Concept Of Mole Very Short Answer Type :
Question 1. Which scientist gave the concept of molecules first?
Answer: Scientist Amadeo Avogadro gave the concept of molecules first.
Question 2. Define a molecule.
Answer:
Molecule: A molecule is the smallest particle of the substance (element or compound) which can exist independently.
Question 3. What is the number of molecules in 1°8 g water? (H=I, O=16)
Answer: 6023 x 10+23.WBBSE Solutions For Class 9 Physical Science And Environment Concept Of Mole
Question 4. Write the name of the scientist who gave the idea of the molecule.
Answer: Avogadro.
Read and Learn all WBBSE Solutions for Class 9 Physical Science And Environment
Question 5. What is the number of molecules in 22°4 liter of ammonia at STP?
Answer: 6.023 x 1023J
Question 6. If the number of molecules in V liters of hydrogen gas at STP is n, what is the number of molecules in \(\frac{v}{10}\) liters of CO2 gas at STP?
Answer: Number of molecules in \(\frac{v}{10}\) litres of CO2 gas =\(\frac{n}{10}\)
Wbbse Class 9 Physical Science Solutions
Question 7. How many atoms are present in one molecule of sulphuric acid?
Answer: 7 (H = 2, S=1, O = 4).
Question 8. What is the number of nitrogen atoms in 14 g of nitrogen gas?
Answer: \(=\frac{6^{.} 023 \times 10^{23}}{2}\) =3.115 x 1023
Question 9. What is the number of molecules of oxygen in one mole of oxygen molecule?
Answer: 6.023 x 1023. molecules.
Question 10. How many molecules of carbon dioxide are present in 11 grams of carbon dioxide?
Answer: 1.50575 x 1023.

WBBSE Class 9 concept of mole solutions
Question 11. What would be the volume of 22 gram of carbon dioxide at standard temperature and pressure -? (C=12, O=16)
Answer: 11.2 liters.
Question 12. 22.4 liter of ammonia and 22.4 liters of carbon dioxide at NTP will
(1) The number of molecules be the same or different
(2) The number of atoms be the same or different?
Answer:
(1) Same
(2) different.
Question 13. What is atomicity?
Answer: Atomicity is the number of atoms present in the elementary molecule of a substance.
Wbbse Class 9 Physical Science Solutions
Question 14. What is the number of electrons in one mole of electrons?
Answer: 6.023 x 1023 electrons. (Avogadro’s Number of electrons).
Question 15. What is the value of Avogadro’s number?
Answer: The value of Avogadro’s number is 6.023 x 1023.
Question 16. What is the mass of 1 gm mole CO2?
Answer: 1 mole CO2 = gm molecular mass of CO2 = molecular mass expressed in gram = 44 gram.
Question 17. What is the atomic mass of oxygen?
Answer: The atomic mass of oxygen is 16 a.m.u.
Question 18. What is the number of molecules in one mole of an element?
Answer: The number of molecules in one mole of any element is 6.023 x 1023.
Question 19. What is the relation between one mole and a gram molecular mass?
Answer: One mole of a molecule means its gram molecular mass.
Question 20. How many atoms of oxygen are present in one mole of it?
Answer.
As oxygen is diatomic, the number of atoms in one mole = 6.023 x 1023 x 2 = 127046 x 1023.
Wbbse Class 9 Physical Science Solutions
Question 21. a.m.u. stands for what?
Answer: a.m.u. is the abbreviation of the atomic mass unit.
Question 22. Name the element which is taken as the standard for measurement of atomic mass.
Answer: The carbon-12 isotope is taken as the standard for measurement of atomic mass.
Question 23. The unit of which physical quantity is the mole?
Answer: A mole is the unit of the amount of a substance.
Question 24. What is the mass of 1 gram H-atom?
Answer: 1 gram H-atom means gm atomic mass of hydrogen, i.e., 1 gm of hydrogen.
Question 25. What is the volume of 1 gram mole of any gas at S.T.P.?
Answer: The volume of one gram mole of any gas at S.T.P. is 22.4 liters.
Wbbse Class 9 Physical Science Solutions
Question 26. How many grams make 1 gram atom of nitrogen?
Answer: The atomic weight of nitrogen = 14. So, 14 grams of nitrogen is equivalent to, 1 gram atom of nitrogen.
Question 27. What is meant by 1 mole H2?
Answer: The molecular mass of Hydrogen (H2) is 2. So, 1 mole H2 is 2 gm hydrogen.
Question 28. How many H, molecules are contained in. one mole of hydrogen gas?
Answer: Number of hydrogen molecules in a mole of hydrogen gas = 6.023 x 1023.
Question 29. What is the relation between molecular weight and vapor density?
Answer: Molecular weight is twice the vapor density of a gas.
Question 30. What is the number of molecules in one gram-molecular weight of a gas at N.T.P.?
Answer: 67023 x 1023
Question 31. What is the vapor density of a gas?
Answer: The vapor density of a gaseous substance is the ratio of the weights of a certain volume of that gas and the same volume of Hydrogen gas at N.T.P.
Question 32. What is meant by ‘1 mole NH3?
Answer: The molecular weight of NH3 = 14 + 3 = 17.
Wbbse Class 9 Physical Science Solutions
17g of ammonia (NH3 ) = 1 mole.
Question 33. What is a mole of a substance?
Answer: The molecular weight of a substance expressed in gram is called mole of the substance.
Question 34. What is the molar volume of a substance?
Answer: The molar volume of a substance is the volume of the gram molecular weight of any substance in a gaseous state.
Concept of mole WBBSE Class 9 solutions with answers
Question 35. What is the vapour density of a gas?
Answer: Vapour density is the ratio of weights of some volume of a gas or vapor to the weight of the same volume of hydrogen at the same temperature and pressure.
Question 35. What is Avogadro’s number?
Answer: Avogadro’s number is the number of molecules present in 1 gram molecule of a substance.
Question 37. What is the atomicity of helium?
Answer: The atomicity of helium is 1.
Question 38. Who postulated the atomic theory?
Answer. John Dalton postulated the atomic theory.
Question 39. Give the name of a monoatomic gas.
Answer: Argon (Ar).
Question 40. Which element has atomicity 4?
Answer: Phosphorus.
Question 41. Which one is heavier between 2-gram-molecule ammonia and 2-gram-molecule carbon dioxide?
Answer: 2 gram-molecule ammonia = (2 17) g = 34 g NH3 .
2 gram-molecule carbon dioxide = (2 44) g = 88 g CO2 .
So, 2-gram-molecule carbon dioxide is heavier than 2-gram-molecule ammonia.
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 42.Determine the moles of atoms for each of the following elements (1) 64 g of O (2) 23.8 g of U [M = 238]
Answer:
(1) 4.0 mole
(2) 0.1 mole.
Question 43. Determine the gram of each element in (1) 2.0 moles of K (2) 0.1 mole of U.
Answer:
(1) 78 g
(2) 23.8 g.
Question 44. A small cup of coffee contains 3.14 mol of water molecules. How many H2 O molecules are present in it
Answer: 1.891 x 1024.
Question 45. The mass of a copper coin is 3.2 g. Suppose it was pure copper. How many moles of Cu atoms would the coin contain, given a molar mass of Cu of 63.54 g mol– ?
Answer. 0.0504 mol Cu.
Question 46. Determine the formula mass of the following compounds.
(1) Na3PO4 (atomic mass : Na = 23, P = 31)
(2) Cu3(PO4)2 (atomic mass : Na = 23, P = 31)
Answer:
(1) 164
(2) 382
Question 47. Determine the moles of molecules in 500 g of CaCO3.
Answer: First, determine the formula mass of CaCO3.
Formula mass =(1×40)+(2 × 14)+(6×16)=164
n=\(\frac{\mathrm{m}}{\mathrm{M}}\)=\(\frac{500 \mathrm{~g}}{100 \mathrm{~g} \mathrm{~mol}^{-1}}\)
Question 48. Determine the moles of molecules in 13.2 g (NH4)2SO4.
Answer: 0.1 mol.
Question 49. Determine the grams of the compound in 2.5 mol of Ca(NO3)2. Formula mass = (1 x 40) + (1 x 12) + (3 x 16) = 100. Apply n= 5 mc
Answer:
Formula mass = (1 x 40) + (2 x 14) + (6 x 16) = 164; n=\(\frac{\mathrm{m}}{\mathrm{M}}\)
∴ m=n x M=2.5 mol– x 164 g mol = 410 g.
WBBSE Class 9 Physical Science mole concept notes
Question 50. Find the molar mass of Na2SO4.
Answer:
Molar mass = (2 x molar mass of Na) + (molar mass of S) + (4 x molar mass of O)
= (2 x 22.99) + (32.06) + 4(16) g mol-1
= 142.04g mol-1
Concept Of Mole 2 Marks Question And Answers:
Question 1. What is the molar volume of a gas at STP?
Answer:
The molar volume of a gas at STP
Molar Volume: The volume of a gram molecule of any gas at STP is called its molar volume. Its value at STP is 22.4 liters.
We know, 1 gram molecule of a gas contains Avagadro’s number of molecules.
So, the molar volume of a gas contains Avogadro’s number of molecules.
Question 2. What is Berzillius’ hypothesis?
Answer:
Berzillius’ hypothesis
Under the same conditions of temperature and pressure, equal volumes of all gases contain the same number of atoms.
Question 3. Two identical jars contain CO2 and NO at the same temperature and pressure. Which jar contains a greater number of oxygen atoms?
Answer:
Given
Two identical jars contain CO2 and NO at the same temperature and pressure.
According to Avogadro’s hypothesis, both gases contain an equal number of molecules. Each molecule of CO2 contains 2 oxygen atoms whereas each molecule of NO contains 1 oxygen atom. So, the jar containing CO2 will have more oxygen atoms.
Question 4. What is the importance of Avogadro’s law in chemistry?
Answer:
Importance of Avogadro’s law in chemistry
(1) It is Avogadro’s law which states the difference between molecules and atoms for the first time.
(2) Gay-Lussac’s law of gaseous volumes can be proved with the help of this law.
(3) With the help of this law we can correlate Gay-Lussac’s law of gaseous volumes and Dalton’s atomic theory.
Question 5. ‘Molecular weight of nitrogen is 28’ – what does it mean?
Answer:
The molecular weight of nitrogen is 28’
The molecular weight of nitrogen is 28, i.e., one molecule of nitrogen is 28 times heavier than 1/16th of the weight of one atom of ‘16O or one molecule of nitrogen is 28 times heavier than 1/12th of the weight of one atom of 12C.
Question 6. Out of 2 g of hydrogen and 16 g of oxygen, whose volume is greater at standard temperature and pressure and why?
Answer:
Given
Out of 2 g of hydrogen and 16 g of oxygen,
The molecular weight of hydrogen = 2 x 1 = 2.
Therefore, the volume of 2 g of hydrogen at STP is 22.4 l.
The molecular weight of oxygen = 2 x 16 = 32.
Therefore, the volume of 32 g of oxygen at STP is 22°4 |, i.e., the volume of 16 g of oxygen at STP is 112 |. So, the volume of 2 g of hydrogen is greater than that of 16 g of oxygen at STP.
Question 7. Define Avogadro’s law.
Answer:
Avogadro’s law
Avogadro’s law states that “under identical conditions of temperature and pressure, equal volumes of all gases contain an equal number of molecules”.
For example: If we enclose equal volumes of three gases Hydrogen (H2), Oxygen (O2), and Chlorine (Cl2) in different flasks of the same capacity (volumes) under similar conditions of temperature and pressure, we find that all the flasks have the same number of molecules. However, these molecules may differ in size and mass.
Wbbse Class 9 Physical Science Question Answer
Question 8. What do you mean by the atomicity of a gas?
Answer:
Atomicity: The number of atoms present in one molecule of a substance is called its atomicity. Avogadro’s law helps in determining the atomicity of gaseous elements such as hydrogen, oxygen, chlorine, etc.
For example, The atomicity of oxygen (O2) is 2, the atomicity of chlorine (Cl2) is 2, etc.
Question 9. Define the atomic mass of an element in the C12 scale.
Answer:
Atomic mass: The atomic mass of an element is defined as the average relative mass of an atom of an element as compared to the mass of an atom of Carbon [C12] taken as 12.
In other words, atomic mass is a number that expresses as to how many times an atom of the element is heavier than 1/12 th of the mass of a Carbon atom [C12]. Therefore,
\(\text { Atomic mass }\)=\(\frac{\text { Mass of atom }}{\left.1 / 12 \text { Mass of Carbon}[ \mathrm{C}^{12}\right]}\)
Question 10. What is the atomic mass unit?
Answer:
Atomic mass unit
One atomic mass unit is defined as the quantity of mass equal to 1/12 of the mass of an atom of Carbon [C12].
The scale of relative masses of atoms is called the atomic mass unit scale. It is abbreviated as a.m.u.
Important questions on mole concept WBBSE Class 9
Question 11. What is average atomic mass? What is the average atomic mass of chlorine?
Answer:
Average atomic mass
Many elements occur in nature as mixtures of several isotopes. To define the atomic mass of these elements is to determine the atomic mass of each isotope separately and then combine them in the ratio of their proportion of occurrence. This is called average atomic mass.
Chlorine occurs in nature in the form of two isotopes with atomic masses 35 and 37 in the ratio of 3:1 respectively. Therefore,
Average atomic mass of chlorine \(=\frac{35 \times 3+37 \times 1}{3+1}\)= 35.5 a.m.u.
Question 12. Define Gram Atomic Mass or Gram Atom.
Answer:
Gram Atomic Mass: The atomic mass of an element expressed in grams is the gram atomic mass or gram atom.
For example The atomic mass of oxygen = 16 a.m.u.
Therefore, the gram atomic mass of oxygen = 16 g.
Question 13. Define the molecular mass of a substance.
Answer:
Molecular mass: The molecular mass of a substance may be defined as “the average relative mass of its molecular as compared to the mass of an atom of Carbon (C12) taken as 12”.
For example, The molecular mass of CO2 is 44 a.m.u.
Question 14. Find the relation between molecular weight, molar volume, and density of a gas.
Answer: The relation between molecular weight, molar volume, and density of a gas is molecular weight = molar volume x density.
Question 15. Which is heavier between 1-mole carbon and 1-mole sodium? Why?
Answer: 1 mole of sodium is heavier. Because the atomic mass of sodium is 23 and the atomic mass of carbon is 12.
Question 16. How many grams of CO, is produced when 12 g of carbon is burnt more than oxygen?
Answer: C + O2= CO2 (CO2= 12 +2 x 16 = 12 + 32 = 44)
44 g CO2 is produced when 12 g carbon is burnt in excess oxygen.
Question 17. The volume of 0°44 grams of a gas at STP is 224 cm. What is its molecular mass?
Answer:
Given
The volume of 0°44 grams of a gas at STP is 224 cm.
The volume of gram molecular mass of the gas = 22.4 liter at STP
= 22400 cm3.
Molecular mass \(=\frac{0.44 \times 22400}{224}\)=44.
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 18. What is the number? of molecules in each of 32 grams of oxygen and 44 grams of carbon dioxide? (C=12, O=16)
Answer:
No. of molecules of 32 gm of oxygen at N.T.P.= 6.023 x 1023 molecules.
No. of molecules of 32 gm of oxygen at N.T.P.=6.023 x 1023molecules.
Question 19. What is the volume of 4 grams of sulfur-di-oxide gas at standard temperature and pressure? (S = 32, O = 16).
Answer: Gram molecular mass of sulfur dioxide (SO2)= 32 + 32 = 64 gm.
At NTP, 64 gm of SO2 occupies a volume of 22.4 liters.
∴\(\frac{22 \cdot 4 \times 4}{64} \text { litre }\)
liter = 1.4 litres.
∴ The required volume is 1.4 liters.
Question 20. How many molecules of carbon dioxide are present in 11 grams of carbon dioxide?
Answer: Gram molecular mass of CO2 = 12x 16 x 2
= 12 + 32 = 44 gm.
At. N.T.P. 44 gm of CO2 will have 6.023 x 1023 no. of molecules.
Question 21. What would be the volume of 22 grams of carbon dioxide at standard temperature and pressure? (C = 12, O = 16)
Answer: Gram molecular mass of CO2 = 12 x 32 = 44 gm.
At, N.T.P. 44 gm of CO2 will occupy 22.4 litre
1 gm of CO2 will occupy=\(\frac{22 \cdot 4}{44}\)
22 gm of CO2 will occupy=\(\frac{22.4 \times 22}{44_2}\)=11.2 litre.
∴The required volume = 11.2 liters.
WBBSE Class 9 solved exercises on concept of mole
Question 22. What is the number of molecules in 1 g mol of hydrogen and also in 1 gm of hydrogen?
Answer: Number of molecules in 1 g mol of H2
= 6023 x 1023.
Now, 1 g mol of H2= 2 g of H2.
∴ Number of molecules in 1 gram of
hydrogen \(=\frac{6.023 \times 10^{23}}{2}\)
= 3.0115 x 1023
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 23. What is the volume of 4 g of SO2 gas? at standard temperature and pressure? (S= 32, O = 16)
Answer: Mass of 1 gm mol of sulfur dioxide (SO2)
=82+2×16=64g.
The volume of 64 g of SO2 at STP = 22.4 L.
∴ Volume of 4 g of SO2 at STP =\(=\frac{22. 4}{64} \times 4\)
=1.4 L.
Question 24. What is the number of molecules present in 1.8 g of water?
Answer:
Gram-molecular weight of water = 18 g.
∴The number of molecules in 18 g of water = 6.023 x 1023.
∴ Number of molecules in 1.8 g of water
\(=\frac{6023 \times 10^{23}}{18} \times 1.8\)= 6.023 x 1023
Question 25. The volume of 0.44 g of a gas at STP. is 224 cm3. What is the molecular weight of the gas?
Answer:
Given
The volume of 0.44 g of a gas at STP. is 224 cm3.
22.4 L = 22.4 x 1000 cm3= 22400 cm3
Here, the mass of 224 cm3 of the gas = 0.44 g.
∴ Mass of 22400 cm3 of the gas
\(=\frac{0.44}{224} \times 22400\) =44g.
∴ The molecular weight of the gas = 44.
Question 26. Determine the mass of one molecule of water.
Answer:
The mass of one molecule of water
The molecular weight of water = 18; therefore, the gram-molecular weight of water = 18 g.
We know, the number of molecules in 1 g. mol of water = 6.023 x 1023
∴ The mass of 6.023 x 1023 molecules of water = 18 g.
∴ The mass of 1 molecule of water
\(=\frac{18}{6^{.023 \times 10^{23}}} \mathrm{~g}\)=2.9885x 10-23 g
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 27. 64 g oxygen = how many g atoms of oxygen?
Answer:
Gram-atomic weight of oxygen =16 g.
Now, 16 g oxygen = 1 g atom of oxygen.
∴ 64 g oxygen \(=\frac{1}{16} \times 64\)
= 4g atom of oxygen.
Question 28. 2 g atom of sulphur.= How many grams of sulfur?
Answer
: Atomic weight of sulfur (S) = 32.
∴1g atom of S = 32 g of S.
∴2g atom of S = 32 x 2= 64 g of S.
Question 29. 5. g mol of water = how many grams of water?
Answer:
The molecular weight of water (H2 O)
=2×1+16=18.
∴ g mol of water = 18 g of water.
∴ 5 g mol of water = 18 x 5 = 90 g of water.
WBBSE Class 9 Physical Science Avogadro’s number solutions
Question 30. 200 g of CaCO3 = how many g mol of CaCO3?
Answer: Molecular weight of CaCO3
= 40+12+16×3= 100.
∴100 g of CaCO3= 1 g mol of CaCO3
∴ 200 g of CaCO3 \(=\frac{200}{100}\)
= 2g mol of CaCO3.
Question 31. What is the mass of 14 | of oxygen at STP?
Answer: Molecular weight of oxygen = 32.
Mass of 22.4 l of oxygen at STP = 32 g.
∴ Mass of 14 l of oxygen at STP \(=\frac{32 \times 14}{22.4}\)
= 20g.
Question 32. If the mass of 2.24 ‘| of a gas at STP is 4.4 g, what would be the molecular weight of that gas?
Answer: Mass of 2.24 l of the gas at STP = 4.4 g.
∴ Mass of 22.4 l of the gas at STP \(=\frac{4.4 \times 22.4}{22.4}\)=44g.
∴ The gram-molecular weight of the gas = 44 g.
∴ The molecular weight of the gas = 44.
Question 33. What is the mass of 4 g mol of oxygen in grams? What would be the volume of that quantity of oxygen at STP?
Answer: Molecular weight of oxygen (O2) = 32.
∴ 1 g mol of O2 = 32 g of O2
∴ 4g mol of O2= 32 x 4 = 128 g of O2
Again, we know, the volume of 1 g mol of O, at STP = 22.4 l
∴ The volume of 4 g mol of O2 at STP
= 22.4 x 4 l = 89.6 l.
Question 34. How many grams of oxygen are there in 2 g atom of oxygen? What is the number of oxygen molecules in it?
Answer: We know, 1 g atom of oxygen = 16 g of oxygen.
∴ 2 g atom of oxygen
= 16 x 2 = 32g of oxygen.
∴ 1 g mole 32 g of oxygen contains 6.023 x 1023 molecules.
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 35. What is the number of molecules in 1 g mol of hydrogen and also in.1 gram of hydrogen?
Answer: Number of molecules in 1 g mol of H2 = 6.023 X 1023
Now, 1 g mol of H2 = 2 g of H2
∴ Number of molecules in 4 gram of hydrogen \(=\frac{6.023 \times 10^{23}}{2}\) = 3.0115 x 1023
Question 36. Determine the moles of atoms for each of the following elements. (1) 36g of C (2) 6.4 g of O.
Answer:
The formula is, n = uM m= mass in g given, M = g atomic mass in g mol
n = number of moles
(1)m=369,M=12g mol-1
∴ n\(=\frac{36 \mathrm{~g}}{12 \mathrm{~g} \mathrm{~mol}^{-1}}\)=3 mole
(2) m= 6.4 g, M= 16 g mol-1
∴ n \(=\frac{6.4 \mathrm{~g}}{16 \mathrm{~g} \mathrm{~mol}^{-1}}\)=0.4 mole
Question 37. Determine the grams of each element in (1) 3.0 moles of He; (2) 0.10 mole of C.
Answer:
m = nM
(1)n=3.0 mol, M=4g mol-1
∴ m=3.0 mol x 4.08 g mol-1= 12.0 g
(2) n= 0.1 mol, M= 12 g mol-1
∴ m=0.1 mol x 12.0g mol-1 = 1.2g
Question 38. Determine the formula mass of the following compounds (1) Fe2O3 , (2) CO2 (atomic mass: Fe = 56, O = 16).
Answer:
(1) Fe2O3 ⇒ (2 x 56 +3 x 16) = 112 + 48 = 160.
(2) CO2 ⇒ (1x 124+ 2x 16) = 12+ 32=44,
Question 39. How many moles of NH3 can be produced from 2.0 mol H, in the reaction? N2(g) + 3H,(g) 2NH3(9)
Answer:
From the balanced chemical equation, we see that 3 mol H, reacts (with 1 mol N2) to give 2 mol NH3.
2.0 mol of H2 \(=\frac{2}{3} \times 2\)=\(\frac{4}{3}\)
=1.33mol of NH3
Concept Of Mole 3 Marks Questions And Answers
Question 1. What are the corollaries obtained from Avogadro’s hypothesis?
Answer:
The following corollaries are obtained from Avogadro’s hypothesis –
(1) The molecules of elementary gases are diatomic, e.g. hydrogen, oxygen, etc.
(2) The difference between a molecule and an atom is shown by this hypothesis.
(3) The molar volume of any gas at N.T.P. is 22.4 lit.
(4) The molecular weight of any gas is twice its vapor density.
(5) The molecular formula of any gas can be found.
Question 2. What is meant by the standard density and vapor density of a gas?
Answer:
Standard density and vapor density of a gas
The mass of 1 liter of a gas at STP expressed in grams is called the standard density of the gas.
Example: The standard density of hydrogen is 0.0898 g L-1. This means that the mass of 1 L of hydrogen at STP is 0.0898 g.
Vapor density or relative density: A gas may be defined as the ratio of the weight of a certain volume of the gas to the weight of the same volume of hydrogen gas, measured under the same conditions of temperature and pressure.
ie., vapour density of a gas, D \(=\frac{\text { mass of } V \text { volume of the gas }}{\text { mass of } V \text { volume of hydrogen }}\) (at the same temperacture and pressure).
Mole-mass and mole-volume relationship Class 9 WBBSE notes
Question 3. What is gram-molecular volume or molar volume? What is its value at STP?
Answer:
Gram-molecular volume or molar volume
At STP the volume occupied by one gram-mole of any gas is called the grammolecular volume or molar volume of that gas.
This volume depends on temperature and pressure but does not depend on the nature or properties of the gas.
The gram-molecular volume of any gas at STP is 22.4L. As,
(1) The volume of one gram-mole of O2 or 32 g of O2 i.e., molar volume of oxygen at STP = 22.4 L.
(2) The volume of one gram-mole of CO2 or 44 g of CO2 i.e., molar volume of CO2 at STP = 22.4 L.
Question 4. In 22.4L of ammonia and 22.4L carbon dioxide at STP,
(1) will the number of molecules be the same or different?
(2) will the number of atoms be the same or different?
Answer:
(1) According to Avogadro’s hypothesis, under the same conditions of temperature and pressure, equal volumes of all gases contain equal numbers of molecules. Therefore, in 22.4 L of ammonia and 22.4 L of carbon dioxide gases at STP, the number of molecules will be the same.
(2) One molecule of ammonia (NH3) contains 1 atom of nitrogen and 3 atoms of hydrogen, i.e., the total number of atoms (1 + 3) = 4. On the other hand, one molecule of carbon dioxide (CO2) contains 1 atom of carbon and 2 atoms of oxygen, i.e., the total number of atoms (1 + 2) = 3. Therefore, the number of atoms in the above two cases will be different.
Question 5. Explain the concept of a molecule.
Answer:
Molecule
Just as the ultimate particle of an element is an atom, the ultimate particle of a chemical compound is called a molecule. The molecules are made up of atoms of the same or different elements and are capable of independent existence. Thus, a molecule
contains two or more atoms. The properties of a substance are due to the properties of its molecules. Molecules can be classified into two types :
(1) Homoatomic molecules: These molecules are made up of the atoms of the same element. For example, molecules_as H2 , N2 , O2 , P4, etc.
(2) Hetroatomic molecules: These molecules are made up of atoms of different elements. For example, molecules of HCI, CO2, NH3, CH4, etc.
Question 6. Explain how Avogadro’s law can be applied in different chemical reactions.
Answer: Avogadro’s taw explains any chemical reaction involving gases. For example, in the case of the formation of hydrogen chloride gas, we observe that one volume of hydrogen gas combines with one volume of chlorine gas to form two volumes of hydrogen chloride
gas.
This can be explained in the following equation :
Hydrogen + Chlorine —> Hydrogen Chloride gas
1 Volume 1 Volume 2 Volume
Suppose, one volume of a gas contains n molecules. Therefore, according to Avogadro’s
law, we have
= Hydrogen + Chlorine → Hydrogen Chloride gas
n molecules + n molecules → 2n molecules
or, 1 molecules + 1 molecule → 2 molecules
or, ½ Molecules + ½ Molecules → 1 molecules
This implies that one molecule of hydrogen chloride gas is composed of ½ a molecule of hydrogen and ½ molecule of chlorine. This is possible because a molecule may contain
more than one atom. One molecule of hydrogen is found to contain 2 hydrogen atoms. Thus, ½ molecule of hydrogen means one atom of it. Similarly, ½ molecule of chlorine
means one atom of it. Therefore, one molecule of hydrogen chloride is formed from one atom of hydrogen and one atom of chlorine. This is in agreement with Dalton’s atomic theory.
Question 7. Explain the mole concept. Define mole with examples.
Answer:
M
ole
It has been found experimentally that one gram atom of any substance contains as many atoms as are present in one gram atom of carbon (12C), ie., in 12 g of 12C. The Value of the number is found to be 6.023 x 1023. Hence, a mole represents 6.023 x 1023 atoms or molecules.
Mole: A mole is the amount of any substance that contains as many elementary particles (atoms, molecules, or ions) as there are atoms in exactly 0.012 Kg (i.e., 12 g) of
carbon (12C).
Hence, one mole = 6.023 x 1023 particles
One mole of atoms = 6.023 x 1023 atoms.
One mole of molecules. = 6.023 x 1023 ions.
The number 6.023 x 1023 is called Avogadro’s number.
8. Give examples of the atomic mass of some common elements referred to as 12C= 12.
Answer:
Table: Atomic mass of a few common elements referred to as 12C = 12.
| Element | Symbol | Atomic mass | Element | Symbol |
Atomic mass |
| Aluminium | A1 | 27.0 | Lead | Pb | 207.2 |
| Antimony | Sb | 121.8 | Lithium | Li | 6.94 |
| Arsenic | AS | 74.9 | Manganese | Mn | 54.9 |
| Argon | Ar | 18 | Magnesium | Mg | 24.3 |
| Beryllium | Be | 9.01 | Mercury | Hg | 200.6 |
| Bismuth | Bi | 209 | Neon | Ne | 20.2 |
| Boron | B | 10.8 | Nickel | Ni | 58.7 |
| Bromine | Br | 79.9 | Nitrogen | N | 14.0 |
| Calcium | Ca | 40.1 | Oxygen | O | 16.0 |
| Carbon | C | 12.0 | Phosphorus | P | 31.0 |
| Chlorine | Cl | 35.5 | Potassium | K | 39.1 |
| Copper | Cu | 63.5 | Silicon | Si | 28.1 |
| Fluorine | F | 19.0 | Silver | Ag | 107.9 |
| Helium | He | 4.0 | Sodium | Na | 23.0 |
| Hydrogen | H | 1.0 | Sulfur | S | 32.1 |
| Iodine | l | 126.9 | Tin | Sn | 118.7 |
| Iron | Fe | 55.8 | Zinc | Zn | 65.4 |
Question 9. How many (1) Molecules, (2) Nitrogen atoms, and (3) Hydrogen atoms are there in 11.2 liters of ammonia at STP?
Answer: 11.2 liters of ammonia contains
(1) ½ x (6.023 x 1023 ) molecules = 3.0115 x 10% molecules.
(2) Number of nitrogen atoms = 3.0115 x 1023.
(3) Number of hydrogen atoms = 9.0345 x 1023.
Question 10. State Avogadro’s law. If there are 6.022x 1023 molecules present in V liter of nitrogen gas at STP, how many molecules are present in 2V liter of sulfur dioxide gas at STP?
Answer:
Avogadro’s law: Equal volumes of all gases under the same condition of temperature and pressure contain the same number of molecules. 6.023 x 1023 molecules of any gas at STP occupy 22.4 lit.
∴ V litre = 22.4 litre
∴ No molecules in 2V litre of sulfur dioxide
= 6.023 x1023 x 2 = 12.046 x 1023 .
Question 11. lf the number of molecules in 22.4 l of CO, gas at STP be 6.023 x 1023, what would be the number of molecules in 11.2 l of O2 gas at STP?
Answer:
Given
lf the number of molecules in 22.4 l of CO, gas at STP be 6.023 x 1023
Number of molecules in 22.4 l of CO2 gas at
STP = 6.023 x 1023.
According to Avogadro’s hypothesis, the number of molecules in 22.4 l of O2 gas at STP would also be 6.023 x 1023
∴The number of molecules in 11.2 of O2 , gas at STP
\(=\frac{6.023 \times 10^{23}}{22.4} \times 112\)=3.011 x 1023
Best study material for concept of mole WBBSE Class 9
Question 12. What is the volume of 7 g of nitrogen at standard temperature and pressure? (N = 14)
Answer:
The atomic weight of nitrogen = 14.
Since nitrogen is a diatomic gas, the molecular weight of nitrogen = 14 x 2= 28.
∴ 28 g of nitrogen = 1g mol of nitrogen
or, 1 g of nitrogen = \(\frac{1}{28}\) g mol of nitrogen
or, 7 g of nitrogen \(\frac{1}{28} \times 7\)= \(\frac{1}{4}\) g mol of nitrogen
We know, the volume of 1 g mol of nitrogen at STP = 22.4 l
∴ volume of g mol of nitrogen at STP \(=\frac{22 \cdot 4}{4} l\)=5.6 l.
Question 13. Calculate the gram molecular mass of sugar (C12 H22 O11)s
Answer:
Since the atomic mass of C = 12, H =1 and O = 16,
molecular mass of sugar (C12 H22 O11)
= 12x 124+22×1+16x 11 = 342.
Therefore, the gram molecular mass of sugar = 342 g.
Question 14. What is the gram-molecular weight of potassium chlorate? (K = 39, Cl = 35.5, O = 16)
Answer:
The molecular formula of potassium chlorate is KCIO3, Therefore, the molecular weight of potassium chlorate
= 39+ 355+16×3
= 39 + 35.5 + 48 = 122.5
∴ Gram-molecular weight of KCIO3 = 122.5 g.
Question 15. How many g mol of sulphuric acid are there in 49 g of sulphuric acid? (5 = 32,O=16, H=1)
Answer:
The molecular formula of sulphuric acid is H2SO4
Therefore, the molecular weight of sulphuric acid
=2×1+32+4×16
=2+ 32+ 64=98
∴ 98 g of H2SO4 = 1 g. mol of H2SO4
or 1 g of H2SO4 =\(\frac{1}{98}\) mol of H2SO4
or, 49 g of H2SO4 = \(\frac{1}{98}\) ×49 mol of H2SO4
= 0.5 g. mol of H2SO4.
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 16. The mass of 11.2 | of a gas at STP is 22 g. What is the molecular weight of that gas?
Answer:
Mass of 11.2 l of the gas at STP = 22 g
∴ Mass of 1 of the gas at STP \(=\frac{22}{11 \cdot 2}\)
∴ Mass of 22.4 l of the gas at STP \(=\frac{22}{11.2} \times 22 \cdot 4\)=44g
Question 17. The vapor density of a gas is 16. What is the mass of one molecule of that gas?
Answer:
Given
Vapor density of the gas = 16.
The molecular weight of that gas
= 2 x vapour density
=2x 16 = 32.
Therefore, 1 g mol of that gas = 32 g.
We know, the number of molecules present in 1 g mol of the gas is 6.023 x 1023 g
∴ Mass of 6.023 x1023 molecules = 32 g
or , Mass of 1 molecule = 32 ⁄ 6.023×1023 g
= 5.3129 x 10-23 g.
Question 18. If in 18 g of water, the number of molecules is n, what is the number of molecules in 100 g of CaCO3 and 44 g of CO2? What is the value of this number? (Ca = 40,C = 12, O = 16)
Answer:
Molecular weight of H2O = 2x 1+ 16 = 18
∴ 1 g mol of water = 18 g of water
Molecular weight of
CaCO3 = 40 + 12+ 16x 3 = 100
∴ 1 g mol of CaCO3 = 100 g of CaCO3
Molecular weight of
CO2 = 12 + 16x 2 = 12 +32 = 44
∴ 1g mol of CO2 = 44 g of CO2
We know, 1 g mol of any substance contains the same number of molecules.
∴ if there are n molecules in 18 g of water, i.e., in 1 g mol of water, there would be n molecules in 100 g of CaCO3 i.e., in 1 g mol of CaCO3 and there are also n molecules in 44
g of CO2 i.e., in 1 g mol of CO2.
This number is called Avogadro’s number. Its value is 6.023 x 1023.
WBBSE Class 9 Physical Science Chapter 4.2 Question Answer
Question 19. What is the mass of 4 g mol of oxygen in grams? What is the volume of that Z quantity of oxygen at STP?
Answer:
The molecular weight of oxygen = 32.
Therefore, the mass of 1 g mol of O2 = 32 g.
Or, the mass of 4 g mol of O2 = 32 x 4 = 128g.
The volume of 1 g mol of O2 at STP = 22.4 l.
Volume of 4 g mol of O2 at STP = 22.4 x 4
= 89.6 l.
Question 20. What are the numbers of oxygen atoms and hydrogen atoms in 0.9 g of water?
Answer:
Molecular weight of H2O = 2x 1+ 16 = 18.
Therefore, 18 g of water = 1 g mol of water
or, 1 g of water \(=\frac{1}{18}\) g. mol of water
or, 0.9 g of water \(=\frac{1}{18}\) × 0.9 or ½ mol of water
Now, 1 g mol of water contains 6.023 x 1023 molecules
\(=\frac{1}{20}\) mol of water contains \(\frac{6.023 \times 10^{23}}{20}\)molecules = 3.0115 x 10 molecules.
Now, since 1 molecule of water consists of 1 atom of oxygen and 2 atoms of hydrogen.
∴ In 3.0145 x 1022 molecules of water, the number of oxygen atoms = 3.0115 x 1022, and the number of hydrogen atoms = 2 x 3.0115 x 1022= 6.023 x 1022.
WBBSE Class 9 Physical Science molar mass and stoichiometry solutions
Question 21. What is the number of molecules in 8 g of oxygen?
Answer:
The molecular weight of oxygen = 32.
∴ 32 g oxygen = 1 g mol of oxygen
or, 8 g oxygen \(=\frac{8}{32}\) mol of oxygen
\(=\frac{1}{4}\) mol of oxygen
Again, we know that 1 g mol of oxygen contains 6.023 x1023 molecules.
\(=\frac{1}{4}\) g mol of oxygen contains \(\frac{6.023 \times 10^{23}}{4}\)
or, 1.506 x 1023 molecules,
ie., there are 1.506 x 1023 molecules in 8 g of oxygen.
Question 22. Explain the significance of Avogadro’s number.
Answer:
Avogadro Number (NA): Its significance in Chemistry, Biology, and Physics: Atoms or molecules can’t be seen by the naked eye but can be visualized or felt the amount of something having NA number of atoms or molecules. We can feel how much water is there in 18 g of water and know that it contains 1 mole or NA, the number of water molecules.
In any chemical change, the molecules change their number, volume, and amount
aA + bB → cC + dD
We say that a mole of A reacts with b mole of B to produce c mole of C and d mole of D. If the species are gaseous, then using NA we can express the volume ratio of A, B, C, and D also.
In biological systems, most of the important changes are nothing but chemical changes and the use of NA is equally applicable here. In photosynthesis reach, carbohydrate is produced according to the following reaction :
\(6 \mathrm{CO}_2+6 \mathrm{H}_2 \mathrm{O} \stackrel{\text { light }}{\longrightarrow} \mathrm{C}_6 \mathrm{H}_{12} \mathrm{O}_6+6 \mathrm{O}_2\)The ratio of amounts or volumes of different species can be given by using the mole concept. In physics, say for example, in radioactive decay, the mole concept is important for the same reason.
West Bengal Board Class 9 Physical Science Book Solution
Question 23. Explain Berzilius’s hypothesis based on Dalton’s atomic theory.
Answer:
Explanation of Berzilius’s hypothesis based on Dalton’s atomic theory: Under the same conditions of temperature and pressure, one volume of hydrogen and one volume of chlorine combine to form two volumes of hydrogen chloride gas.
According to Berziliu’s hypothesis, suppose that under the same conditions of temperature and pressure, each volume of gas contains number of atoms. We can say,
n-number of hydrogen atoms + n-number of chlorine atoms
= 2n number of hydrogen chloride atoms
∴ 1 atom of hydrogen + atom of chlorine
= 2 atoms of hydrogen chloride
∴ ½ atom of hydrogen + ½ atom of chloride
= 1 atom of hydrogen chloride
Now, the existence of ½ atom of hydrogen and ½ atom of chlorine is impossible according to
Dalton’s atomic theory, so the hypothesis clashed with the basic concept of atomic theory. Hence, it was rejected by Dalton.
Question 24. Explain Gay-Lussac’s law with the help of Avogadro’s hypothesis.
Answer:
Explanation of Gay-Lussac’s law with the help of Avogadro’s hypothesis : From experimental result it has been found that under the same conditions of temperature and Pressure, one volume of hydrogen combines with one volume of chlorine to produce two volumes of hydrogen chloride gas.
Suppose, under the same conditions of temperature and pressure, each volume of gas contains n-number of molecules.
So, n-number of hydrogen molecules + n-number of chlorine molecules.
= 2 n-number of hydrogen chloride molecules.
∴ 1 molecule of hydrogen + 1 molecule of chlorine
= 2 molecules of hydrogen chloride gas.
∴ ½ molecule of hydrogen +½ molecule of chlorine
= 1 molecule of hydrogen chloride
This is to say,
1 molecule of hydrogen chloride must contain ½ molecule of hydrogen and ½ molecule of chlorine.
This fact does not go against Dalton’s atomic theory. Later on, it is known from Avogadro’s hypothesis that each molecule of hydrogen and chlorine contains two atoms. .
So, 1 atom of hydrogen + 1 atom of chlorine
= 1 molecule of hydrogen chloride gas.
In this way Dalton’s atomic theory and Gay-Lussac’s law were harmonized with the help of Avogadro’s hypothesis.
Wb Class 9 Physical Science Question Answer
Question 25. Prove that hydrogen molecule is diatomic in nature.
Answer:
Proof of diatomic nature of hydrogen molecule: From experimental result we know that under the same conditions of temperature and pressure one volume hydrogen and one volume of chlorine combine chemically to form two volumes of hydrogen chloride gas.
Let us suppose that under the same conditions of pressure and temperature at the time of experiment one volume of hydrogen gas contains n-molecules.
So, according to Avogadro’s hypothesis under the same conditions of temperature and pressure, one volume of chlorine contains n-molecules and two volumes of hydrogen chloride gas contain 2n molecules.
This fact can be represented by the following equation :
n-molecules of hydrogen + n-molecules of chlorine
= 2n molecules of hydrogen chloride gas.
∴ 1 molecule of hydrogen + 1-molecule of chlorine
= 2 molecules of hydrogen chloride gas.
So, 1 molecule of hydrogen chloride
½ molecule of hydrogen + ½ molecule of chlorine.
Now, according to Dalton’s atomic theory an atom is indivisible.
So, one molecule of hydrogen chloride contains at least one atom of hydrogen and one atom
of chlorine. This one atom of hydrogen comes from ½ molecule of hydrogen.
So, one molecule of hydrogen must contain two atoms.
Question 26. What are the differences between molecular weight and acutal weight (mass) of a molecule ?
Answer:
Differences between molecular weight and actual weight (mass) of a molecule :
| Molecular weight | Actual weight (mass) of a Molecule |
| (1) It is a number which represents how many times a molecule of a substance is heavier than ½ th Part of the weight (mass) of carbon (C = 12) | (1) It is the number of times one molecule of a substance is heavier than one atomic mass unit (a.m.u.) which is 1.6603 x10-24 g. |
| (2) It is the ratio of two weights, i.e., a pure number and has no unit. | (2) It represents the actual mass of a molecule and so it is not unitless. |
| (3) It is a number which denotes the actual weight (mass) in gram of 6.023 x 1023or Avogadro’s number of molecules of the substance. | (3) it represents the mass of a single molecule of a substance. |
West Bengal Board Class 9 Physical Science Book Solution
Question 27. Gram-molecular volume of all gases at NTP is 22.4 litres—prove it.
Answer:
If the molecular mass of a gas is M, then we know that the limiting density of the gas =0.0898× \(\frac{M}{2} g\)/litre.
From the definition of limiting density it can be written that mass of 1 litre gas at NTP =0.0898× \(\frac{M}{2} g\)
So, at NTP volume of 0.0898 x g gas = 1 litre.
∴ At NTP volume of 1 g gas \(\frac{2}{M \times 0.0898}\)litre
∴ At NTP volume of M g or 1 gram molecule gas
\(=\frac{2 \times M}{M \times 0.0898}\)=\(\frac{2}{0.0898}\)=22.27 litre
Atomic mass of hydrogen in oxygen scale is 2.016. In that case, calculating in a similar way we can obtain .
Volume of 1 gram-molecule gas at NtP=\(\frac{2.016}{0.0898}\)=22.4litre.
So, at NTP volume of 1 gram-molecule of any gas is 22.4 litre.
Question 28. Deduce the relation between density and vapour density of a gas on the basis of hydrogen.
Answer:
Relation between density and vapour density of a gas on the basis of hydrogen:
Vapour density(D)\(=\frac{\text { Weight of a gas of volume V (at NTP) }}{\text { Weight of } V \text { volume of hydrogen (at NTP) }}\)
∴ \(D\)=\(\frac{\text { Weight of } 1 \text { litre of a gas (at NTP) }}{\text { Weight of } 1 \text { litre of a hydrogen (at NTP) }}\) \(=\frac{\text { Density of gas }(\mathrm{d})}{\text { Density of hydrogen gas }}\)
∴ Density of gas (d) = D × 0.00898
(Density of hydrogen gas= 0.0898)
∴ d= D × 0.089.
Question 29. What is the actual weight of a molecule ? What is the actual weight of an atom ?
Answer:
Actual weight of a molecule : Suppose, the molecular mass of any subsiance = M. So, gram-molecular weight of the substance = M g.
We know that there are present 6.023 x 1023 molecules in gram-molecular weight.
∴ Weight of 6.023 x 1023 molecules = M g.
∴ Weight of 1 molecule \(=\frac{M}{6.023 \times 10^{23}} g\)
Actual weight of an atom :
Suppose, the atomic mass of an element = A
So, gram-atomic weight of the element = A g
We know that there are 6.023x 1023 toms present in gram-atomic weight.
∴ Weight of 6.023 x 1023 atoms = A g
∴ Weight of 1 atom\(=\frac{A}{6.023 \times 10^{23}} g\).
West Bengal Board Class 9 Physical Science Book Solution
Question 30. Deduce the relation between vapour density and the molecular weight of a gas.
Answer:
Relation between vapour density and the molecular weight of a gas : From the definition of vapour density,
D\(=\frac{\text { Weight of } V \text { volume of a gas at NTP }}{\text { Weight of } V \text { volume of hydrogen at NTP }}\)
Suppose ,V volume of the gas contains ‘n’ molecules then by Avogadro’s hypothesis, V volume of hydrogen will also contain ‘n’ molecules of hydrogen.
∴ D\(=\frac{\text { Weight of ‘ } n \text { ‘ molecules of the gas (at NTP) }}{\text { Weight of ‘ } n \text { ‘ molecules of hydrogen gas (at NTP) }}\)
\(=\frac{n \times \text { weight of one molecule of the gas }}{n \times \text { weight of one molecule of hydrogen }}\) \(=\frac{\text { weight of one molecule of the gas }}{\text { weight of one molecule of hydrogen }}\)
\(=\frac{\text { weight of one molecule of the gas }}{\text { weight of } 2 \text { atoms of hydrogen }}\) (Hydrogen is a diatomic element)
∴ D= ½ ×\(=\frac{\text { weight of one molecule of the gas }}{\text { weight of one molecule of hydrogen }}\)
½ × M (M=molecular weight of the gas )
∴ D\(=\frac{M}{2}\)
or, M= 2D
i.e., Molecular weight=2 × its vapour density.
Question 31. Potassium superoxide, KO2 is an air purifier as it reacts with CO2 and releases O2, 4KO2(s) + 2CO2(g) —> 2K2CO3(s) + 3O2(g)
Answer:
Strategy: Find Vm at given (T, P)
Use n\(=\frac{V}{V_m}\) to find n of CO2
Use mKO2 = nKO2 .Mko2
nKO2 is obtained from stoichiometry.
Vm(CO2)\(=\frac{R T}{P}\)=0.0821 293.15/1 l=24.47 l mol-1
nCO2 \(=\frac{V}{V_m}\)\(=\frac{501}{24.47 \mid \mathrm{mol}^{-1}}\)= 2.043 mole.
From stoichimetry,2 mol CO2 reacts with 4 mol KO2
∴ nKO2 \(=\frac{4}{2}\)×nKO2 =2 × 2.043mol=4.086 mol.
Wb Class 9 Physical Science Question Answer
Question 32. What mass of Al is needed to reduce 10.0 kg of Cr(IIl) oxide to produce chromium metal ? \(2 \mathrm{Al}(I)+\mathrm{Cr}_2 \mathrm{O}_3(\mathrm{~s}) \stackrel{\Delta}{\longrightarrow} \mathrm{Al}_2 \mathrm{O}_3(\mathrm{~s})+2 \mathrm{Cr}(I)\)
Answer:
M(Cr2 O3)= 152.0g mol-1 ; 1 mol Cr2 O3=2mol AI; MAI =26.98 g mol-1
Now apply the formula:
\(\mathrm{n}_{\mathrm{c}_2} \mathrm{O}_3\)=\(\frac{10.0 \times 10^3 \mathrm{~g}}{152 \mathrm{gmol}^{-1}}\)
nAI =2×\(\mathrm{n}_{\mathrm{c}_2} \mathrm{O}_3\)
2×65.78mol=131.58.
∴ mAI= nAI×MAI
131.58 mol ×26.98 g mol-1
= 3,550 g
=3.55Kg.
WBBSE Solutions for Class 9 Physical Science And Environment
- Chapter 1 Measurement
- Chapter 2 Force And Motion
- Chapter 3 Matter: Structure And Properties
- Chapter 4 Matter: Atomic Structure; Physical & Chemical Properties of Matter
- Chapter 5. Energy In Action: Work, Power & Energy
- Chapter 6 Heat
- Chapter 7 Sound


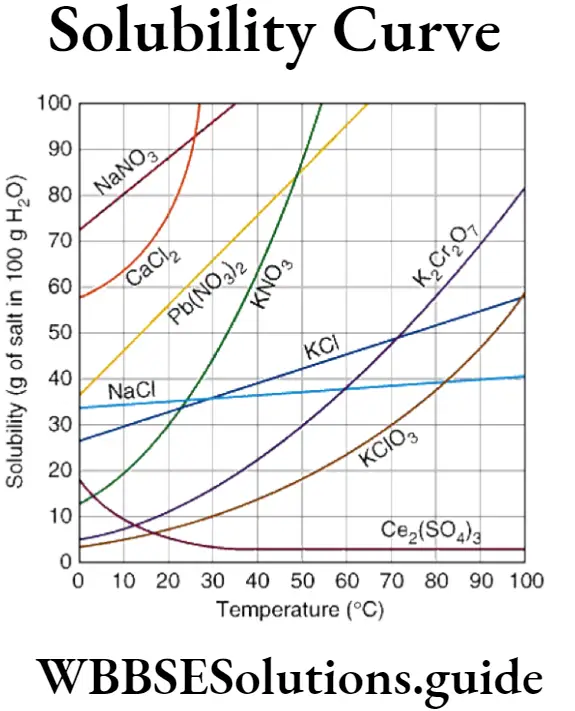
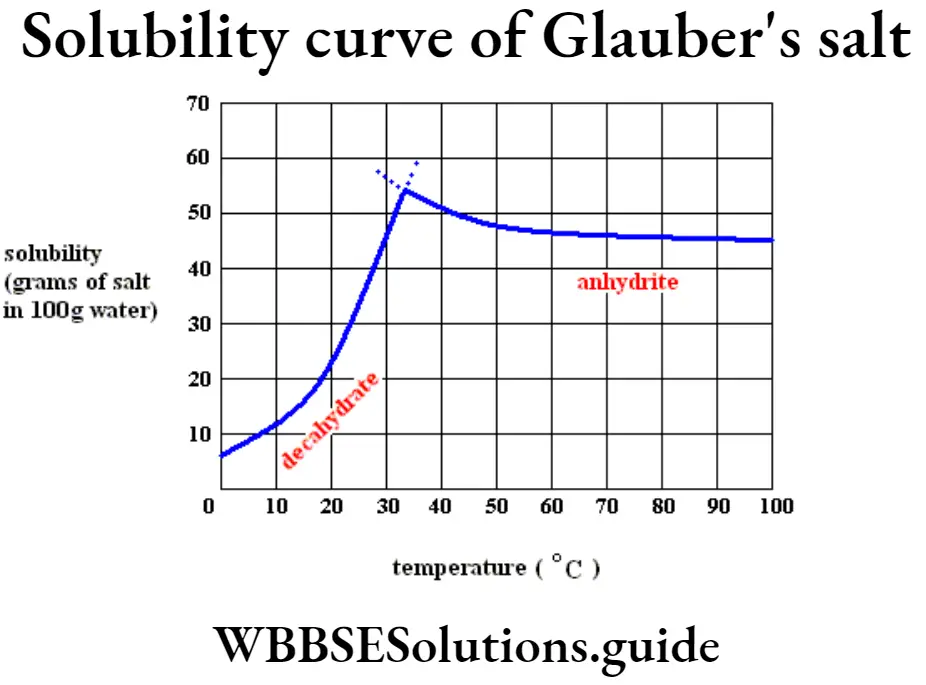 (4) The solubility curve of Ca(OH)2 shows there is a decrease of solubility after attaining certain temperature.
(4) The solubility curve of Ca(OH)2 shows there is a decrease of solubility after attaining certain temperature.








 Inference:
Inference:

 Uses of barometer :
Uses of barometer :

