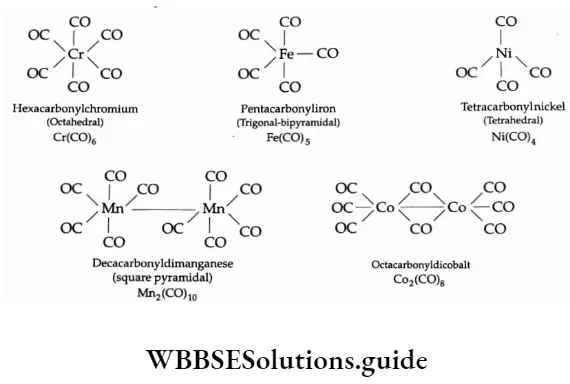
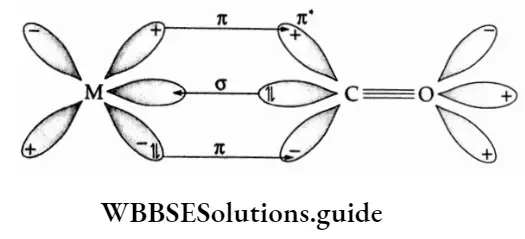
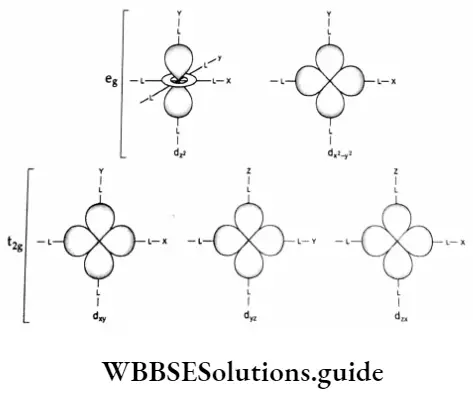
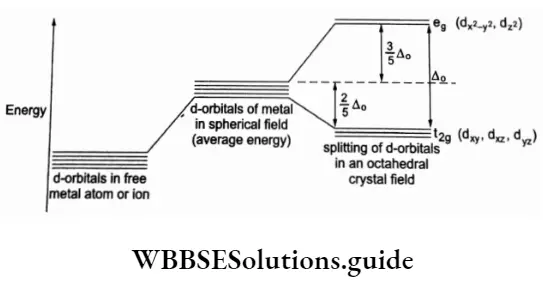
Some Aspects Of The French Revolution Introduction 8 Marks Questions & Answers
Question 1. Why did the Revolution break out in France? What was its significance?
Answer:
The Revolution break out in France Because
(1) New ideas: The French Revolution of 1789 broke out in France because the country was prepared for such a situation. It should be noted that the characteristics of the old Regime were not peculiar to France. Monarchical absolutism, oppression of the peasantry and several other features existed in most of the countries of Western Europe. So there was nothing exceptional in the character or in the extent of the grievances of the French people.
Hence we must not imagine that the people of France suffered more grievously than those of many other lands. What was peculiar to France was the fact that the people there were more enlightened and intelligent and so were more conscious of their sufferings.
That was why the evils of the Old Regime came under greater scrutiny in France than elsewhere. Hence the motive force ‘of the Revolution was not the intensity of the sufferings of the French people, but a new unwillingness to submit to the anomalies of the Old Regime. The change in the angle of vision was largely due to the influence of the writings of the French philosophers.
The material revolution of 1789 was preceded by a revolution in the realm of ideas. Thus intellectual revolt was brought about by a group of French philosophers, of whom the most prominent were Montesquieu, Voltaire and Rousseau. Their writings gave expression to the discontent and indignation which lay latent in the hearts of the French people.
| Class 9 English Bliss | Class 9 Life Science |
| Class 3 English | Class 9 Geography |
| Class 10 Life Science | Class 9 History |
| Class 9 History | Class 9 Maths |
The manifold ills and anomalies from which the-station suffered, coupled with the mistakes of the Government, brought about the catastrophe. Nevertheless, these writers were a powerful factor in the Revolution. Into a soil prepared by social grievances and political abuses, the French philosophers flung broadcast the seed of philosophical speculation.
Their new ideas and theories upset men’s convictions and changed their outlook. Their writings exposed the evils of the Old Regime, focussed public attention upon them, compelled discussion and aroused the passion of the people instilling into them new ideas and doctrines. Thus the French philosophers habituated the people to the destructive criticism of the existing institutions and thereby prepared their minds for a revolutionary “change.
Wbbse Class 9 History Chapter 1 Some Aspects Of The French Revolution Long Questions With Answers
Question 2. What was the condition of France on the eve of the French Revolution of 1789?
Answer:
The condition of France on the eve of the French Revolution of 1789
(1) Evils of Absolutism: The Government of France was a highly centralised monarchy in France since the days of the reign of Louis 14. The representative institutions that France had at one time possessed had either been destroyed or brought under the King’s control. The States General (the feudal Parlement of France) had ceased to exist Hence the King ruled like an absolute monarch. His function was to command the people to obey.
All the functions of the Government being absorbed by the king required a ruler of exceptional ability to carry on the duties of the royal office. Louis 14, whatever might have been his defects, was never wanting in assiduity and anxious care for his country. But his successor, Louis 15 was weak and frivolous, enjoying the sweets of the royal office, while shirking the responsibilities attached to it The consequences of centralised Government under an incapable ruler soon manifested themselves.
The task of governing fell to a greedy horde of courtiers who sacrificed every interest of the state to advance their selfish ends. To the evils of corrupt administration were added the evils of oppression. Anybody might be imprisoned by the mere issue of warrants called letters de cachet, one of the most odious features of the Old Regime. A Government at once corrupt and arbitrary could not but produce widespread discontent and the people remained in sullen silence.
(2) Inequality: The French society was graded from top to bottom and each grade differed from others in legal rights and the enjoyment of powers and privileges. Broadly speaking, it may be said that France was divided into two classes, privileged and unprivileged. The privileged class included the nobility and the higher clergy.
They enjoyed a total or partial exemption from taxation and also had a monopoly of honours and emoluments. The unprivileged class included the bourgeoisie or the middle-class citizens, the labourers and the peasants. They formed what was called the Third Estates. They practically bore the whole burden of taxation and were at the same time excluded from all places of authority.
(3) The Nobility: The feudal nobles had mostly degenerated into a body of greedy courtiers. In feudal times the nobility enjoyed exemption from taxation as well as other feudal privileges in return for services they rendered to the Provincial Government. But as the monarchy became centralised the local services were performed by the Central Government, the nobles being deprived of all local power.
But they continued to enjoy the privileges of feudalism though they were not called upon to perform the duties attached to the system. The absence of duties made the continuance of rights anomalous and irritating. It was this that roused the indignation of the peasants when they were subjected to the feudal exactions of nobles.
(4) The clergy: The clergy along with the nobility formed the whole privileged class. In the Church as elsewhere, we find the same inequality and favouritism. The higher clergy monopolised all the lucrative Church offices but were quite indifferent to their spiritual duties.
They were recruited from the younger sons of the nobility and they conducted themselves as typical men of the world, being devoted to self-advancement and given to the pleasures, intrigues and dissipation of the court On the other hand, the lower clergy, who did the real work of spiritual consolation and instruction, were wretchedly recompensated. They had to work in poverty without any hope of promotion. Hence they formed a discontented class ready to join hands with the common people against the abuse of favouritism and absolutism.
(5) The Third Estate: Below the two privileged orders, there was the vast majority of the population called the Third Estate. It was not a homogeneous body. It comprised the bourgeoisie or the upper middle class, the artisans and the peasants.
(1) The bourgeoisie or the upper middle class formed the well-to-do, intelligent and energetic section of the community. They were practical businessmen who had gotten with wealth a secured monopoly of municipal appointments. Conscious that they were as good as the nobles, they keenly resented the existing system under which they were made to feel in numerous ways their social inferiority. Although their interests differed from those of the other members of the Third Estate, they were a discontented class and wanted political and social reforms.
(2) The artisans and labourers, although belonging to the Third Estate, were much worse off than the bourgeoisie. They were completely at the mercy of the rich middle class which controlled commerce and industry through guilds and similar close corporations.
(3) The condition of the peasantry, which formed by far the largest section of the population, was deplorable in the extreme. The peasant had to pay rent to his feudal lord, tithes to the Church and taxes to the king. The whole burden of taxation fell with a crushing weight upon him, especially because the privileged orders were more or less exempt from taxation.
(6) Inequality of taxation: The system of taxation had almost every conceivable defect. It was unjust, odious and honey-combed with favouritism. The privileged classes being more or less exempt from taxation, the burden fell more heavily on the common people. Thus the rich who could support the state paid the least, while the poor who were already over-burdened were crushed with the weight of taxation. The oppressive nature of taxation was further heightened by the method of its collection.
The state often sold the right of collection to private individuals who paid a lump sum to the Government and then collected the taxes themselves, seeking, of course, to exact as much as possible from the people. It is not unnatural that inequality of taxation and its oppressiveness should prove one of the most decisive causes of the Revolution.
Wbbse French Revolution Class 9 Short Question Answers
Question 3. What were the causes of the French Revolution?
Answer:
The causes of the French Revolution
(1) Introduction: Next to the Reformation, the French Revolution was undoubtedly the greatest event in the history of Europe. It is of such far-reaching importance that the West is still under the influence of the forces, the Revolution let loose — Nationality and Democracy. The death of the Old Regime in France which was honey-combed with abuses of every sort was gone due to a combination of circumstances.
(2) Political Causes :
(1) Arbitrary rule: 1) Absolute divine-right monarchy was the rule all over the continent. The kings of France ruled arbitrarily. They imprisoned people without trial by a mere order, a letter de cachet There was no intellectual or civil liberty in France.
(2) The Palace of Versailles: The kings of France lived isolated twelve miles away from the capital amid the splendours of the palace of Versailles. The magnificent court of the French King was a riot of extravagance and the envy of Europe. It proved to be the veritable nation’s grave.
(3) The rule of weak kings: The kings that claimed absolute rights in France preceding the Revolution were not ‘grand monarchs’ like Louis XIV but weaklings like Louis XV and Louis XVI. They became puppets in the hands of the nobles and favourites. Louis XV was under the evil influence of Madam de Pompadour.
(4) Defective organisation of the State: The Government of France was highly corrupt, complicated and inefficient It was full of disunity, diversity and variety. France was split into forty provinces-‘forty Governments’. Weight and measures had different names in different parts of the country. Something like 285 codes of law prevailed.
(5) Over-centralisation: France was an over-centralised State but lacked unity. Local self-government did not exist in France. The Government was determined and t directed by Versailles which was the source of endless delays. The Central Government was represented by the Intendants on whom depended, in large measure, the happiness or misery of the people.
(6) Army: The French army was greatly reduced in numbers and was influenced by revolutionary ideas.
(7) Foreign policy: France, since the time of Richelieu, followed an ambitious foreign policy. It aimed at the glory of the Bourbons regardless of the French people. The various and costly wars of Louis 14 left France practically a desert.
(3) Social causes: The structure of French society was essentially feudal. It was divided into three classes, rigidly cut off from one another—the Nobles, the Bourgeoisie and the Peasantry. Graded from top to bottom, each social section sharply differed from another in the enjoyment of power and privileges. French society was cut into two distinct classes. One the privileged and the other unprivileged.
While the first included the nobility and the higher clergy, the second comprised the middle-class citizens or bourgeoisie, the labourers and the peasants. The third estate was the convenient name given to the second class. There was a strange relationship between the two classes. While the privileged class was affluent, it paid little to the State treasury, whereas the poor second class was heavily overburdened with taxes.
(1) The Nobility: The Nobles and the upper clergy were the most privileged class. They led lives of luxury in the court of the King and everybody was jealous of them. The nobles enjoyed the privilege of exemption. At the same time, they could levy certain taxes on their Estates. Another feudal privilege was the corvee or feudal labour on roads.
The lord could summon any of his peasants to work at road¬making, without wages. One more privilege was the exclusive right of hunting. The peasant had no right to disturb the game although it was destroying their crops. Thus, aristocratic privilege in France ruined agriculture and trade.
(2) The bourgeoisie: The bourgeoisie was the middle class, and was, like the nobles, small in numbers. But they were educated thinkers and comprised lawyers, physicians, teachers, merchants, bankers, etc. They were rich and intelligent and favoured a political reorganisation and social revolution. It was from this class that the revolutionary leaders sprang – Rousseau, Danton and Robespierre.
(3) The peasantry and the artisans: The peasants and the artisans were the lowest and the largest class. They constituted 4/5 of the entire population. The burden of society fell with a crushing weight upon them. They paid 55% of their earnings to their State. In addition, they had paid tithes to the Clergy, numerous feudal dues and salt and excise duties. In all, they paid 4/5 of their income by way of taxes and were left with only 1/5 to support their families. No wonder, they were the most discontented.
(4) Religious Causes: The Church in France, like the other institutions of the country, was corrupt Privilege and favouritism were found in the Church and elsewhere. The higher clergy resided in court and lived a gay and worldly life. The lower clergy were poorly paid. Hence, they sympathised with plans for reforms and powerfully aided the popular cause in the Revolution.
(5) Taxation: Those who could pay the taxes — the nobility and the royal prince—were exempt from taxation. So the burden fell upon the peasantry and the middle class. The system of taxation varied from region to region and from year to year.
Class 9 History Some Aspects Of The French Revolution Long Answer Questions Wbbse
Question 4. How far was the intellectual movement responsible for the Revolution of France? Or, Can you trace the influence of French philosophers on the course and events between 1789-1795? Or, Critically examine the influence of French philosophy on the causes of the French Revolution.
Answer:
(1) Ideas of the philosophers: The material revolution of France of 1789 was preceded by a revolution in the realm of ideas. Within the French society of discontents were the writers stimulating them, pointing their discontents, dissolving the traditions which alone held them together, voicing their grievances, giving them a lead of faith, for in a land of no Parliament the men of letters had come to be the politicians.
In innumerable ways, they demonstrated the rottenness of French institutions — by satire and wit, criticism and comparison, analogy and innuendo, scientific exposition, sociological theory, and downright abuse. The literature made excellent gunpowder for destroying the State, and never before was a revolution so armed with words and phrases.
Writers of all kinds prepared for the French Revolution. Early in the century, there was Montesquieu. He was not a revolutionary and he was both a Catholic and a monarchist. He criticised the abuses of the church and the despotism of the State. He criticised by satire.
He believed in the British system of Government, and he believed in the principle of liberty and equality which were the safeguards of freedom. In his famous book. ‘The Spirit of Laws’, he analysed the principle of Government and traced the evolution of laws and the Constitution with special reference to the theory of separation of powers.
A contrast to Montesquieu in method and temperament was his more famous junior contemporary Voltaire. He was the most honoured man in Europe in the middle of the 18th century. He was an indefatigable writer-poet, historian, philosopher, dramatist, correspondent and pre-eminently satirist.
He was inspired by a wide philanthropy, a caustic wit and a bitter hostility to the church. But he was not a Democrat. To attack the altar was ultimately to undermine the throne which rested on it, but Voltaire never consciously aimed at the monarchy and he was fully aware of the dangers of popular Government
The authority which Voltaire respected was destroyed by Jean Jacques Rousseau. Rousseau began where Voltaire left off; the latter harnessed the horses of reason, and the former unchained the tigress of emotion. The political influence of Rousseau’s works especially on Social contracts was incalculable not only in France but in all of Europe.
His fundamental theory was that of the inherent virtue of the “natural man” whom civilisation and institutions had corrupted and deprived of his natural rights to liberty and happiness. “Man is born free, but everywhere he is in chains.” According to Rousseau, laws should be the “general expression” of the deep, instinctive conscience of society.
Kings were the creations of an original contract framed by the people for the promotion of their welfare and a sovereign’s title was dependent upon his guardianship of the rights of his subjects. The people had lost their rights, the contract had been violated, and the crown had forfeited the allegiance of the nation. This revolutionary, though unhistorical, idea was destined to destroy the monarchy of France.
Montesquieu, Voltaire and Rousseau were the giants of the age. But there were other thinkers also whose contributions could not be neglected. Denis Diderot was a brilliant and versatile scholar. He organised a team of researchers and with their collaboration published the Encyclopaedia in seventeen volumes. In this book, Diderot collected together all human knowledge including the latest findings. It became a very popular work. In 1765, when it first appeared, four thousand copies were subscribed.
The philosophers directed their search enquiry also in the field of economics. A school of economists called physiocrats preached their theories. They were led by Quesnay. They criticised mercantilism and State regulation of trade and industry which prevailed in France.
They believed that nature was the single source of all wealth. They strongly urged free trade and free circulation of goods without Government interference. They denounced State paternalism of industry and advocated laissez-faire. Thus the French philosophers created a mental revolution by exposing the hollowness of the ancient regime.
Wbbse French Revolution Class 9 Short Question Answers
(2) Nature of the influence of the philosophers: Historians differ about the extent of the influence of the philosophers on the causation of the French Revolution. According to some of them, is a mistake to ascribe the French Revolution to the influence of the writings of these philosophers.
The manifold ills and anomalies from which the nation suffered, coupled with the mistakes of the Government brought about the catastrophe. Nevertheless, these writings were a powerful factor in the Revolution. Into a soil prepared by social grievances ended by political abuses, the French philosophers flung broadcast the seed of philosophical speculation.
Their new ideas and theories upset convictions and changed their outlook. Their writings exposed the evils of the Old Regime focusing public attention upon them, compelled discussion and aroused the passion of the people by instilling into them new ideas and doctrines.
Thus the French philosophers habituated the people to the destructive criticism of the existing institutions and thereby prepared their minds for a revolutionary change. In short, the idea of the philosophers filtered down among the Frenchmen and destroyed their faith in the old order in France.
Wbbse Class 9 History Chapter 1 Long Answer Type Questions With Solutions
Question 5. Describe the work of the National Assembly during the French Revolution.
Answer:
The work of the National Assembly during the French Revolution
(1) Introduction: Louis 16, unable to face the financial crisis in France, was at last forced to summon the States General in May 1789, which had not met for 175 years. A new chapter was opened up in the history of France. The King recalled Necker to head the ministry. The leaders of the Third Estate at that time were Mirabeau, Abbe Seiyes and Lafayette.
(2) The National Assembly: Friction arose between the Third Estate and the King on the question of voting. The nobility and the clergy wanted separate chambers in the assembly while the Third Estate insisted on only one. In June, it took the momentous and revolutionary step of declaring itself the National Assembly.
(3) Tennis Court Oath: At this, the king blocked the entrance of the Assembly Hall with soldiers. Thereupon the members of the Third Estate met at the adjoining Tennis Court and took the famous Tennis Court Oath, never to separate until the Constitution of the kingdom was established.
(4) The Royal Session: In the Royal Session of June 23, 1789, the king ordered the members of the Third Estate ‘to vote by order’. At their refusal, he sent a message demanding their withdrawal. The occasion brought forth.
(5) The National Constituent Assembly: The King yielded to the demands of the Third Estate and he ordered the Nobility and the Clergy to sit with the Third Estate in a single assembly. The National Assembly was now complete. It immediately appointed a committee to frame a Constitution and adopted the title of Constituent Assembly.
(6) The Storming of the Bastille: The King, inspired by the Court, made a second attempt to suppress the Assembly. A considerable number of mercenary soldiers began to appear near Versailles and Paris. On July 11, Necker was suddenly dismissed. The Paris mob became violent and on July 14, the Bastille, the notorious State person was stormed. It was the triumph of liberty. The 14th of July was declared a national holiday and the Bourbon flag was displaced by the new tricolour flag.
(7) Reforms of the National Assembly :
(1) The August Days: The National Assembly in the “August Days” began tearing down the old regime and building up a new order in society, Church and State.
(2) Abolition of Feudalism: On August 4, 1789, a frenzy of generosity seized the Assembly and thirty decrees were passed which brought about an extraordinary social revolution in France. The Feudal dues and tithes were abolished. The guilds with their narrow restrictions were swept away, public offices were thrown open to all. Provinces as well as individuals were awarded equality. Distinctions of the class were abolished and the principle of equality was made the basis of the State.
(3) Declaration of the Rights of Man: The Declaration of the Rights of Man by the Constituent Assembly of France has been regarded as the gospel of modern times. This declaration reflected the influence of the philosophy of Rousseau and borrowed ideas and phrases from the English Bill of Rights and the American Declaration of Independence. It proclaimed the freedom of religion, expression, press, and property, and security and equality before the law. It announced that men are born free and equal in rights.
(4) The Civil Constitution of the Clergy: The Civil Constitution of the Clergy was promulgated by an Act of Assembly. By this new law, the bishops and priests were reduced in number. They were to be chosen by the Electors elected by the people, paid by the State and only nominally subjected to the Pope. All clergymen must take an oath to support this civil Constitution of the clergy. Naturally, such a Constitution was not acceptable to sincere Catholics.
(5) Financial Reforms: The primary function for which the National Assembly was summoned in 1789, was to solve the financial problem. It tried to solve this in two ways — firstly, by issuing paper currency, and secondly, by confiscating church lands. The paper money proved a total failure by 1791. The Assembly did nothing to improve the finances. It left national finance in a worse condition.
(6) Reorganisation of Administration: The old 32 provinces were abolished and France was divided into 83 departments of nearly uniform size, for administration and local Government. The departments were subdivided into Arrondissements, cantons and communes. Local officials were to be elected by the people. A new system of courts with elected judges was established. A highly centralised State became one highly decentralised. The local divisions made by the National Assembly exist in France even to this day.
(8) The Constitution of 1791: A new Constitution was promulgated in 1791 which incorporated Montesquieu’s theory of Separation of Powers. It introduced a Constitutional monarchy in France. The English system of Parliamentary Government was deliberately avoided. The executive or the ministers were separated from the legislature. The King was deprived of the law-making function, but he was given a suspensive veto. The Assembly was to consist only of one House. Its 745 members were to be elected for years indirectly by active citizens or taxpayers. This excluded the poor.
Wbbse French Revolution Class 9 Short Question Answers
Question 6. How far is it true to say that the old regime in France could not fit with the spirit of the time of 1789?
Answer:
With the meeting of the Estates General at Versailles in May 1789, the Ancient Regime came to an end together with a good many institutions and ideas to which Europe had grown accustomed during the previous three hundred years. But it is a paradox that no important people or forces in France of 1789 wanted revolution. Revolutions may begin as wars often begin a lot because people positively want them. They happen because people want other things that, in a certain set of circumstances, implicate them in war or revolution.
There had been growing in Europe, throughout most of the eighteenth century, what has been called the “revolutionary spirit.” This spirit, a spirit of rationalist criticism and resistance to the established powers of the Roman Catholic Church, the absolutist monarchy, and the privileged nobility was fostered particularly by the work of a remarkable series of French thinkers and literary men, the philosophers. The writings of men like Voltaire, Montesquieu, Diderot and Rousseau were widely read throughout Europe, and they became European figures of eminence and influence.
But the connection between their ideas and the outbreak of the revolution in 1789 is somewhat remote and indirect They did not preach revolution and were usually ready to lend support to any absolute monarch who was prepared to patronize them and adopt their teachings.
But no doubt they had fostered a critical and irreverent attitude towards all existing institutions. They made men more ready, when the need arose, to question the whole foundation of the old order. What mattered in 1789 and what made men revolutionary almost despite themselves, was the whole revolutionary situation and in producing that situation the works of the philosophers played no very important role.
Louis XVI won fresh popularity when he made known his intention of summoning the Estates General. His action aroused hopes of liberal and constitutional reforms and just as nobody of importance wanted revolution, so nobody of importance wanted a republic before 1792. Yet the king’s well-received action of summoning the Estates General precipitated revolution.
“The nobles fight, the clergy pray, the people pay” -this feudal idea was openly challenged by the Third Estate, the people, the non-noble and non-clerical class. Feudalism had come to mean social inequality and social privilege, beginning with class feelings and ending in the class war. The summoning of the Estates General suddenly allowed the leaders of the Third Estate to make their social and economic weight politically effective. It was a chance they seized with both hands.
By not only arousing hopes of liberal reform but also drawing together many of the people most eager for an overhand of the social and political system, Louis crystallized the revolutionary situation. It was only after he had forfeited his original public support by his lack of policy, his wavering and disappointing conduct and in the end the open betrayal of his own promises, that republicanism grew. But the King and his ministers were also in dilemma. They could not satisfy the demands of the middle class without destroying the ancient rights of the nobles and the clergy.
They could not do this without changing and challenging the feudal structure of French society. The Divine Right of the king was questioned, his autonomy in jurisdiction was denied and his financial powers were also a subject of criticism. So only a monarch prepared to be a revolutionary could have escaped from the dilemma. That is also a paradox. And it has been aptly remarked, “France presented indeed the combination of grievance and independence of obscurantism and enlightenment of irritation and incentive, which makes the best material for revolution.”
Wbbse Solutions For Class 9 History Chapter 1 Long Questions And Answers
Question 7. Examine the work of the Constituent Assembly in the French Revolution up to 1791.
Answer:
The work of the Constituent Assembly in the French Revolution up to 1791
The Assembly was unaware of the need for rules, it tolerated disorder in its meetings, it laid itself open to pressure from the galleries, and it forbade the King to choose his ministers from among its members — an insane decision which deprived France of a Mirabeau Cabinet. In short, the Assembly wanted parliamentarians without any of the conditions which make parliament possible.
In religious toleration the Constituents went beyond the Declaration on Dec. 27, 1789, protestants were given civic rights which were granted one month later to the Jews in southern France. Freedom of belief, however, did not triumph completely, and public worship remained a Catholic monopoly.
The Declaration recognised all citizens the right to take part in person or through their representatives in the making of laws. But the Constituent Assembly established a wholly representative system, national sovereignty was only exercised when elections were held, and thereafter the people’s delegates wielded unlimited power.
However, the Assembly could not establish a limited monarchy in France. The King was responsible to no one and was inviolable. No measures were taken to govern the case of high treason on his part The King was granted diplomatic initiative and the right to appoint military leaders, ambassadors and six ministers according to his own choice.
In defiance of the separation of powers, he was even given a suspensive veto to be valid for two legislatures over decisions of the Assembly, yet he was denied the exercise of powers and the Legislative Assembly was made master of the State, and the Legislative Assembly was the French bourgeoisie. Thus Constitutional monarchy was a bourgeoisie republic with no real government.
The Constituent Assembly, in effect, completely decentralised the administration of France. Provinces and local communities long ruled by intendants unanimously demanded the end of royal authority. Decree of Dec. 14, 1789, accordingly granted wide powers to municipalities. They were to levy and collect taxes, maintain public order and have the right to requisition troops and proclaim martial law.
Venality in office was forbidden and litigants would not have tolerated royal appointment of justices from the old regions to the new Courts. Judges were, therefore, elected like administrators. Central authority over administrative bodies was almost eliminated. The most the king could do was to suspend them, and the Assembly could reinstate them. By separating civil and criminal justice from administrative functions, an important improvement was made in that sphere.
Yet another great problem was before the Assembly to deal with, it was the reform of the clergy. On July 12, the Civil Constitution of the clergy was passed. The ecclesiastic organisation was adapted to the administrative framework. Each department had a bishop, and each commune had one or more local priests.
They would be elected like other civil servants. The Pope would no longer be able to draw on France for money, his supremacy was recognised but not his jurisdiction. But this arrangement did not satisfy the Pope and a permanent conflict continued with the Pope till the rise of Napoleon the Great
Class 9 History Chapter 1 Important Long Questions With Answers Wbbse
Question 8. Explain the specific features of the Constitutions of France of 1789, 1793 and 1795.
Answer:
The specific features of the Constitutions of France of 1789, 1793 and 1795
(1) Introduction: The deputies of the Legislative Assembly, just after their election, began to consider the new Constitution. They determined on the motion of Lafayette to publish a Statement of general principles, after the example of the American colonists. On August 12, therefore, was issued a Declaration of the Rights of Man, inspired by Geneva and Philadelphia, which proclaimed that all men being born equal should have equal rights.
But the cause of democracy was to suffer dearly for the comprehensiveness of its claims and for the reason it was a mistaken measure of policy. There was administered inevitable contradiction between the Declaration and the New Constitution which led to conflict for five years.
(2) Constitution of 1789: The political framework of the new Constitution was completed by the end of 1789. The legislative power was to be vested in a single chamber, chosen for two years, not by universal suffrage, but by a tax-paying electorate of a little over four million. Thus was the curtain immediately dropped upon the equal rights of man. Only men who paid at least fifty lives in taxes were landed proprietors. The same civil rights were extended to protestants, Jews and along with the abolition of titles, all careers were open to talent
The hereditary monarchy was retained, and the King, who was to adopt the title of King of the French, was to be the head of the army and the administration. He could nominate the highest officials and choose and dismiss ministers. He could coin money, direct the military forces, and if authorised by the Assembly, he could declare war. He was given only a suspensive veto, that is, he could delay the passing of a measure by holding it up until it had been passed by three successive assemblies.
Despite these limitations, the king retained as head of the executive a certain measure of power. In practice it was difficult for him to exercise it He had no taxing power; he could hardly initiate legislation, for none of his ministers could be members of the Assembly—a check upon the executive inspired by Montesquieu’s theory and America’s example.
The National Convention met on September 21, 1792, on the dissolution of the Legislative Assembly. It was summoned to draft a new Constitution, necessitated by the suspension of Louis XVI. Its first duty was to abolish the monarchy and declare France a Republic. It passed a decree of perpetual banishment against the emigres and adopted a revolutionary calendar from the birth of the republic.
In the meantime, Louis was found guilty by a narrow majority on the 18th of January 1793 and executed on the 21st Robespierre, now rapidly advancing to the leadership of the Jacobins, justified the judicial murder of the king. A Revolutionary Tribunal was set up in March 1793 and the Committee of Public Safety in April, and France was in the grip of the Terror for twelve months from September 1793 to September 1794. After the fall of Robespierre, the Jacobin club in Paris was closed in November. The terror was ended.
In 1795, the Republic attempted to establish a settled government in France by promulgating the Constitution of the year. The Executive was vested in a Directory, of five members, and profiting by previous blunders, a two-chamber legislature was set up. But the Directorial Constitution had a weakness of its own. With the help of a young Corsican, the Republic successfully suppressed the insurrection of the 13th Vendemiaire (5th October 1795). But in 1799, the Directory itself succumbed to a coup de tat effected by the gunner who had saved it in 1795.
Some Aspects Of The French Revolution Class 9 Long Questions Solutions
Question 9. Give an account of the Convention. Or, Describe the Reign of Terror during the Revolution.
Answer:
Convention
(1) Introduction: The National Convention was the third Revolutionary Assembly. It was assembled on September 20, 1792. It was in existence for three years. It became the task of the Convention to abolish royalty, establish a republic, consolidate the Revolution in France and wage a successful foreign war. These tasks were accomplished by terror, bloodshed and dictatorship.
(2) Girondins and the Jacobins: The majority of the members of the Convention belonged to the Girondins and the Jacobins. The Girondins and the Jacobins were entirely distinct revolutionary groups. The former was moderate and the latter extreme radicals. It is around the rivalry between the two that the period of the National Convention centres. The Girondins hated the leaders of the Jacobins— Robespierre, Marat and Danton.
(3) Work of the Convention :
(1) Abolition of Kingship: The first act of the Convention was the abolition of monarchy as an institution. It constructed a republican form of Government based on the principle of democracy.
(2) Execution of Louis 16: Louis XVI was brought to trial before the Convention on a charge of treason, found guilty and condemned to death. He was beheaded on January 21, 1793.
(3) The New Republican Army: Under the leadership of Carnot, the Convention inaugurated militarism which was quite novel in the world’s annals. In 1793, by conscription, an army of 7,70,000 was raised. A new group of dashing republican generals like Moreau, Pichegru and Jourdan rose to distinction. On the other hand, the first coalition of the European powers was formed this time and it began to attack the frontiers of France.
(4) Collapse of the Coalition: The country was cleared of foreign enemies and Carnot, the organiser of defence, led the country to victory. The first Coalition against France was dissolved. Spain and Russia made peace with France. Only Britain, Austria and Sardinia remained in arms against France.
(5) Effects: The military success of the Republic was achieved at too great a cost. The National Army became the chief concern and the pride of the revolution arose. It led to a serious peasant revolt in La Vendee to restore the monarchy and the Catholic Church. The revolt was crushed.
(4) Creation of a strong Central government: The Convention established a strong Central Government and powered a policy of terrorism. For this purpose, it created three organs :
(1) The Committee of Public Safety: It consisted of 12 members elected for one month. It discharged an enormous mass of business of every kind hour after hour.
(2) The Committee of General Security: Its business was the police duty of maintaining law and order throughout the country. A multitude of suspected persons was thrown into prison.
(3) The Revolutionary Tribunal: This was an extraordinary criminal court for trying traitors and conspirators rapidly by applying the Law of Suspects. No appeal could be taken from its decisions. Its sentences were always death sentences.
(5) The Reign of Terror-Danton: The creation of the instruments of despotism commenced in France, which is known as the Reign of Terror. Danton was the chief architect of the Reign of Terror in the first stage of the reign. It lasted from the summer of 1793 to that of 1794. The Terror began in Paris and spread to the Provinces. About 5000 in Paris and about 15000 in the Provinces were guillotined. Among those guillotined was Marie Antoinette, the hated ‘Austrian woman’ and wife of Louis 16. The chief spokesman of these atrocities was Danton, the Dictator, and the leader of the Cordelier Club.
All counsels of clemency were counted as treason. The Girondins became the victim of their moderation. Incited by Marat, a Parisian mob expelled the Girondins from the Convention. Many of them were afterwards guillotined. Among them was Madam Roland, the romantic woman leader of the Girondins. The worse ferocities were reserved for La Vendee. The Duke of Orleans who, as Philip Equality voted for the death of his cousin, Louis XVI, was executed.
Many distinguished men like Bailly and Barnaves were killed and Condercit, fleeing from the Terror, committed suicide. The Herbeitits, a group of extremists and socialists who favoured the destruction of all churches and the establishment of atheism, were executed jointly by Danton and Robespierre. Yet all this did not constitute the ‘great Terror’ that came later.
(6) The Great Terror: Robespierre: Danton who began the Reign of Terror himself paid the penalty. He grew sick of bloodshed and advocated moderation. Robespierre, his secret enemy, condemned Danton on a charge of treason and he was guillotined. Robespierre, the leader of the Jacobin Club, became supreme in the Convention and the Committee of the Public Safety with his advent, began what is called the Great Terror.
Day after day, the butchery went on. It brought about the fall of Robespierre, and he like Danton, went to the guillotine. With the death of Robespierre, the terror was gradually abandoned. The ‘Thermidorian Reaction’ set in and a milder regime began.
Wbbse Class 9 History Chapter 1 Essay Type Questions And Answers
Question 10. How do you explain the emergence of the Directory?
Answer:
(1) Introduction: After the end of the reign of Terror, the Convention sat until 1795. It reorganised its committees and readmitted surviving Girondins to positions of power. The Convention gave up attempts to enforce the laws of the Maximum and some of the emigres began to find their way back into France.
Abandoning the draft Constitutions of both of Girondins and the Jacobins, the Convention now drew up a third, which betrayed not only fear of the executive but also fear of the mob. It began with a declaration of duties as well as of rights. The Constitution came into operation in October 1795 and lasted until November 1799.
(2) The Directory: The Directory of the five, which held executive power under this new Constitution, was ill-fated from birth. The men who successively became Directors were, except for the patriotic organising genius of Carnot, disreputable and self-seeking politicians of little ability.
The corrupt leaders of a period when the moral standards of social and political life were exceptionally low-ebb, they presided over the final liquidation of the Revolution, opposed equality to royalist reaction and furthered mob violence, their aim was a Constitutional parliamentary system on a narrow base, moderate in action and so devised as to prevent dictatorship. They succeeded in preventing a repetition of Robespierre’s revolutionary dictatorship. Explanation of how this came about lies in their success in crushing rebellion at home and their failure to produce victory abroad.
At home, the Directory faced its first political crisis with the elections of 1797, which it was expected would result in an anti-Jacobin majority. Only 13 out of 216 retiring members of the Council were returned, a clear enough protest against the failure of the Government to restore French credit and currency and to alleviate the widespread social distress. In September 1797, the Directory forestalled the arrival of the new hostile and royalist majority.
With the help of Napoleon Bonaparte, it expelled the newly elected members from the assembly. By this coup, the directors forfeited their last shreds of legality and henceforth relied more openly on armed force. Bonaparte’s seizure of power was brought one step nearer. The political system was as bankrupt as the treasury. Of the five Directors, Barras and Sieyes were resolved to resort to the ultimate sanction, an open alliance with Napoleon Bonaparte, the most popular personality in France with a victorious army at his command.
In foreign affairs, the army assumed an ever greater role. By the beginning of 1796, France’s only active enemies on land were Austria and Sardinia and at sea Great Britain. The Convention made peace with Holland, Spain and Prussia. By incorporating the former Austrian Netherlands into France in October, it was committed to continuing the war against Austria, which would not accept this loss, while the British refusal to make peace, even after the break up of the first coalition, kept the war at sea.
Peace had also been made with Portugal and the German States, Saxony and the two Houses, with the Italian States of Naples, Parma and the Papacy. By the beginning of 1796, the Directory was able to concentrate all its efforts against Austria. On October 17, 1797, Austria signed the peace of Campo Formio and it was a settlement as characteristic of sly Napoleonic diplomacy as the campaign had been of Napoleonic generalship.
Then Napoleon departed on an expedition to Egypt designed to cut off the British from India. By May 1799, he was severely defeated by Nelson, the British admiral. The campaign produced a second coalition against France. Despite his losses and reverses, he was the only man in France who enjoyed general confidence and in the new conditions of emergency, it was to him that men burned.
(3) Unpopularity of the Directory: The tragedy of the Egyptian campaign cast no shadow on the fortunes of Bonaparte. A new Coalition had been formed against France in his absence, and the French were driven out of Italy and France itself was threatened with invasion. The Directory was discredited because of its corruption, incompetence and blunders.
When Bonaparte landed at Frejus, a thrill of delight and relief passed through France. Every peasant enquired about the news of General Bonaparte, but no one enquired after the Directory.
(4) Coup d’etat of Bonaparte : On November 9-10, 1799, Napoleon, assisted by Abbe Sieyes, successfully overthrew the Directory, an incident known as the Coup d’etat of Brumaire. A provisional consulate was appointed and a new Constitution was drawn up for France with Napoleon as First Consul and Ducos and Abbe Sieyes being the other Consuls.


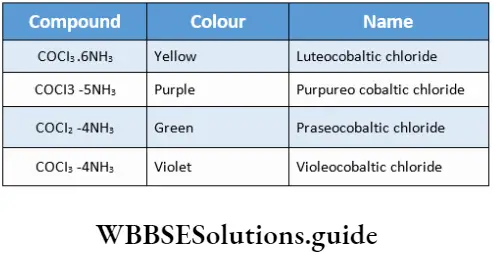
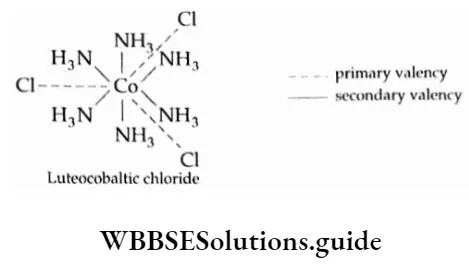
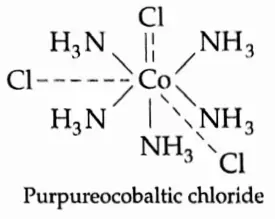
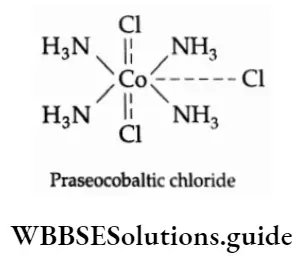

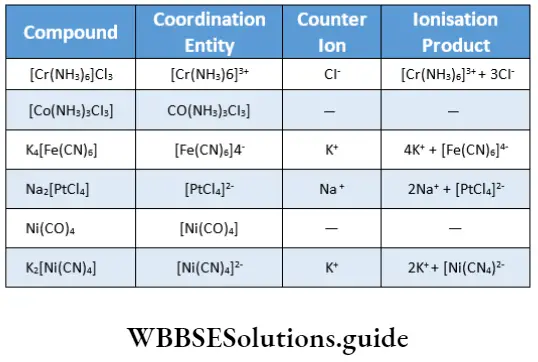
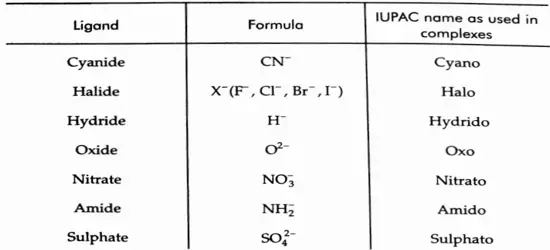

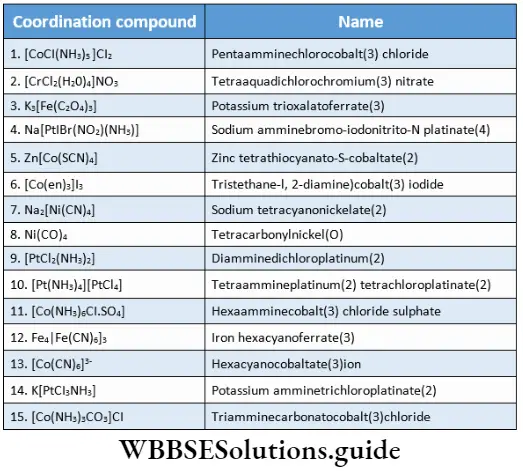
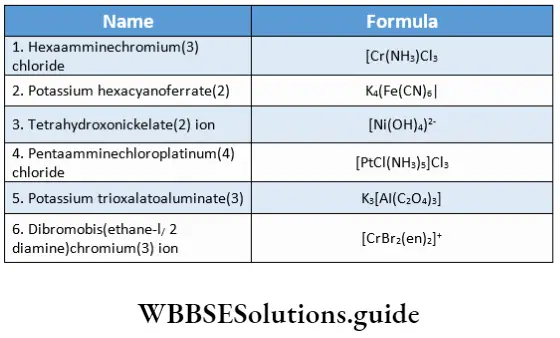

![Coordination Compounds and Organometallics The cis and trans isomers of the square-planar [Ma2b2] type and The cis and trans isomers](https://wbbsesolutions.guide/wp-content/uploads/2023/10/Coordination-Compounds-and-Organometallics-The-cis-and-trans-isomers-of-the-square-planar-Ma2b2-type-and-The-cis-and-trans-isomers.png)