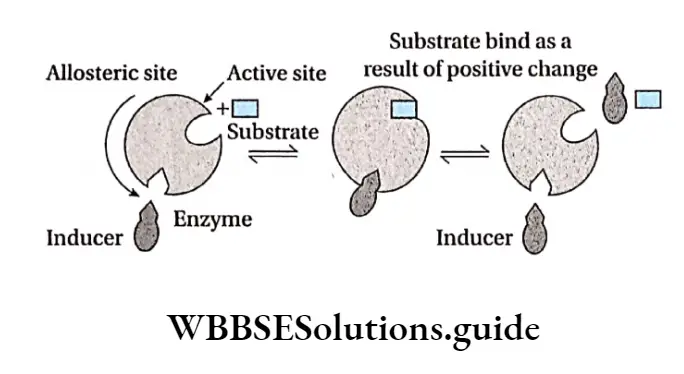
This in turn increases the enzyme activity and hence the rate of the reaction increases. An example of positive allosterism is the binding of oxygen molecules to hemoglobin.
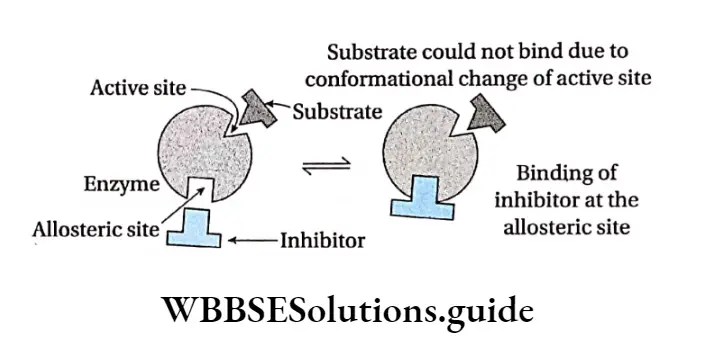
Negative allosteric: Sometimes, when an inhibitor molecule binds with the allosteric site of an enzyme, the conformation of the active site changes. As a result, the substrate cannot bind to it. This is known as negative allosterism.
In this type, the reaction is inhibited so, the product is not formed. This, in turn, reduces the rate of the reaction. For example, when 2,3-bisphosphoglyceric acid binds to an allosteric site of hemoglobin, the affinity towards oxygen decreases in all subunits of the protein.

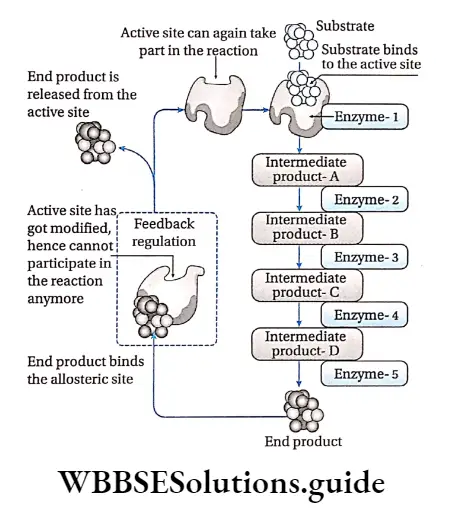
Feedback regulation: When the concentration of the end product of a series of enzymatic reactions regulates the activity of the enzymes involved, the phenomenon is called feedback regulation.
When a large amount of product is produced, it binds to an allosteric site of the first enzyme in that reaction series, thus, inhibiting its activity. When the concentration of the end product decreases, it frees the allosteric site and the enzyme reverses back to its active state.
For example, the amino acid isoleucine is synthesized through a series of biochemical reactions from the amino acid threonine. An adequate amount of isoleucine as an end product in the cell, inhibits the activity of the enzyme threonine deaminase.
It is caused by feedback inhibition which prevents the further synthesis of isoleucine. A decrease in the concentration of isoleucine in the cell, activates the threonine deaminase enzyme by feedback mechanism.
Carbohydrates, proteins, lipids, and nucleic acids notes
Enzyme Inhibitors And Enzyme Inhibition
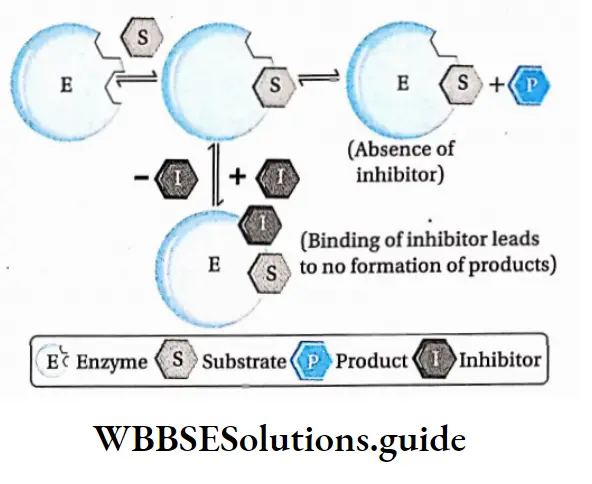
The substances that inhibit the activity of the enzymes are called enzyme inhibitors and the mechanism by which they do so is called enzyme inhibition.
Types Of Enzyme Inhibition
Enzyme inhibition is of two types—
- Irreversible and
- Reversible.
Irreversible inhibition: The method of inhibition by which the activity of the enzymes is inhibited permanently and cannot be restored, is called irreversible inhibition.
Characteristics: An irreversible inhibitor binds to the active site and changes its structure permanently or destroys the protein structure of an enzyme.
It stops the enzymatic activity and the enzyme cannot be recovered.
Heavy metal ions, SH groups, radiations like UV rays, X-rays, are responsible for irreversible inhibition.
For example, salts of cyanides inhibit the activity of cytochrome oxidase essential for cellular respiration, resulting in cell death.
Reversible inhibition: The method of inhibition by which the activity of an enzyme is inhibited temporarily is called reversible inhibition.
Characteristics: In this type of inhibition, some inhibitor molecules may attach to the active site by non-covalent interactions.
It may also involve feedback inhibition or modification of the active site by positive modulator, etc.
When these inhibitors are released from the active site, the enzyme reverses back to the functional state.
Types: There are three types of reversible inhibition—competitive, non-competitive and uncompetitive.
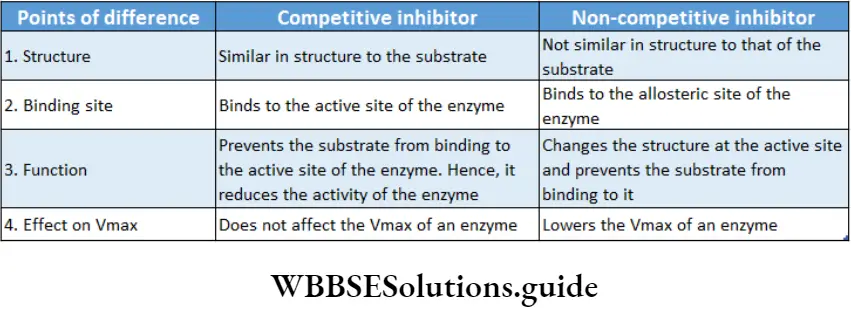
Competitive inhibition: Competitive inhibitors are molecules that are structurally similar to the substrates and compete with the substrates to bind at active sites.
This leads to slowing down of the reactions. The mechanism is called competitive inhibition. Competitive inhibitors increase KM value, thus decrease substrate affinity of the active site. Vmax remains unchanged.
For example, malonate is the competitive inhibitor of succinate dehydrogenase. Malonate binds to the active site of succinate dehydrogenase, preventing succinate from binding to it.
Non-competitive inhibition: Non-competitive inhibitors have no structural similarity with the substrate but may form an inhibitor-enzyme complex at the allosteric site of the enzyme.
It inactivates an enzyme by changing the structure of the active site. Thus, the substrate can no longer recognise or bind to the active site of the enzyme.
This prevents the enzyme-substrate complex formation. Non-competitive inhibitors lower the Vmax of an enzyme but keep the value of KM unchanged.
For example, non-nucleoside reverse transcriptase inhibitors or NNRTIs (like Etravirine) can bind to the reverse transcriptase enzyme of HIV, inhibiting the synthesis of viral RNA without interfering with substrate binding. This inhibitor is used as an anti-HIV drug.
Uncompetitive inhibition: Uncompetitive inhibitors bind to the enzyme after the formation of the enzyme-substrate complex.
It binds close to the active site. This stops the action of the enzyme by altering the structure of the enzyme-substrate complex.
As a result, KM decreases. The Vmax also decreases as a result of the removal of activated complexes.
Significance Of Enzyme Inhibition
- Excess products can be regulated by feedback inhibition.
- An idea about the different metabolic reactions within the body can be derived.
- Structure and activity of the enzymes can also be explained by these theories.
- Different medicines and pesticides can be designed applying the knowledge of enzyme inhibition.
Denaturation of enzymes
High temperature, high energy radiation (UV rays, X-rays, etc.), heavy metals (Ag+, Hg 2+, As+), salts, concentrated acids, alkali, etc., affect the active site of the enzymes. This in turn changes the structure of the proteins, breaking the hydrogen bonds. This finally destroys the enzyme integrity permanently.
Isoenzyme or Isozyme
- The different forms of an enzyme, differing only in certain amino add sequences but catalyzing the same reaction, are called isoenzymes.
- Generally, different isoenzymes are found within different cells and tissues.
- Due to slight differences in the molecular structure, they have different properties (like favorable pH, affinity towards the substrate, the effect of inhibition, etc.).
- For example, the lactate dehydrogenase enzyme of human cells has five isoenzymes.
- An enzyme, a-amylase, of wheat endosperm has 16 isoenzymes. Alcohol dehydrogenase of maize has 4 isoenzymes.
Allozyme
Allozymes or allozymes are variant forms of an enzyme that are coded by different genes of the same locus. E.g., DNA polymerase.
Proenzymes or Zymogens
Proenzymes or zymogens are biologically inactive substances which are metabolized into enzymes. For example, HCl of gastric juice alters the inactive pepsinogen to an active pepsin enzyme.
Biomolecules Notes
- Amphiphilic: A molecule having both hydrophilic and hydrophobic parts.
- Calorific value: The energy contained in food or fuel, determined by heat produced by the complete combustion of its unit quantity. This is expressed in joules per kilogram.
- Chiral molecule: A molecule that is non-superposable on its mirror image.
- Culture medium: It is a solid or liquid mixture of various substances, designed to support the growth of microscopic organisms, plants, etc., in artificial environmental situations.
- Furfural: It is a heterocyclic (ring structure) aldehyde compound having a chemical formula of C5H4O2. It is a colorless oily liquid, which quickly darkens when exposed to air.
- Glycocaiyx.’The glycocalyx is a glycoprotein-polysaccharide covering that surrounds the cell membranes of some bacteria.
- Hexosamine: It is an amino sugar produced by the addition of an amine group to a hexose. Example glucosamine (based upon glucose), fructosamine (based upon fructose), etc.
- Fmmunog robin: Any of several classes of structurally related proteins, found in serum, other body fluids, and tissues, that help in fighting diseases is called immunoglobin or antibody.
- Melanin: A pigment, produced by melanocyte cells, that gives color to human skin, hair, and eyes.
- Mucopolysaccharides: These are heteropolysaccharides composed of hexosamines and repeating units of monosaccharides.
- Mutation: A relatively permanent, heritable alteration in the genetic material involving a physical or a biochemical change.
- Myelin sheath: It is a covering made of myelin, a fatty colorless substance, around the axon(long slender projection) of some neurons and acts as an electrically insulating layer.
- Neurotransmitters: The chemicals that enable transmission, i.e., conduction of electrical impulses through the nerve cells and across chemical synapses or junctions between adjacent nerve cells.
- Non-polar molecule: If electrons are shared equally by the atoms forming a molecule, then there is no resulting charge and the molecule is non-polar.
- Nucleosome: A structural unit of an eukaryotic chromosome, consisting of a length of DNA coiled around a core of histone proteins.
- Polar molecule; A polar molecule has a partial positive charge in one part of the molecule and a complementary negative charge in the other part.
- Polarized light: The light waves in which vibrations occur in a single plane.
- Spectrophotometer: An apparatus for measuring the intensity of light in a part of the spectrum, especially as transmitted or emitted by particular substances.
- Stereoisomer: Each of two or more compounds differing only in the spatial arrangement of their atoms.
- Sugar acids: A monosaccharide containing -COOH group. Thermolabile: A substance that can be destroyed by the application of heat.
- X-ray diffraction: It is a rapid analytical technique primarily used for the identification of crystalline substances and crystalline phases in a material and can provide information on its atomic and molecular structure. It is also called X-ray crystallography.
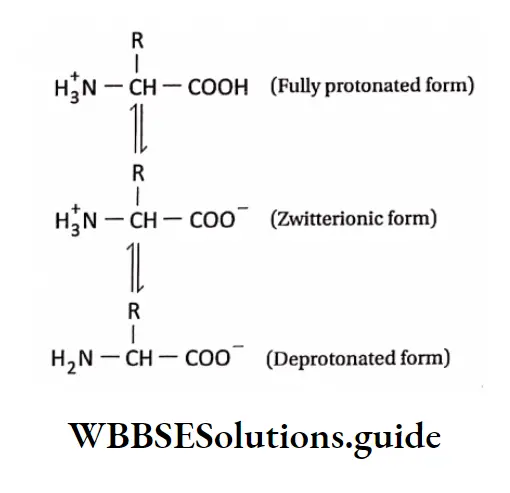
- Zwitterion: A neutral molecule having both positively and negatively charged groups.
Points of remember
- Sugar acids: A monosaccharide containing -COOH group.
- Thermolabile: A substance that can be destroyed by the application of heat.
- X-ray diffraction: It is a rapid analytical technique primarily used for the identification of crystalline substances and crystalline phases in a material and can provide information on its atomic and molecular structure of it.
- It is also called X-ray crystallography.
- Zwitterion: A neutral molecule having both positively and negatively charged groups.
- Most enzymes are conjugated proteins.
- The activity of an enzyme can be determined by its turnover number. It is the maximum number of chemical conversions of substrate molecules per second that an active site will execute for a given enzyme concentration.
- The activity of an enzyme depends on the nature and concentration of substrate and products, temperature, and pH.
- The iodine test is performed to determine the presence of starch. Iodine reacts with starch to produce a deep blue color.
- To determine the presence of reducing sugars, Benedict’s test is performed.
- Copper sulfate reacts with the reducing sugars to produce a brown/red-colored precipitate of Cu20. This determines the presence of reducing sugars.
- To distinguish between the aldose and the ketose sugars, Seliwanoff’s test is done, if a red-colored precipitate is obtained by a reaction between the sample solution and Seliwanoff’s reagent (HCL+ resorcinol), the sample sugar is a ketose sugar.